FLEXParvoPRRS
This treatment applies to the following species:Porcine Reproductive & Respiratory Syndrome-Parvovirus Vaccine
Respiratory Form, Modified Live Virus, Killed Baculovirus Vector
This product has been shown to be effective for the vaccination of healthy swine 6 months of age or older against the reproductive failure caused by porcine parvovirus (PPV) and reproductive disease caused by Porcine Reproductive and Respiratory Syndrome (PRRS) virus. The duration of immunity is at least 4 months for the reproductive form of PRRS virus. The duration of immunity has not been determined for porcine parvovirus. For more information regarding efficacy and safety data go to productdata.aphis.usda.gov.
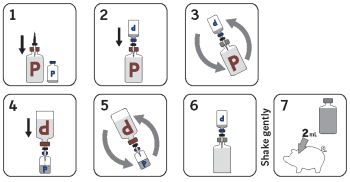
Directions and dosage: Rehydrate the vaccine by adding a portion of the contents of the ReproCyc® ParvoFLEX vial to the Ingelvac PRRSFLEX® MLV vaccine vial. Shake well and transfer the reconstituted suspension back into the ReproCyc® ParvoFLEX vial. Mix with the remaining ReproCyc® ParvoFLEX contents to complete the total volume. Shake well and use immediately. Using aseptic technique, inject a single 2 mL dose of vaccine intramuscularly. Administer a second 2 mL intramuscular dose of ReproCyc® ParvoFLEX 3 weeks later. For more information on revaccination frequency, consult your veterinarian.
Precautions
Store out of direct sunlight at 35 - 46°F (2 - 8°C). Do not freeze. Use entire contents when first opened. Do not vaccinate within 21 days before slaughter. Do not mix with other products, except as specified on the carton. In case of anaphylactoid reaction, administer epinephrine. For use in PRRS virus positive herds only. Do not vaccinate breeding age boars. PRRS vaccine virus may be shed and transmitted to other populations of swine which are in contact with vaccinated swine. The duration of potential vaccine virus transmission may vary. Use of the vaccine in herds intended to remain PRRS virus seronegative is contraindicated. Introduction of vaccinated pigs into herds intended to remain PRRS virus seronegative is contraindicated. Vaccine is safe in pregnant swine. Specific herds may be particularly sensitive. Fetal health risks associated with the vaccination of pregnant animals with this vaccine cannot be unequivocally determined during clinical trials conducted for licensure. Appropriate strategies to address the risks associated with vaccine use in pregnant animals should be discussed with a veterinarian. In case of human exposure, contact a physician. Inactivate unused contents before disposal.Preservative: Neomycin
Boehringer Ingelheim Animal Health USA Inc., St. Joseph, MO 64506
Phone: 1 (888) 637-4251
VLN/PCN 124/1P29.R0
|
50 Doses/Rehydrate to 100 mL |
124600-03 |
CPN: 1028384.1
3239 SATELLITE BLVD., BLDG 500, DULUTH, GA, 30096
| Telephone: | 800-325-9167 | |
| Customer Service: | 888-637-4251 | |
| Technical Service: | 888-637-4251 | |
| Fax: | 816-236-2717 | |
| Website: | www.boehringer-ingelheim.com |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". DVMetrics assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the DVMetrics service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-08-27
