OstiLox Oral Suspension 1.5 mg/mL
This treatment applies to the following species: Company: VetOne
Company: VetOne
(meloxicam oral suspension)
Professional Insert
ANADA 200-497, Approved by FDA
Non-steroidal anti-inflammatory drug for oral use in dogs only
OstiLox Oral Suspension 1.5 mg/mL Caution
Federal law restricts this drug to use by or on the order of a licensed veterinarian.
WarningRepeated use of meloxicam in cats has been associated with acute renal failure and death. Do not administer additional injectable or oral meloxicam to cats. See Contraindications, Warnings, and Precautions for detailed information. |
Description
Meloxicam is a non-steroidal anti-inflammatory drug (NSAID) of the oxicam class. Each milliliter of OstiLox™ Oral Suspension contains meloxicam equivalent to 1.5 milligrams and sodium benzoate (1.5 milligrams) as a preservative. The chemical name for Meloxicam is 4-Hydroxy-2-methyl-N-(5-methyl-2-thiazolyl) -2H-1,2-benzothiazine-3-carbox-amide-1, 1-dioxide. The formulation is a yellowish viscous suspension.
OstiLox Oral Suspension 1.5 mg/mL Indications
OstiLox Oral Suspension is indicated for the control of pain and inflammation associated with osteoarthritis in dogs.
Dosage and Administration
Always provide client information sheet with prescription. Carefully consider the potential benefits and risk of OstiLox Oral Suspension and other treatment options before deciding to use OstiLox Oral Suspension. Use the lowest effective dose for the shortest duration consistent with individual response. OstiLox Oral Suspension should be administered initially at 0.09 mg/lb (0.2 mg/kg) body weight only on the first day of treatment. For all treatments after day 1, OstiLox Oral Suspension should be administered once daily at a dose of 0.045 mg/lb (0.1 mg/ kg). The syringes are calibrated to deliver the daily maintenance dose in lbs. Because the first dose (0.2 mg/kg) is two times the amount of the daily maintenance dose (0.1 mg/kg), two syringes containing the 0.1 mg/kg dose should be administered at the first dose.
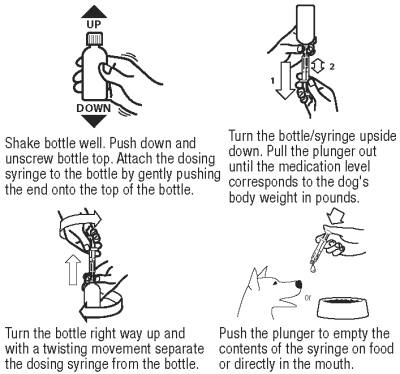
Directions for Administration:
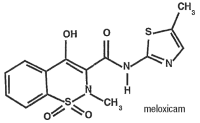
OstiLox Oral Suspension is packaged with 2 sizes of dosing syringes. The small syringe is calibrated in 1-lb increments for use in dogs under 30 lbs. The large syringe is calibrated in 5-lb increments (up to 160 lbs.) and should be used for dosing dogs that are 30 lbs and over. Only administer OstiLox with the provided syringes. The container should never be used as a dropper bottle for administration of OstiLox.
Dogs Under 30 Lbs (13.6 Kg)
Shake well before use, then remove cap. OstiLox Oral Suspension can be given either mixed with food or placed directly into the mouth. Particular care should be given with regard to the accuracy of dosing. To prevent accidental overdosing of small dogs, only use the small dosing syringe. The large syringe provided should not be used to measure doses for dogs weighing less than 30 lbs (13.6 kg). For dogs under 30 lbs, use the small dosing syringe provided in the package (see dosing procedure below). The small dosing syringe fits onto the bottle and has dosing marks in 1-lb increments, designed to deliver the daily maintenance dose of 0.045 mg/lb (0.1 mg/kg). For dogs between 1 - 29 lbs, OstiLox can be given using the marks on the small dosing syringe. When using the small dosing syringe, the dog’s weight should be rounded down to the nearest 1-lb increment. Replace and tighten cap after use.
Dogs 30 Lbs (13.6 Kg) And Over
Shake well before use, then remove cap. OstiLox may be either mixed with food or placed directly into the mouth. Particular care should be given with regard to the accuracy of dosing. To prevent accidental overdosing of small dogs, do not use the large syringe in animals weighing less than 30 pounds.
For dogs 30 lbs or greater, the large dosing syringe provided in the package should be used (see dosing procedure below). The large dosing syringe fits onto the bottle and has dosing marks in 5-lb increments (up to 160 lbs), designed to deliver the daily maintenance dose of 0.045 mg/lb (0.1 mg/kg). When using the large syringe, the dog’s weight should be rounded down to the nearest 5-lb increment. Replace and tighten cap after use.
Contraindications
Dogs with known hypersensitivity to meloxicam should not receive OstiLox Oral Suspension. Do not use OstiLox Oral Suspension in cats. Acute renal failure and death have been associated with the use of meloxicam in cats.
Warnings
Not for use in humans. Keep this and all medications out of reach of children. Consult a physician in case of accidental ingestion by humans. For oral use in dogs only.As with any NSAID all dogs should undergo a thorough history and physical examination before the initiation of NSAID therapy.
Appropriate laboratory testing to establish hematological and serum biochemical baseline data is recommended prior to and periodically during administration. Owner should be advised to observe their dog for signs of potential drug toxicity and be given a client information sheet about OstiLox.
Precautions
The safe use of OstiLox Oral Suspension in dogs younger than6 months of age, dogs used for breeding, or in pregnant or lactating dogs has not been evaluated. Meloxicam is not recommended for use in dogs with bleeding disorders, as safety has not been established in dogs with these disorders. As a class, cyclo-oxygenase inhibitory NSAIDs may be associated with gastrointestinal, renal and hepatic toxicity. Sensitivity to drug-associated adverse events varies with the individual patient.
Dogs that have experienced adverse reactions from one NSAID may experience adverse reactions from another NSAID. Patients at greatest risk for renal toxicity are those that are dehydrated, on concomitant diuretic therapy, or those with existing renal, cardiovascular, and/or hepatic dysfunction. Concurrent administration of potentially nephrotoxic drugs should be carefully approached. NSAIDs may inhibit the prostaglandins that maintain normal homeostatic function. Such anti-prostaglandin effects may result in clinically significant disease in patients with underlying or pre-existing disease that has not been previously diagnosed. Since NSAIDs possess the potential to induce gastrointestinal ulcerations and/or perforations, concomitant use with other anti-inflammatory drugs, such as NSAIDs or corticosteroids, should be avoided. If additional pain medication is needed after administration of the total daily dose of OstiLox Oral Suspension, a non-NSAID or non-corticosteroid class of analgesia should be considered. The use of another NSAID is not recommended. Consider appropriate washout times when switching from corticosteroid use or from one NSAID to another in dogs. The use of concomitantly protein-bound drugs with OstiLox Oral Suspension has not been studied in dogs. Commonly used protein-bound drugs include cardiac, anticonvulsant and behavioral medications. The influence of concomitant drugs that may inhibit metabolism of OstiLox Oral Suspension has not been evaluated. Drug compatibility should be monitored in patients requiring adjunctive therapy.
Adverse Reactions
Field safety was evaluated in 306 dogs. Based on the results of two studies, GI abnormalities (vomiting, soft stools, diarrhea, and inappetance) were the most common adverse reactions associated with the administration of meloxicam. The following table lists adverse reactions and the numbers of dogs that experienced them during the studies. Dogs may have experienced more than one episode of the adverse reaction during the study.|
Adverse Reactions Observed During Two Field Studies |
||
|
Clinical Observation |
Meloxicam (n = 157) |
Placebo (n = 149) |
|
Vomiting |
40 |
23 |
|
Diarrhea/Soft Stool |
19 |
11 |
|
Bloody Stool |
1 |
0 |
|
Inappetance |
5 |
1 |
|
Bleeding gums after dental procedure |
1 |
0 |
|
Lethargy/Swollen Carpus |
1 |
0 |
|
Epiphora |
1 |
0 |
In foreign suspected adverse drug reaction (SADR) reporting over a 9 year period, incidences of adverse reactions related to meloxicam administration included: auto-immune hemolytic anemia (1 dog), thrombocytopenia (1 dog), polyarthritis (1 dog), nursing puppy lethargy (1 dog), and pyoderma (1 dog).
Post-Approval Experience (Rev. 2010): The following adverse events are based on post-approval adverse drug experience reporting. Not all adverse reactions are reported to FDA/CVM. It is not always possible to reliably estimate the adverse event frequency or establish a causal relationship to product exposure using these data. The following adverse events are listed in decreasing order of frequency by body system.
Gastrointestinal: vomiting, anorexia, diarrhea, melena, gastrointestinal ulceration
Urinary: azotemia, elevated creatinine, renal failure
Neurological/Behavioral: lethargy, depression
Hepatic: elevated liver enzymes
Dermatologic: pruritus
Death has been reported as an outcome of the adverse events listed above. Acute renal failure and death have been associated with use of meloxicam in cats. To report suspected adverse reactions, to obtain a Safety Data Sheet, or for technical assistance, call 1-866-591-5777. For a complete listing of adverse reactions for meloxicam reported to the CVM see: http://www.fda.gov/ADEreports.
Information For Dog Owners
OstiLox, like other drugs of its class, is not free from adverse reactions. Owners should be advised of the potential for adverse reactions and be informed of the clinical signs associated with drug intolerance. Adverse reactions may include vomiting, diarrhea, decreased appetite, dark or tarry stools, increased water consumption, increased urination, pale gums due to anemia, yellowing of gums, skin or white of the eye due to jaundice, lethargy, incoordination, seizure, or behavioral changes. Serious adverse reactions associated with this drug class can occur without warning and in rare situations result in death (see Adverse Reactions). Owners should be advised to discontinue OstiLox and contact their veterinarian immediately if signs of intolerance are observed.
The vast majority of patients with drug related adverse reactions have recovered when the signs are recognized, the drug is withdrawn, and veterinary care, if appropriate, is initiated. Owners should be advised of the importance of periodic follow up for all dogs during administration of any NSAID.
Clinical Pharmacology
Meloxicam has nearly 100% bioavailability when administered orally with food. The terminal elimination half-life after a single dose is estimated to be approximately 24 hrs (+/-30%) regardless of route of administration. There is no evidence of statistically significant gender differences in drug pharmacokinetics. Drug bioavailability, volume of distribution, and total systemic clearance remain constant up to 5 times the recommended dose for use in dogs. However, there is some evidence of enhanced drug accumulation and terminal elimination half-life prolongation when dogs are dosed for 45 days or longer.Peak drug concentrations can be expected to occur within about 7.5 hrs after oral administration. Corresponding peak concentration is approximately 0.464 mcg/mL following a 0.2 mg/kg oral dose. The drug is 97% bound to canine plasma proteins.
Effectiveness
The effectiveness of meloxicam was demonstrated in two field studies involving a total of 277 dogs representing various breeds, between six months and sixteen years of age, all diagnosed with osteoarthritis. Both of the placebo-controlled, masked studies were conducted for 14 days. All dogs received 0.2 mg/kg on day 1. All dogs were maintained on 0.1 mg/kg oral meloxicam from days 2 through 14 of both studies. Parameters evaluated by veterinarians included lameness, weight-bearing, pain on palpation, and overall improvement. Parameters assessed by owners included mobility, ability to rise, limping, and overall improvement. In the first field study (n=109), dogs showed clinical improvement with statistical significance after 14 days of meloxicam treatment for all parameters. In the second field study (n=48), dogs receiving meloxicam showed a clinical improvement after 14 days of therapy for all parameters; however, statistical significance was demonstrated only for the overall investigator evaluation on day 7, and for the owner evaluation on day 14.Safety:
Six Week Study
In a six week target animal safety study, meloxicam was administered orally at 1, 3, and 5X the recommended dose with no significant clinical adverse reactions. Animals in all dose groups (control, 1, 3 and 5X the recommended dose) exhibited some gastrointestinal distress (diarrhea and vomiting). No treatment-related changes were observed in hematological, blood chemistry, urinalysis, clotting time, or buccal mucosal bleeding times. Necropsy results included stomach mucosal petechiae in one control dog, two dogs at the 3X and one dog at the 5X dose. Other macroscopic changes included areas of congestion or depression of the mucosa of the jejunum or ileum in three dogs at the 1X dose and in two dogs at the 5X dose. Similar changes were also seen in two dogs in the control group. There were no macroscopic small intestinal lesions observed in dogs receiving the 3X dose. Renal enlargement was reported during the necropsy of two dogs receiving the 3X and two receiving the 5X dose.
Microscopic examination of the kidneys revealed minimal degeneration or slight necrosis at the tip of the papilla in three dogs at the 5X dose. Microscopic examination of the stomach showed inflammatory mucosal lesions, epithelial regenerative hyperplasia or atrophy, and submucosal gland inflammation in two dogs at the recommended dose, three dogs at the 3X and four dogs at the 5X dose. Small intestinal microscopic changes included minimal focal mucosal erosion affecting the villi, and were sometimes associated with mucosal congestion. These lesions were observed in the ileum of one control dog and in the jejunum of one dog at the recommended dose and two dogs at the 5X dose.
Six Month Study
In a six month target animal safety study, meloxicam was administered orally at 1, 3, and 5X the recommended dose with no significant clinical adverse reactions. All animals in all dose groups (controls, 1, 3, and 5X the recommended dose) exhibited some gastrointestinal distress (diarrhea and vomiting). Treatment related changes seen in hematology and chemistry included decreased red blood cell counts in seven of 24 dogs (four 3X and three 5X dogs), decreased hematocrit in 18 of 24 dogs (including three control dogs), dose-related neutrophilia in one 1X, two 3X and three 5X dogs, evidence of regenerative anemia in two 3X and one 5X dog. Also noted were increased BUN in two 5X dogs and decreased albumin in one 5X dog. Endoscopic changes consisted of reddening of the gastric mucosal surface covering less than 25% of the surface area. This was seen in three dogs at the recommended dose, three dogs at the 3X dose and two dogs at the 5X dose.
Endoscopic changes consisted of reddening of the gastric mucosal surface covering less than 25% of the surface area. This was seen in three dogs at the recommended dose, three dogs at the 3X dose and two dogs at the 5X dose.
Two control dogs exhibited reddening in conjunction with ulceration of the mucosa covering less than 25% of the surface area.
Gross gastrointestinal necropsy results observed included mild discoloration of the stomach or duodenum in one dog at the 3X and in one dog at the 5X dose. Multifocal pinpoint red foci were observed in the gastric fundic mucosa in one dog at the recommended dose, and in one dog at the 5X dose.
No macroscopic or microscopic renal changes were observed in any dogs receiving meloxicam in this six month study. Microscopic gastrointestinal findings were limited to one dog at the recommended dose, and two dogs at the 3X dose. Mild inflammatory mucosal infiltrate was observed in the duodenum of one dog at the recommended dose. Mild congestion of the fundic mucosa and mild myositis of the outer mural musculature of the stomach were observed in two dogs receiving the 3X dose.
How Supplied
OstiLox Oral Suspension 1.5 mg/mL: 10, 32 and 100 mL bottles with small and large dosing syringes.
Storage
Store at controlled room temperature 68-77°F (20-25°C). Excursions permitted between 59°F and 86°F (15°C and 30°C). Brief exposure to temperature up to 104° F (40°C) may be tolerated provided the mean kinetic temperature does not exceed 77°F (25°C); however such exposure should be minimized.U.S. Patent No. 9,399,013
Manufactured for: MWI, Boise, ID 83705
www.VetOne.net
Made in the UK.
Manufactured by: Norbrook Laboratories Limited, Newry, Co. Down, BT35 6PU, Northern Ireland
000000I01
Label Rev. 04/18
|
Net Contents: |
NDC |
|
|
|
10 mL |
13985-613-10 |
V1 501081 |
|
|
32 mL |
13985-613-32 |
V1 501082 |
|
|
100 mL |
13985-613-01 |
V1 501083 |
Rev. 04/18 020684L03 |
|
2 x 100 mL |
13985-613-02 |
V1 502083 |
Iss. 08/18 6220420C01 |
CPN: 1315199.1
Distributed by MWI Animal Health
3041 W. PASADENA DR., BOISE, ID, 83705
| Customer Service: | 888-694-8381 | |
| Toll-Free: | 800-824-3703 | |
| Website: | www.mwiah.com |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". DVMetrics assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the DVMetrics service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-08-27
