DiroCHEK
This treatment applies to the following species:HEARTWORM ANTIGEN TEST KIT
For the detection of adult Dirofilaria immitis antigen in canine and feline plasma or serum.
General Information And Intended Uses
The reaction wells are coated with antibodies directed against D. immitis antigen. Another antibody is labeled with the enzyme horseradish peroxidase. Any antigen present in the specimen (plasma or serum) is bound by the antibody coated well and the enzyme-linked antibody to form a specific complex. Any free enzyme-linked antibody is washed away and a chromogenic substrate is added. In the absence of D. immitis antigen, no color change will be observed. The development of a blue color specifically indicates the presence of D. immitis antigen and heartworms.
DiroCHEK™ is highly specific, sensitive and simple to use. Test results can be obtained in 15 minutes.
Kit Composition And Conservation
Contains materials sufficient to test 48 to 140 samples.
|
ITEM |
REAGENT NATURE |
AMOUNT |
RECONSTITUTION AND CONSERVATION |
|
A |
Anti-D. immitis Coated Wells |
144 wells |
Ready to use. |
|
CONTROL + |
Positive Control; preserved with Gentamicin Sulfate and Amphotericin B |
2.5 mL |
Ready to use. Red Cap. |
|
CONTROL - |
Negative Control; preserved with Gentamicin Sulfate and Amphotericin B |
2.5 mL |
Ready to use. Gray Cap. |
|
1 |
Conjugate; preserved with Gentamicin Sulfate |
7.0 mL |
Ready to use. Blue Cap. |
|
2 |
Substrate Buffer* |
2 x 7.0 mL |
Ready to use. Purple Cap. |
|
|
Precision Pipetter |
1 |
|
|
|
Pipette Tips |
150 |
|
|
|
Well holder |
|
|
Store all reagents provided in the kit at 2 - 8 °C. Reagents should not be frozen.
*1,2-propylene Carbonate 5-10%, N-methyl-2-pyrrolidone 5-10%, 2,2-oxybisethanol diethylene glycol 1-5%
REAGENTS REQUIRED TO PERFORM 48 TESTS
a) 48 Anti-D. immitis Coated Wells
b) 1.0 mL Positive Control
c) 1.0 mL Negative Control
d) 2.5 mL Conjugate
e) 1 x 7.0mL Substrate Buffer
EQUIPMENT AND MATERIALS REQUIRED BUT NOT PROVIDED
a) Marking Pen
b) Deionized or distilled water
c) Wash Bottle
d) Timer
WARNINGS TO THE USERS OF REAGENTS AND ANTIBODY COATED MICROPLATES
● Handle all reagents and samples as biohazardous material. It is recommended to dispose of reagents and contaminated material according to the applicable regulations.
● Wear suitable protective clothing.
● Irritating to skin and eyes. Keep all reagents away from skin and eyes. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
● Take care not to contaminate any test reagents with samples or bacterial agents.
● The best results are achieved by following the protocols described below, using good, safe laboratory techniques.
● Do not use this kit or any of its components after the expiration date and do not intermix components from different serial numbers
● Do not expose kit to direct sunlight
● Follow instructions exactly. Improper washing or contamination of reagents may produce nonspecific color development.
● NEVER PIPETTE BY MOUTH. Harmful if swallowed.
● NOTE: Allow all components to come to 21 - 25 °C before starting.
● In vitro use only
● For Veterinary Use Only
SAMPLE COLLECTION AND STORAGE
● Follow proper sample collection procedures.
● Harvest serum and store properly (up to seven days at 2 - 7 °C.
● For prolonged storage, samples should be kept frozen (-20 °C or colder).
● Test only good quality serum or plasma (i.e. avoid bacterial contamination, heavy hemolysis or lipemia). Slightly hemolyzed and lipemic samples may be used; however, they may produce background color. When in doubt, obtain a better quality sample.
TEST PROCEDURE
|
STEP |
NOTES |
|
|
PREPARATION |
||
|
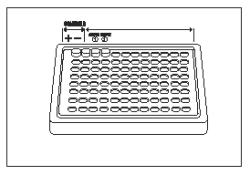
1) |
Remove and place in well holder one well for Positive Control, one well for Negative Control, and one well for each sample. Leave the wells attached to each other. |
 |
|
CONTROLS |
||
|
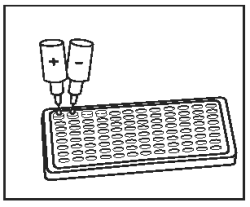
2) |
Add 1 drop of Positive Control (Red Cap) into the first well. Add 1 drop of Negative Control (Gray Cap) into the second well. |
 |
|
SAMPLE(S) |
||
|
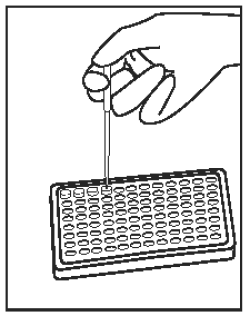
3) |
Using the 0.050 mL precision pipetter dispense 50 µL (0.05 mL) of sample into the next well following the controls. Discard pipette tip. Repeat for each sample into subsequent wells. One well is used for each sample. |
 |
|
NOTE: USE OF THE 0.050 mL PRECISION PIPETTER Place pipette tip firmly onto pipette. Release button slowly to aspirate. Depress button to dispense. |
||
|
CONJUGATE |
||
|
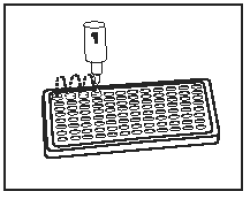
4) |
Add 1 drop of Conjugate (Bottle 1 - Blue Cap) into each well. Tap microplate (without splashing) for 15 seconds to mix. |
 |
|
5) |
Incubate for 10 minutes at 21 - 25 °C. |
|
|
BLOT AND WASH |
||
|
6) |
Discard the fluid from wells into appropriate container. Invert holder and blot firmly onto a paper towel to remove final drops. |
 |
|
7) |
Wash by vigorously filling the wells to overflowing with distilled or deionized water. Direct a forceful stream into each well. (Oversplashing will not contaminate adjacent wells). |
 |
|
8) |
Discard the fluid from the wells into an appropriate container after each wash. |
|
|
9) |
Repeat the washing procedure five (5) times. |
|
|
10) |
Discard excess fluid into appropriate container. Invert holder and blot firmly onto a paper towel to remove final drops. |
|
|
DEVELOP |
||
|
11) |
Add 2 drops Substrate Buffer (Bottle 2 - Purple Cap) into each well. Tap well holder (without splashing) for 15 seconds to mix. |
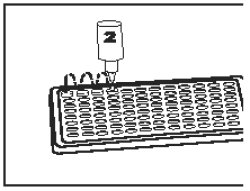 |
|
12) |
Incubate for 5 minutes at 21 - 25 °C. |
|
|
13) |
Read results immediately. |
|
DANGER
Substrate Buffer: DANGER. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. May damage the unborn child. May cause damage to organs (kidney, liver) through prolonged or repeated exposure. Obtain special instructions before use. Do not handle until all safety precautions have been read and understood. Do not breathe mist or vapor. Wear protective gloves/protective clothing/eye protection/face protection. Use personal protective equipment as required. Wash thoroughly after handling. Use only outdoors or in a well-ventilated area. Avoid release to the environment. IF exposed or concerned: Get medical advice/attention. IF ON SKIN: Wash with plenty of soap and water. If skin irritation occurs: Get medical advice/attention. IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for breathing. Call a poison center/doctor if you feel unwell. IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing. If eye irritation persists: Get medical advice/attention. Take off contaminated clothing and wash before reuse. Store in a well-ventilated place. Keep container tightly closed. Store locked up. Dispose of contents/container in accordance with local/regional/national/international regulations.
INTERPRETATION OF RESULTS
Controls:
● POSITIVE control should be distinctly blue.
● NEGATIVE control should be completely clear.
Samples:
● POSITIVE samples will be blue. Color intensity will vary with level of heartworm antigen present. This may reflect worm burden.
● NEGATIVE samples will be clear. Compare directly with the negative control against a white background.
NOTES
● When mixing well contents in Steps 4 and 11, tap microplate rapidly for a full 15 seconds.
● Washing is the most important step. Microwells cannot be overwashed. Underwashing will result in nonspecific color development in the negative control and sample wells.
● Read results at 5 minutes. If no color is seen at 5 minutes, the sample is negative.
SYMBOL DESCRIPTIONS

Zoetis Inc., Kalamazoo, MI 49007, USA
www.zoetis.com
VLN/PCN 190/5018.00
EC REP
Zoetis Belgium SA, Rue Laid Burniat 1, 1348 Louvain-la-Neuve, Belgium
Zoetis Australia Pty Ltd, L6, 5 Rider Bvd, RHODES NSW 2138, AUSTRALIA
1800 814 883
40028768
CPN: 3690613.0
333 PORTAGE STREET, KALAMAZOO, MI, 49007
| Telephone: | 269-359-4414 | |
| Customer Service: | 888-963-8471 | |
| Website: | www.zoetis.com |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". DVMetrics assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the DVMetrics service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-08-27
