Nobivac Intra-Trac3
This treatment applies to the following species:CANINE ADENOVIRUS TYPE 2-PARAINFLUENZA-BORDETELLA BRONCHISEPTICA VACCINE
Modified Live Virus and Avirulent Live Culture
FOR ANIMAL USE ONLY
IN THE ABSENCE OF A VETERINARIAN-CLIENT-PATIENT RELATIONSHIP, FEDERAL REGULATIONS PROHIBIT THE RELABELING, REPACKAGING, RESALE OR REDISTRIBUTION OF THE INDIVIDUAL CONTENTS OF THIS PACKAGE.
THIMEROSAL FREE
DIRECTIONS FOR USE - PLEASE READ CAREFULLY
Nobivac Intra-Trac3 Indications
This product has been shown to be effective for vaccination of healthy dogs 3 weeks of age or older against canine adenovirus type 2, canine parainfluenza virus and Bordetella bronchiseptica. Duration of Immunity for Bordetella bronchiseptica is at least 1 year. Duration of Immunity for canine adenovirus type 2 and canine parainfluenza has not been established. To review efficacy and safety data, go to productdata.aphis.usda.gov.
When To Vaccinate
Vaccinate dogs at 3 weeks of age or older. Historically, annual revaccination has been recommended for this product. The need for this booster has not been established. For more information on revaccination frequency consult your veterinarian or the manufacturer.
PREPARATION OF THE VACCINE
1. Rehydrate live vaccine to desired volume with the accompanying Sterile Diluent.*
2. Draw the vaccine back into the syringe.
3. Remove needle from syringe.
4. Apply nasal applicator tip to syringe.
Nobivac Intra-Trac3 Dosage And Administration
1 Dose - *Standard dose is 1 mL administered intranasally. Vaccine may be rehydrated with less than 1 mL of diluent to minimize discomfort when vaccinating small dogs and puppies. For one 1 mL dose, rehydrate vaccine with 1 mL sterile diluent or for one 0.5 mL dose, rehydrate vaccine with 0.5 mL sterile diluent. Instill the rehydrated vaccine into one or both nostrils of dogs as illustrated. rehydrate vaccine with 1 mL sterile diluent or for one 0.5 mL dose, rehydrate vaccine with 0.5 mL sterile diluent. Instill the rehydrated vaccine into one or both nostrils of A dogs as illustrated below.
5 Dose - *Standard dose is 1 mL administered intranasally. Vaccine may be rehydrated with less than 5 mL of diluent to minimize discomfort when vaccinating small dogs and puppies. For five 1 mL doses, rehydrate vaccine with 5 mL sterile diluent or for five 0.5 mL doses, rehydrate vaccine with 2.5 mL sterile diluent. Instill the rehydrated vaccine into one or both nostrils of five dogs as illustrated below.
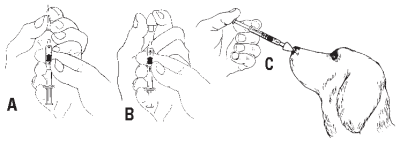
Nobivac Intra-Trac3 Caution
1. THIS PRODUCT IS DESIGNED FOR INTRANASAL USE ONLY WITH THE ENCLOSED APPLICATORS. Systemic reactions resulting from inadvertent intramuscular or subcutaneous injection have been reported. Symptoms may include vomiting, diarrhea, lethargy, inappetence, jaundice and death associated with liver failure. Localized tissue necrosis at the injection site has also been reported. If inadvertent injection occurs, monitor the dog closely. Supportive therapy including IV fluids and treatment with gentamicin, tetracycline, trimethoprim/sulfa, or amoxicillin/clavulanic acid may be indicated. If anaphylactoid reaction occurs, use epinephrine.
2. Post vaccinal reactions consisting of mild canine cough syndrome may occur following use of this vaccine.
3. For animal use only. Store at 2° - 8°C (35° - 46°F). Do not freeze. Do not mix with other products, except as specified on the label. This product has not been tested in pregnant bitches. Inactivate unused contents before disposal. In case of human exposure, contact a physician.
4. This vaccine contains penicillin, streptomycin and nystatin as preservatives.
FOR INTRANASAL USE ONLY
Intervet Inc. d/b/a Merck Animal Health
Omaha, NE 68103 USA
VLN 165A/PCN 12X1.20
1 800 224-5318 (USA)
For patent information:
http://www.merck.com/product/patent/home.html
|
|
Code |
|
|
2x5 DOSE - VARIED DOSE VOLUME |
065314 |
368402-04, 374990-04 |
|
25x1 DOSE - VARIED DOSE VOLUME |
065313 |
325630-04 |
|
150x1 DOSE - VARIED DOSE VOLUME |
065310 |
301148-04, 323622-04 |
CPN: 1047240.7
Intervet Inc.
126 E. LINCOLN AVENUE, PO BOX 2000, Rahway, NJ, 07065
| Customer Service: | 800-521-5767 | |
| Technical Service (Companion Animal): | 800-224-5318 | |
| Technical Service (Livestock): | 800-211-3573 | |
| Website: | www.merck-animal-health-usa.com |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". DVMetrics assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the DVMetrics service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-08-27
