Bovikalc
This treatment applies to the following species:Oral Calcium Supplement
For Freshening Cows
FAST & SUSTAINED RELEASE OF CALCIUM
Product Characteristics: BOVIKALC is an oral calcium supplement for use around parturition in fresh dairy cows.
Actions: After administration of BOVIKALC the bolus disintegrates completely within 30 minutes.
The calcium chloride dissolves quickly in the rumen, while the calcium sulfate dissolves only after being degraded by the rumen microflora, providing a sustained release of calcium.
Ingredients: Calcium chloride, calcium sulfate, water, fat product, xanthan gum.
Analytical content:
Calcium, minimum 21.2%
Calcium, maximum 25.4%
How to use BOVIKALC?
Minimum protocol:
● 1st bolus immediately after calving
● 2nd bolus 12 - 24 hours after calving
Extended protocol: For high yielding cows and cows with previous history of hypocalcemia.
● 1st bolus at the first signs of parturition
● 2nd bolus immediately after calving
● 3rd bolus 12 - 24 hours after calving
● 4th bolus 24 - 48 hours after calving
Please discuss protocol with Herd Nutritionist and/or Herd Veterinarian before implementation.
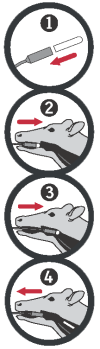
Place the bolus in the bolus applicator with the rounded end pointing outward.
If right- handed, place yourself at the cow’s right side.
Grab the cow’s upper jaw with your left hand. Keep the cow’s head at a natural position and a little towards yourself. Gently guide the loaded BOVIKALC applicator into the cow’s mouth.
When you feel resistance, gently guide the applicator over the tongue. When the applicator is behind the tongue, squeeze the handle to release the bolus.
Wait a moment for the cow to swallow and then remove the applicator.
Each 192 g bolus provides 43 g of calcium, 71% from calcium chloride and 29% from calcium sulfate.
BOVIKALC does not contain any sodium, phosphorus or magnesium.
Precautions: Follow label instructions carefully. Incorrect use or handling may cause serious side effects.
User assumes risk of use or handling contrary to instructions, or under abnormal conditions or conditions not reasonably foreseeable.
Only administer using the BOVIKALC branded applicator.
BOVIKALC must not be given to cows with clinical signs of milk fever as the swallowing reflex may be impaired.
Do not give BOVIKALC to cows lying down.
Do not administer BOVIKALC if the coating on the bolus becomes damaged or the bolus is broken.
Storage
Store at or below 30°C (86°F).The bolus should be stored in the original plastic tube until immediately before use, as it is susceptible to humidity.
Keep out of the reach of children.
BOVIKALC is not for human use.
Do not use broken or damaged boluses.
Please note the caution on the packaging.
BOVIKALC is a registered trademark of Boehringer Ingelheim GmbH, used under license.
© 2019 Boehringer Ingelheim Vetmedica, Inc. All rights reserved.
Manufactured in Denmark by: Boehringer Ingelheim Animal Health Denmark A/S
Marketed by: Boehringer Ingelheim Animal Health USA Inc., Duluth, GA 30096
Toll Free 1-888-637-4251
MADE IN DENMARK
GTIN: (01)05703655003185
|
Net Contents: |
|
|
4 boluses |
113459-005 434502-04 |
|
48 boluses FRAGILE: HANDLE WITH CARE |
119053-004 434505-02 |
CPN: 1028157.4
3239 SATELLITE BLVD., BLDG 500, DULUTH, GA, 30096
| Telephone: | 800-325-9167 | |
| Customer Service: | 888-637-4251 | |
| Technical Service: | 888-637-4251 | |
| Fax: | 816-236-2717 | |
| Website: | www.boehringer-ingelheim.com |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". DVMetrics assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the DVMetrics service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-08-27
