Repaglinide and Metformin Tablets: Package Insert / Prescribing Info
Package insert / product label
Generic name: repaglinide and metformin hydrochloride
Dosage form: tablet
Drug class: Antidiabetic combinations
Medically reviewed by Drugs.com. Last updated on Mar 25, 2025.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Drug Interactions
- Use In Specific Populations
- Overdosage
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
REPAGLINIDE and METFORMIN hydrochloride tablets, for oral use
Initial U.S. Approval: 2008
WARNING: LACTIC ACIDOSIS
See full prescribing information for complete boxed warning
- Post-marketing cases of metformin-associated lactic acidosis have resulted in death, hypothermia, hypotension, and resistant bradyarrhythmias. Symptoms included malaise, myalgias, respiratory distress, somnolence, and abdominal pain. Laboratory abnormalities included elevated blood lactate levels, anion gap acidosis, increased lactate/pyruvate ratio; and metformin plasma levels generally >5 mcg/mL. (5.1)
- Risk factors include renal impairment, concomitant use of certain drugs, age > 65 years old, radiological studies with contrast, surgery and other procedures, hypoxic states, excessive alcohol intake, and hepatic impairment. Steps to reduce the risk of and manage metformin-associated lactic acidosis in these high risk groups are provided in the Full Prescribing Information. (5.1)
- If lactic acidosis is suspected, discontinue repaglinide and metformin hydrochloride tablets and institute general supportive measures in a hospital setting. Prompt hemodialysis is recommended. (5.1)
Recent Major Changes
Indications and Usage for Repaglinide and Metformin Tablets
Repaglinide and metformin hydrochloride tablets are a combination of repaglinide, a glinide, and metformin, a biguanide, indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus who are already treated with a glinide and metformin or who have inadequate glycemic control on a glinide alone or metformin alone. (1)
Limitation of Use:
- Not for treatment of type 1 diabetes mellitus or diabetic ketoacidosis (1)
Repaglinide and Metformin Tablets Dosage and Administration
- Instruct patients to take repaglinide and metformin hydrochloride tablets within 30 minutes before meals. Instruct patients to skip the scheduled dose of repaglinide and metformin hydrochloride tablets if a meal is skipped. (2.1)
- For patients inadequately controlled on metformin, start with 1 mg repaglinide/500 mg metformin twice daily and gradually titrate dose based on glycemic response. (2.1)
- For patients inadequately controlled on repaglinide, start with 1 mg repaglinide/500 mg metformin or 2 mg repaglinide/500 mg metformin twice daily and gradually titrate dose to reduce gastrointestinal side effects (2.1)
- For patients taking repaglinide and metformin, start repaglinide and metformin hydrochloride tablets at the dose of repaglinide and metformin similar to (but not exceeding) the patient's current doses. Titrate as needed to achieve glycemic control (2.1)
- Do not exceed 10 mg repaglinide/2500 mg metformin daily or 4 mg repaglinide/1000 mg metformin per meal. (2.1)
- Assess renal function with estimated glomerular filtration rate (eGFR) before initiation of repaglinide and metformin hydrochloride tablets and periodically thereafter (2.2)
- Contraindicated in patients with eGFR below 30 mL/minute/1.73 m2
- Initiation is not recommended in patients with eGFR between 30 to 45 mL/minute/1.73 m2
- Assess risk/benefit of continuing if eGFR falls below 45 mL/minute/1.73 m2
- Discontinue if eGFR falls below 30 mL/minute/1.73 m2
- Repaglinide and metformin hydrochloride tablets may need to be discontinued at time of, or prior to, iodinated contrast imaging procedures (2.3)
- Dose modifications are required when used concominantly with some medications. (2.4, 7)
Dosage Forms and Strengths
Contraindications
Warnings and Precautions
- Lactic acidosis : See boxed warning. (4, 5.1)
- Hypoglycemia : Repaglinide and metformin hydrochloride tablets may cause hypoglycemia. Skip the scheduled dose if a meal is skipped to reduce the risk of hypoglycemia. Reduce the dose if hypoglycemia occurs. (5.2)
- Vitamin B12deficiency : Metformin can lower vitamin B12levels. Monitor hematological parameters annually. (5.3)
- Serious Cardiovascular Adverse Reactions with Concomitant NPH-insulin : Repaglinide and metformin hydrochloride tablets are not indicated for use with NPH insulin. (5.4)
- Macrovascular outcomes : There have been no clinical studies establishing conclusive evidence of macrovascular risk reduction with repaglinide and metformin hydrochloride tablets. (5.5)
Adverse Reactions/Side Effects
- Hypoglycemia and headache were the most common adverse reactions (≥10%) reported among patients treated with repaglinide in combination with metformin. (6.1)
- Gastrointestinal reactions (e.g., diarrhea, nausea and vomiting) are the most common adverse reactions with metformin (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Lupin Pharmaceuticals, Inc. at 1-800-399-2561 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
- Carbonic anhydrase inhibitors may increase risk of lactic acidosis. Consider more frequent monitoring. (7)
- Drugs that reduce metformin clearance (such as ranolazine, vandetanib, dolutegravir, and cimetidine) may increase the accumulation of metformin. Consider the benefits and risks of concomitant use. (7)
- Alcohol can potentiate the effect of metformin on lactate metabolism. Warn patients against excessive alcohol intake. (7)
- Clopidogrel: Avoid concomitant use; if used concomitantly limit total daily dose of repaglinide to 4 mg (7)
- Cyclosporine : Limit daily dose of repaglinide to 6 mg and increase frequency of glucose monitoring when co-administered (7)
- CYP2C8 and CYP3A4 Inhibitors and Drugs That May Increase the Risk of Hypoglycemia : Co-administration may require dose reductions and increased frequency of glucose monitoring (7)
- CYP2C8 and CYP3A4 Inducers and Drugs That May Decrease the Blood Glucose Lowering Effect of Repaglinide and Metformin Hydrochloride Tablets : Co-administration may require dose increases and increased frequency of glucose monitoring (7)
- Drugs That May Blunt Signs and Symptoms of Hypoglycemia : Increased frequency of glucose monitoring may be required when co-administered (7)
Use In Specific Populations
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 3/2018
Full Prescribing Information
WARNING: LACTIC ACIDOSIS WARNING: LACTIC ACIDOSIS
Post-marketing cases of metformin-associated lactic acidosis have resulted in death, hypothermia, hypotension, and resistant bradyarrhythmias. The onset of metformin-associated lactic acidosis is often subtle, accompanied only by nonspecific symptoms such as malaise, myalgias, respiratory distress, somnolence, and abdominal pain. Metformin-associated lactic acidosis was characterized by elevated blood lactate levels (> 5 mmol/Liter), anion gap acidosis (without evidence of ketonuria or ketonemia), an increased lactate/pyruvate ratio; and metformin plasma levels generally >5 mcg/mL [see WARNINGS AND PRECAUTIONS (5.1)].
Risk factors for metformin-associated lactic acidosis include renal impairment, concomitant use of certain drugs (e.g., carbonic anyhydrase inhibitors such as topiramate), age 65 years old or greater, having a radiological study with contrast, surgery and other procedures, hypoxic states (e.g., acute congestive heart failure), excessive alcohol intake, and hepatic impairment.
Steps to reduce the risk of and manage metformin-associated lactic acidosis in these high risk groups are provided in the full prescribing information [see DOSAGE AND ADMINISTRATION (2.2), CONTRAINDICATIONS (4), WARNINGS AND PRECAUTIONS (5.1), DRUG INTERACTIONS (7), and USE IN SPECIFIC POPULATIONS (8.6, 8.7)].
If metformin-associated lactic acidosis is suspected, immediately discontinue repaglinide and metformin hydrochloride tablets and institute general supportive measures in a hospital setting. Prompt hemodialysis is recommended [see WARNINGS AND PRECAUTIONS (5.1)].
1. Indications and Usage for Repaglinide and Metformin Tablets
Repaglinide and metformin hydrochloride tablets are indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus who are already treated with a glinide and metformin or who have inadequate glycemic control on a glinide alone or metformin alone.
Limitation of Use
Repaglinide and metformin hydrochloride tablets should not be used in patients with type 1 diabetes mellitus or for the treatment of diabetic ketoacidosis.
2. Repaglinide and Metformin Tablets Dosage and Administration
2.1 Recommended Dosage and Administration
General Dosage and Administration information
Administer repaglinide and metformin hydrochloride tablets orally 2 to 3 times a day with meals up to a maximum daily dose of 10 mg repaglinide/2500 mg metformin. No more than 4 mg repaglinide/1000 mg metformin should be taken per meal.
Instruct patients to take repaglinide and metformin hydrochloride tablets within 30 minutes before meals. In patients who skip meals, instruct patients to skip the scheduled dose of repaglinide and metformin hydrochloride tablets to reduce the risk of hypogylcemia. In patients who experience hypoglycemia, the dose of repaglinide should be reduced [see WARNINGS AND PRECAUTIONS (5.2)].
Patients Inadequately Controlled with Metformin Monotherapy
The recommended starting dose of repaglinide and metformin hydrochloride tablets 1 mg repaglinide/500 mg metformin twice daily with meals. Gradually increase dose based on glycemic response to reduce the risk of hypoglycemia with repaglinide.
Patients Inadequately Controlled with Glinide Monotherapy
The recommended starting dose of metformin component of repaglinide and metformin hydrochloride tablets is 500 mg metformin twice daily with meals. Gradually increase dose based on glycemic response to reduce gastrointestinal side effects associated with metformin.
Patients Currently Using Repaglinide and Metformin Concomitantly
Initiate repaglinide and metformin hydrochloride tablets at the dose of repaglinide and metformin similar to (but not exceeding) the patient's current doses. Titrate as needed to achieve glycemic control up to the maximum daily dose.
2.2 Recommended Dosage for Patients with Renal Impairment
Assess renal function with an estimated glomerular filtration rate (eGFR) prior to initiation of repaglinide and metformin hydrochloride tablets and periodically thereafter.
Repaglinide and metformin hydrochloride tablets is contraindicated in patients with an eGFR below 30 mL/min/1.73 m2.
Initiation of repaglinide and metformin hydrochloride tablets in patients with an eGFR between 30 to 45 mL/min/1.73 m2 is not recommended.
In patients taking repaglinide and metformin hydrochloride tablets whose eGFR later falls below 45 mL/min/1.73 m2, assess the benefit-risk of continuing therapy.
Discontinue repaglinide and metformin hydrochloride tablets if the patient's eGFR later falls below 30 mL/min/1.73 m2 [see CONTRAINDICATIONS (4) and WARNINGS AND PRECAUTIONS (5.1)].
2.3 Discontinuation for Iodinated Contrast Imaging Procedures
Discontinue repaglinide and metformin hydrochloride tablets at the time of, or prior to, an iodinated contrast imaging procedure in patients with an eGFR between 30 and 60 mL/min/1.73 m2; in patients with a history of liver disease, alcoholism or heart failure; or in patients who will be administered intra-arterial iodinated contrast. Re-evaluate eGFR 48 hours after the imaging procedure; restart repaglinide and metformin hydrochloride tablets if renal function is stable [See WARNINGS AND PRECAUTIONS (5.1)].
2.4 Dose Modifications for Drug Interactions
Concomitant use with gemfibrozil is contraindicated [see CONTRAINDICATIONS (4)].
Avoid concomitant use of repaglinide and metformin hydrochloride tablets with clopidogrel. If concomitant use can not be avoided, initiate repaglinide at 0.5 mg before each meal. Although repaglinide and metformin hydrochloride tablets are not available in that strength, repaglinide
0.5 mg tablets are available. Do not exceed a total daily dose of 4 mg of repaglinide [see DRUG INTERACTIONS (7), CLINICAL PHARMACOLOGY (12.3)].
Do not exceed a total daily dose of 6 mg of repaglinide in patients receiving cyclosporine [see DRUG INTERACTIONS (7), CLINICAL PHARMACOLOGY (12.3)].
Dosage adjustments are recommended in patients taking concomitant strong CYP3A4 or CYP2C8 inhibitors or strong CYP3A4 or CYP2C8 inducers [see DRUG INTERACTIONS (7), CLINICAL PHARMACOLOGY (12.3)].
3. Dosage Forms and Strengths
- 1 mg repaglinide/500 mg metformin tablets are yellow coloured, biconvex, oval shaped, film coated tablets, debossed with 'V41' on one side and 'LU' on the other side.
- 2 mg repaglinide/500 mg metformin tablets are pink coloured, biconvex, oval shaped, film coated tablets, debossed with 'V42' on one side and 'LU' on the other side.
4. Contraindications
Repaglinide and metformin hydrochloride tablets are contraindicated in patients with:
- Severe renal impairment (eGFR below 30 mL/min/1.73 m2) [see WARNINGS AND PRECAUTIONS (5.1)]
- Acute or chronic metabolic acidosis, including diabetic ketoacidosis. Diabetic ketoacidosis should be treated with insulin [see WARNINGS AND PRECAUTIONS (5.1)] .
- Concomitant use of gemfibrozil [see DRUG INTERACTIONS (7.2)]
- Known hypersensitivity to repaglinide, metformin or any inactive ingredients
5. Warnings and Precautions
5.1 Lactic Acidosis
There have been post-marketing cases of metformin-associated lactic acidosis, including fatal cases. These cases had a subtle onset and were accompanied by nonspecific symptoms such as malaise, myalgias, abdominal pain, respiratory distress, or increased somnolence; however, hypothermia, hypotension and resistant bradyarrhythmias have occurred with severe acidosis.
Metformin-associated lactic acidosis was characterized by elevated blood lactate concentrations (>5 mmol/Liter), anion gap acidosis (without evidence of ketonuria or ketonemia), and an increased lactate:pyruvate ratio; metformin plasma levels generally >5 mcg/mL. Metformin decreases liver uptake of lactate increasing lactate blood levels which may increase the risk of lactic acidosis, especially in patients at risk.
If metformin-associated lactic acidosis is suspected, general supportive measures should be instituted promptly in a hospital setting, along with immediate discontinuation of repaglinide and metformin hydrochloride tablets.
In repaglinide and metformin hydrochloride tablets treated patients with a diagnosis or strong suspicion of lactic acidosis, prompt hemodialysis is recommended to correct the acidosis and remove accumulated metformin (metformin is dialyzable, with a clearance of up to 170 mL/minute under good hemodynamic conditions). Hemodialysis has often resulted in reversal of symptoms and recovery.
Educate patients and their families about the symptoms of lactic acidosis and if these symptoms occur instruct them to discontinue repaglinide and metformin hydrochloride tablets and report these symptoms to their healthcare provider.
For each of the known and possible risk factors for metformin-associated lactic acidosis, recommendations to reduce the risk of and manage metformin-associated lactic acidosis are provided below:
Renal Impairment
The post-marketing metformin-associated lactic acidosis cases primarily occurred in patients with significant renal impairment. The risk of metformin accumulation and metformin-associated lactic acidosis increases with the severity of renal impairment because metformin is substantially excreted by the kidney. Clinical recommendations based upon the patient's renal function include [see CLINICAL PHARMACOLOGY (12.3)]:
- Before initiating repaglinide and metformin hydrochloride tablets, obtain an estimated glomerular filtration rate (eGFR).
- Repaglinide and metformin hydrochloride tablets is contraindicated in patients with an eGFR less than 30 mL/minute/1.73 m2[see CONTRAINDICATIONS (4)].
- Initiation of repaglinide and metformin hydrochloride tablets is not recommended in patients with eGFR between 30 to 45 mL/minute/1.73 m2.
- Obtain an eGFR at least annually in all patients taking repaglinide and metformin hydrochloride tablets. In patients at increased risk for the development of renal impairment (e.g., the elderly), renal function should be assessed more frequently.
- In patients taking repaglinide and metformin hydrochloride tablets whose eGFR later falls below 45 mL/min/1.73 m2, assess the benefit and risk of continuing therapy.
The concomitant use of repaglinide and metformin hydrochloride tablets with specific drugs may increase the risk of metformin-associated lactic acidosis: those that impair renal function, result in significant hemodynamic change, interfere with acid-base balance or increase metformin accumulation [see DRUG INTERACTIONS (7)]. Therefore, consider more frequent monitoring of patients.
Age 65 or Greater
The risk of metformin-associated lactic acidosis increases with the patient's age because elderly patients have a greater likelihood of having hepatic, renal, or cardiac impairment than younger patients. Assess renal function more frequently in elderly patients [see USE IN SPECIFIC POPULATIONS (8.5)].
Radiological Studies with Contrast
Administration of intravascular iodinated contrast agents in metformin-treated patients has led to an acute decrease in renal function and the occurrence of lactic acidosis. Stop repaglinide and metformin hydrochloride tablets at the time of, or prior to, an iodinated contrast imaging procedure in patients with an eGFR between 30 and 60 mL/min/1.73 m2; in patients with a history of hepatic impairment, alcoholism, or heart failure; or in patients who will be administered intra-arterial iodinated contrast. Re-evaluate eGFR 48 hours after the imaging procedure, and restart repaglinide and metformin hydrochloride tablets if renal function is stable.
Surgery and Other Procedures
Withholding of food and fluids during surgical or other procedures may increase the risk for volume depletion, hypotension and renal impairment. Repaglinide and metformin hydrochloride tablets should be temporarily discontinued while patients have restricted food and fluid intake.
Hypoxic States
Several of the postmarketing cases of metformin-associated lactic acidosis occurred in the setting of acute congestive heart failure (particularly when accompanied by hypoperfusion and hypoxemia). Cardiovascular collapse (shock), acute myocardial infarction, sepsis, and other conditions associated with hypoxemia have been associated with lactic acidosis and may also cause prerenal azotemia. When such events occur, discontinue repaglinide and metformin hydrochloride tablets.
Excessive Alcohol Intake
Alcohol potentiates the effect of metformin on lactate metabolism and this may increase the risk of metformin-associated lactic acidosis. Warn patients against excessive alcohol intake while receiving repaglinide and metformin hydrochloride tablets.
Hepatic Impairment
Patients with hepatic impairment have developed with cases of metformin-associated lactic acidosis. This may be due to impaired lactate clearance resulting in higher lactate blood levels. Therefore, avoid use of repaglinide and metformin hydrochloride tablets in patients with clinical or laboratory evidence of hepatic disease.
5.2 Hypoglycemia
All glinides, including repaglinide and metformin hydrochloride tablets, can cause hypoglycemia [see ADVERSE REACTIONS (6.1)]. Severe hypoglycemia can cause seizures, may be life-threatening, or cause death. Hypoglycemia can impair concentration ability and reaction time; this may place an individual and others at risk in situations where these abilities are important (e.g., driving or operating other machinery).
Hypoglycemia can happen suddenly and symptoms may differ in each individual and change over time in the same individual. Symptomatic awareness of hypoglycemia may be less pronounced in patients with longstanding diabetes, in patients with diabetic nerve disease, in patients using medications that block the sympathetic nervous system (e.g., beta-blockers) [see DRUG INTERACTIONS (7)], or in patients who experience recurrent hypoglycemia.
Factors which may increase the risk of hypoglycemia include changes in meal pattern (e.g., macronutrient content), changes in level of physical activity, changes to co-administered medication [see DRUG INTERACTIONS (7)], and concomitant use with other antidiabetic agents. Patients with renal or hepatic impairment may be at higher risk of hypoglycemia [see USE IN SPECIFIC POPULATIONS (8.6, 8.7)].
Patients should administer repaglinide and metformin hydrochloride tablets before meals and be instructed to skip the dose of repaglinide and metformin hydrochloride tablets if a meal is skipped. In patients who experience hypoglycemia, the dose of repaglinide and metformin hydrochloride tablets should be reduced [see DOSAGE AND ADMINISTRATION (2.1)]. Patients and caregivers must be educated to recognize and manage hypoglycemia. Self-monitoring of blood glucose plays an essential role in the prevention and management of hypoglycemia. In patients at higher risk for hypoglycemia and patients who have reduced symptomatic awareness of hypoglycemia, increased frequency of blood glucose monitoring is recommended.
5.3 Vitamin B12 Levels
In controlled clinical trials of metformin of 29 weeks' duration, a decrease to subnormal levels of previously normal serum vitamin B12 levels, without clinical manifestations, was observed in approximately 7% of patients. This finding, possibly due to interference with B12 absorption from the B12-intrinsic factor complex, is rarely associated with anemia and appears to be rapidly reversible with discontinuation of metformin or vitamin B12 supplementation. Measurement of hematologic parameters on an annual basis is advised in patients on repaglinide and metformin hydrochloride tablets and any apparent abnormalities should be appropriately investigated and managed.
Certain individuals (those with inadequate vitamin B12 or calcium intake or absorption) appear to be predisposed to developing subnormal vitamin B12 levels. In these patients, routine serum vitamin B12 measurements at 2- to 3- year intervals may be useful.
5.4 Serious Cardiovascular Adverse Reactions with Concomitant Use with NPH-insulin
Across seven controlled trials, there were six serious adverse events of myocardial ischemia in patients treated with repaglanide plus NPH-insulin from two studies, and one event in patients using insulin formulations alone from another study [see ADVERSE REACTIONS (6.1)]. repaglinide and metformin hydrochloride tablets is not indicated for use in combination with NPH-insulin.
6. Adverse Reactions/Side Effects
The following adverse reactions are discussed in more detail in other sections of the labeling.
- Lactic acidosis [see WARNINGS AND PRECAUTIONS (5.1)]
- Hypoglycemia [see WARNINGS AND PRECAUTIONS (5.2)]
- Vitamin B12levels [see WARNINGS AND PRECAUTIONS (5.3)]
6.1 Clinical Trial Experience
Because clinical trials are conducted under varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Repaglinide and metformin hydrochloride tablets was administered to 374 patients with type 2 diabetes during clinical trials. Table 1 summarizes the most common adverse reactions occurring in a 6-month randomized trial of repaglinide as add-on therapy to metformin in patients inadequately controlled on metformin alone.
| Table 1: Adverse Reactions (%) occurring > 10% in Patients Treated with Repaglinide and Metformin Together or Repaglinide and Metformin Monotherapy during a 6 Month Trial
|
|||
| Repaglinide and Metformin | Metformin Monotherapy | Repaglinide Monotherapy |
|
| N = 27 | N = 27 | N = 28 |
|
| Diarrhea Nausea | 19 15 | 30 7 | 7 4 |
| Symptomatic Hypoglycemia* | 33 | 0 | 11 |
| Headache | 22 | 15 | 11 |
| Upper Respiratory Tract Infection | 11 | 11 | 11 |
| * Hypoglycemia with symptoms which included, but were not limited to, anxious feeling, dizziness, sweating, tremor, hunger and difficulty in concentration. None of the symptomatic hypoglycemia events listed in the table required the assistance of another person. |
|||
In clinical trials with repaglinide, hypoglycemia is the most commonly observed adverse reaction. Mild or moderate hypoglycemia occurred in 31% of repaglinide treated patients and 7% of placebo treated patients.
Gastrointestinal Adverse Reactions
Gastrointestinal reactions (e.g., diarrhea, nausea, vomiting) are the most common adverse reactions (> 5%) with metformin treatment and are more frequent at higher metformin doses.
Weight Gain
In a clinical trial, 83 patients were randomized to add-on repaglinide, repaglinide monotherapy, or continued treatment with metformin monotherapy. A statistically significant weight gain was observed for the combination of repaglinide and metformin in a pairwise comparison with metformin monotherapy (see Table 2).
| Table 2: Repaglinide as Add-on to Metformin: Mean Changes from Baseline in Body Weight After 4 to 5 Months of Treatment1
|
|||
| Repaglinide add-on to Metformin | Repaglinide monotherapy | Metformin HCl monotherapy |
|
| N | 27 | 28 | 27 |
| Weight (kg) | |||
| Baseline | 93 | 87 | 91 |
| Change from Baseline | 2.4# | 3.0 | -0.9 |
| 1: based on intent-to-treat analysis #: p<0.05, for pairwise comparison with metformin monotherapy. |
|||
Cardiovascular Events in repaglinide monotherapy trials
The incidence of total serious cardiovascular adverse events, including ischemia, was higher for repaglinide (51/1228 or 4%) than for sulfonylurea drugs (13/498 or 3%) in controlled clinical trials.
Seven controlled clinical trials included repaglinide combination therapy with NPH-insulin (n=431), insulin formulations alone (n=388) or other combinations (sulfonylurea plus NPH-insulin or repaglinide plus metformin ) (n=120). There were six serious adverse events of myocardial ischemia in patients treated with repaglinide plus NPH-insulin (1.4%) from two studies, and one event in patients using insulin formulations alone from another study (0.3%) [see WARNINGS AND PRECAUTIONS (5.4)].
6.2 Postmarketing Experience
The following additional adverse reactions have been identified during postapproval use. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or a causal relationship to drug exposure.
Repaglinide
Alopecia, hemolytic anemia, pancreatitis, Stevens-Johnson Syndrome, and severe hepatic dysfunction including jaundice and hepatitis.
Metformin
Cholestatic, hepatocellular, and mixed hepatocellular liver injury
Related/similar drugs
7. Drug Interactions
Table 3 includes a list of drugs with clinically important drug interactions when administered concomitantly with repaglinide and metformin hydrochloride tablets and instructions for preventing or managing them.
| Carbonic Anhydrase Inhibitors
|
|
| Clinical Impact:
| Carbonic anhydrase inhibitors frequently causes a decrease in serum bicarbonate and induce non-anion gap, hyperchloremic metabolic acidosis. Concomitant use of these drugs with repaglinide and metformin hydrochloride tablets may increase the risk for lactic acidosis. |
| Intervention:
| Consider more frequent monitoring of these patients. |
| Examples:
| Topiramate, zonisamide, acetazolamide or dichlorphenamide. |
| Drugs that reduce metformin clearance
|
|
| Clinical Impact:
| Concomitant use of drugs that interfere with common renal tubular transport systems involved in the renal elimination of metformin could increase systemic exposure to metformin and may increase the risk for lactic acidosis [see CLINICAL PHARMACOLOGY (12.3)].
|
| Intervention:
| Consider the benefits and risks of concomitant use. |
| Examples:
| E.g., organic cationic transporter-2 [OCT2] / multidrug and toxin extrusion [MATE] inhibitors such as ranolazine, vandetanib, dolutegravir, and cimetidine. |
| Alcohol
|
|
| Clinical Impact:
| Alcohol is known to potentiate the effect of metformin on lactate metabolism. |
| Intervention:
| Warn patients against excessive alcohol intake while receiving repaglinide and metformin hydrochloride tablets. |
| Gemfibrozil
|
|
| Clinical Impact:
| Gemfibrozil significantly increased repaglinide exposures by 8.1 fold [see CLINICAL PHARMACOLOGY (12.3)]
|
| Intervention:
| Do not administer repaglinide and metformin hydrochloride tablets to patients receiving gemfibrozil [see CONTRAINDICATIONS (4)] . |
| Clopidogrel
|
|
| Clinical Impact:
| Clopidogrel increased repaglinide exposures by 3.9 to 5.1 fold [see CLINICAL PHARMACOLOGY (12.3)]
|
| Intervention:
| Avoid concomitant use of repaglinide and metformin hydrochloride tablets with clopidogrel. If concomitant use can not be avoided, initiate repaglinide at 0.5 mg before each meal. Although repaglinide and metformin hydrochloride tablets is not available in that strength, repaglinide 0.5 mg tablets are available. Do not exceed a total daily repaglinide dose of 4 mg. Increased frequency of glucose monitoring may be required during concomitant use [see DOSAGE AND ADMINISTRATION (2.4)] . |
| Cyclosporine
|
|
| Clinical Impact:
| Cyclosporine increased low dose repaglinide exposures by 2.5 fold [see CLINICAL PHARMACOLOGY (12.3)]
|
| Intervention:
| Daily maximum repaglinide dose should be limited to 6 mg, and increased frequency of glucose monitoring may be required when repaglinide and metformin hydrochloride tablets is co-administered with cyclosporine. |
| CYP2C8 and CYP3A4 Inhibitors
|
|
| Intervention:
| Repaglinide and metformin hydrochloride tablets dose reductions and increased frequency of glucose monitoring may be required when co-administered. |
| Examples:
| Drugs that are known to inhibit CYP3A4 include antifungal agents (ketoconazole, itraconazole) and antibacterial agents (clarithromycin, erythromycin). Drugs that are known to inhibit CYP2C8 include trimethoprim, gemfibrozil, montelukast, deferasirox, and clopidiogrel. |
| CYP2C8 and CYP3A4 Inducers
|
|
| Intervention:
| Repaglinide and metformin hydrochloride tablets dose increases and increased frequency of glucose monitoring may be required when co-administered. |
| Examples:
| Drugs that induce the CYP3A4 and/or 2C8 enzyme systems include rifampin, barbiturates, and carbamezapine |
| Drugs That May Increase the Risk of Hypoglycemia
|
|
| Intervention:
| Repaglinide and metformin hydrochloride tablets dose reductions and increased frequency of glucose monitoring may be required when co-administered. |
| Examples:
| Antidiabetic agents, ACE inhibitors, angiotensin II receptor blocking agents, disopyramide, fibrates, fluoxetine, monoamine oxidase inhibitors, nonsteroidal anti-inflammatory agents (NSAIDs), pentoxifylline, pramlintide, propoxyphene, salicylates, somatostatin analogs (e.g., octreotide), and sulfonamide antibiotics |
| Drugs That May Decrease the Blood Glucose Lowering Effect of Repaglinide and Metformin Hydrochloride Tablets
|
|
| Intervention:
| Repaglinide and metformin hydrochloride tablets dose increases and increased frequency of glucose monitoring may be required when co-administered. |
| Examples:
| Atypical antipsychotics (e.g., olanzapine and clozapine), calcium channel antagonists, corticosteroids, danazol, diuretics, estrogens, glucagon, isoniazid, niacin, oral contraceptives, phenothiazines, progestogens (e.g., in oral contraceptives), protease inhibitors, somatropin, sympathomimetic agents (e.g., albuterol, epinephrine, terbutaline), and thyroid hormones. |
| Drugs That May Blunt Signs and Symptoms of Hypoglycemia
|
|
| Intervention:
| Increased frequency of glucose monitoring may be required when repaglinide and metformin hydrochloride tablets is co-administered with these drugs. |
| Examples:
| beta-blockers, clonidine, guanethidine, and reserpine |
8. Use In Specific Populations
8.1 Pregnancy
There are no adequate and well-controlled studies in pregnant women with repaglinide and metformin hydrochloride tablets or its individual components. Because animal reproduction studies are not always predictive of human response, repaglinide and metformin hydrochloride tablets like other antidiabetic medications, should be used during pregnancy only if clearly needed.
No animal studies have been conducted with the combined products in repaglinide and metformin hydrochloride tablets. The following data are based on findings in studies performed with repaglinide or metformin individually.
Repaglinide
Repaglinide was not teratogenic in rats at doses 40 times, and rabbits approximately 0.8 times the clinical exposure (on a mg/m2 basis) throughout pregnancy. Offspring of rat dams exposed to repaglinide at 15 times clinical exposure on a mg/m2 basis during days 17 to 22 of gestation and during lactation developed nonteratogenic skeletal deformities consisting of shortening, thickening, and bending of the humerus during the postnatal period. This effect was not seen at doses up to 2.5 times clinical exposure (on a mg/m2 basis) on days 1 to 22 of pregnancy or at higher doses given during days 1 to 16 of pregnancy. Relevant human exposure has not occurred to date and therefore the safety of repaglinide administration throughout pregnancy or lactation cannot be established.
Metformin
Metformin alone was not teratogenic in rats or rabbits at doses up to 600 mg/kg/day. This represents an exposure of approximately two and six times the near-maximal efficacious human daily dose of 2000 mg of the metformin component of repaglinide and metformin hydrochloride tablets based on body surface area comparisons for rats and rabbits, respectively. Determination of fetal concentrations demonstrated a partial placental barrier to metformin.
8.3 Nursing Mothers
No studies in lactating animals have been conducted with the repaglinide and metformin hydrochloride tablets fixed dose combination. In studies performed with individual components, both repaglinide and metformin are excreted into milk of lactating rats. It is not known whether repaglinide or metformin are excreted in human milk. Repaglinide and metformin hydrochloride tablets are not recommended in nursing mothers because it may potentially cause hypoglycemia in nursing infants.
Repaglinide
In rat reproduction studies, measurable levels of repaglinide were detected in the breast milk of the dams and lowered blood glucose levels were observed in the pups. Cross fostering studies indicated that skeletal changes could be induced in control pups nursed by treated dams, although this occurred to a lesser degree than those pups treated in utero.
Metformin
Studies in lactating rats with metformin show that it is excreted into milk and reaches levels comparable to those in plasma.
8.5 Geriatric Use
In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy and the higher risk of lactic acidosis. Assess renal function more frequently in elderly patients. [see WARNINGS AND PRECAUTIONS (5.1), CONTRAINDICATIONS (4), CLINICAL PHARMACOLOGY (12.3)].
8.6 Renal Impairment
Metformin is substantially excreted by the kidney, and the risk of metformin accumulation and lactic acidosis increases with the degree of renal impairment. Repaglinide and metformin hydrochloride tablets are contraindicated in severe renal impairment, patients with an estimated glomerular filtration rate (eGFR) below 30 mL/min/1.73 m2. [see DOSAGE AND ADMINISTRATION (2.2), CONTRAINDICATIONS (4), WARNINGS AND PRECAUTIONS (5.1) and CLINICAL PHARMACOLOGY (12.3)]
8.7 Hepatic Impairment
Use of metformin in patients with hepatic impairment has been associated with some cases of lactic acidosis. Repaglinide and metformin hydrochloride tablets is not recommended in patients with hepatic impairment. [see WARNINGS AND PRECAUTIONS (5.1)]
10. Overdosage
Severe hypoglycemic reactions with coma, seizure, or other neurological impairment may occur and constitute medical emergencies requiring immediate hospitalization. Hypoglycemic symptoms without loss of consciousness or neurologic findings should be treated aggressively with oral glucose and adjustments in drug dosage and/or meal patterns. Close monitoring may continue until the physician is assured that the patient is out of danger. Patients should be closely monitored for a minimum of 24 to 48 hours, since hypoglycemia may recur after apparent clinical recovery. There is no evidence that repaglinide is dialyzable using hemodialysis.
Metformin
Overdose of metformin has occurred, including ingestion of amounts greater than 50 grams. Hypoglycemia was reported in approximately 10% of cases, but no causal association with metformin has been established. Lactic acidosis has been reported in approximately 32% of metformin overdose cases [see WARNINGS AND PRECAUTIONS (5.1)]. Metformin is dialyzable with a clearance of up to 170 mL/min under good hemodynamic conditions. Therefore, hemodialysis may be useful for removal of accumulated drug from patients in whom metformin overdosage is suspected.
11. Repaglinide and Metformin Tablets Description
Repaglinide and metformin hydrochloride tablets for oral use contain repaglinide, a glinide, and metformin, a biguanide.
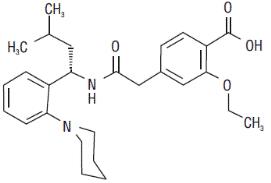
Repaglinide, S(+)2-ethoxy-4(2((3-methyl-1-(2-(1-piperidinyl) phenyl)-butyl) amino)-2-oxoethyl) benzoic acid, is chemically unrelated to the oral sulfonylurea insulin secretagogues. Repaglinide is a white to off-white powder with molecular formula C27H36N2O4 and a molecular weight of 452.6 with the structural formula as shown below. Repaglinide is freely soluble in methanol and ethanol. The pKa of repaglinide in acid is 3.9, and the pKa in amine is 6.0.
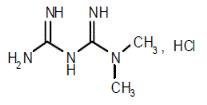
Metformin hydrochloride (N,N-dimethylimidodicarbonimidic diamide hydrochloride) is a white to off-white crystalline compound with a molecular formula of C4H11N5•HCl and a molecular weight of 165.63. Metformin HCl is freely soluble in water and is practically insoluble in acetone, ether, and chloroform. The pKa of metformin hydrochloride is 12.4. The pH of a 1% aqueous solution of metformin hydrochloride is 6.68. The structural formula of metformin hydrochloride is:
Repaglinide and metformin hydrochloride tablets contain 1 mg repaglinide with 500 mg metformin HCl (1 mg/500 mg) or 2 mg repaglinide with 500 mg metformin HCl (2 mg/500 mg) formulated with the following inactive ingredients: hypromellose 6cp, magnesium stearate, meglumine, microcrystalline cellulose, polacrillin potassium, poloxamer 188, polyethylene glycol, povidone, sorbitol, talc and titanium dioxide. Iron oxide yellow is present in the 1 mg/500 mg repaglinide and metformin hydrochloride tablets. Iron oxide red and propylene glycol are present in the 2 mg/500 mg repaglinide and metformin hydrochloride tablets.
12. Repaglinide and Metformin Tablets - Clinical Pharmacology
12.1 Mechanism of Action
Repaglinide and Metformin Hydrochloride Tablets
Repaglinide lowers blood glucose levels by stimulating the release of insulin from the pancreas. This action is dependent upon functioning beta (β) cells in the pancreatic islets.
Repaglinide closes ATP-dependent potassium channels in the β-cell membrane by binding at characterizable sites. This potassium channel blockade depolarizes the β -cell, which leads to an opening of calcium channels. The resulting increased calcium influx induces insulin secretion. The ion channel mechanism is highly tissue selective with low affinity for heart and skeletal muscle.
Metformin improves glucose tolerance in patients with type 2 diabetes by lowering both the basal and postprandial plasma glucose. Metformin decreases hepatic glucose production, decreases intestinal absorption of glucose, and improves insulin sensitivity by increasing peripheral glucose uptake and utilization. With metformin therapy, insulin secretion remains unchanged while fasting insulin levels and day-long plasma insulin response may actually decrease.
12.3 Pharmacokinetics
Repaglinide and Metformin Hydrochloride Tablets
The pharmacokinetic profiles of repaglinide and metformin from repaglinide and metformin hydrochloride tablets doses are listed in Table 4 below. The results of a pharmacokinetic
single-dose crossover study in healthy subjects demonstrated that repaglinide has dose proportional pharmacokinetics (AUC and Cmax) for the combination of repaglinide/metformin in repaglinide and metformin hydrochloride tablets (2 mg/500 mg and 1 mg/500 mg).
| Table 4: Pharmacokinetic Parameters for Repaglinide and Metformin
|
|||||
| Repaglinide | Metformin |
||||
| Repaglinide and Metformin Hydrochloride Tablets | N | AUC (ng⸱h/mL) | Cmax (ng/mL) | AUC (ng⸱h/mL) | Cmax (ng/mL) |
| 2 mg/500 mg tablet | 55 | 34.5 (13.3) | 26.0 (13.7) | 6041.9 (1494.6) | 838.8 (210.2) |
| 1 mg/500 mg tablet | 55 | 17.6 (6.6) | 12.9 (6.9) | 5948.9 (1442.0) | 799.4 (174.6) |
Absorption and Bioavailability:
Repaglinide
After single and multiple oral doses in healthy subjects or in patients with type 2 diabetes, peak plasma drug levels (Cmax) occur within 1 hour (Tmax). Repaglinide is eliminated from the blood stream with a half-life of approximately 1 hour. The mean absolute bioavailability is 56%. When repaglinide was given with food, the mean Tmax was not changed, but the mean Cmax and AUC (area under the time/plasma concentration curve) were decreased 20% and 12.4%, respectively.
Metformin
The absolute bioavailability of a 500 mg metformin tablet given under fasting conditions is approximately 50% to 60%. Studies using single oral doses of metformin tablets of 500 mg to 1,500 mg, and 850 mg to 2,550 mg, indicate that there is a lack of dose proportionality with increasing doses, which is due to decreased absorption rather than an alteration in elimination. Food decreases the extent of and slightly delays the absorption of metformin, as shown by approximately a 40% lower peak concentration (Cmax), a 25% lower area under plasma concentration (AUC) and a 35-minute prolongation of time to peak plasma concentration (Tmax) following administration of a single 850 mg tablet of metformin with food, compared to the same tablet strength administered fasting. The clinical relevance of these decreases is unknown.
Distribution:
Repaglinide
After intravenous (IV) dosing in healthy subjects, the volume of distribution at steady state (Vss) was 31 L, and the total body clearance (CL) was 38 L/h. Protein binding and binding to human serum albumin was greater than 98%.
Metformin
The apparent volume of distribution (V/F) of metformin following single oral dose of 850 mg averaged 654 ± 358 L. Metformin is negligibly bound to plasma proteins. Metformin partitions into erythrocytes, most likely as a function of time. At usual clinical doses and dosing schedules of metformin, steady state plasma concentrations of metformin are reached within 24 to 48 hours and are generally < 1 mcg/mL. During controlled clinical trials, maximum metformin plasma levels did not exceed 5 mcg/mL, even at maximum doses.
Metabolism and Elimination:
Repaglinide
Repaglinide is completely metabolized by oxidative biotransformation and direct conjugation with glucuronic acid after either an intravenous or oral dose. The major metabolites are an oxidized dicarboxylic acid (M2), the aromatic amine (M1), and the acyl glucuronide (M7). The cytochrome P-450 enzyme system, specifically 2C8 and 3A4, have been shown to be involved in the N-dealkylation of repaglinide to M2 and the further oxidation to M1. Metabolites do not contribute to the glucose-lowering effect of repaglinide. Within 96 hours after dosing with 14C-repaglinide as a single, oral dose, approximately 90% of the radiolabel was recovered in the feces and approximately 8% in the urine. Only 0.1% of the dose is cleared in the urine as parent compound. The major metabolite (M2) accounted for 60% of the administered dose. Less than 2% of parent drug was recovered in feces. Repaglinide appears to be a substrate for active hepatic uptake transporter (organic anion transporting protein OATP1B1).
Metformin
Intravenous single-dose studies in normal subjects demonstrate that metformin is excreted unchanged in the urine and does not undergo hepatic metabolism (no metabolites have been identified in humans) or biliary excretion. Renal clearance is approximately 3.5 times greater than creatinine clearance, which indicates that tubular secretion is the major route of metformin elimination. Following oral administration, approximately 90% of the absorbed drug is eliminated via the renal route within the first 24 hours, with a plasma elimination half-life of approximately 6.2 hours. In blood, the elimination half-life is approximately 17.6 hours, suggesting that the erythrocyte mass may be a compartment of distribution.
Specific Populations:
Renal Impairment
Repaglinide:
Single-dose and steady-state pharmacokinetics of repaglinide were compared between patients with type 2 diabetes and normal renal function (CrCl > 80 mL/min), mild to moderate renal function impairment (CrCl = 40 to 80 mL/min), and severe renal function impairment (CrCl = 20 to 40 mL/min). Both AUC and Cmax of repaglinide were similar in patients with normal and mild to moderately impaired renal function (mean values 56.7 ng/mL*hr vs 57.2 ng/mL*hr and 37.5 ng/mL vs 37.7 ng/mL, respectively). Patients with severely reduced renal function had elevated mean AUC and Cmax values (98.0 ng/mL*hr and 50.7 ng/mL, respectively), but this study showed only a weak correlation between repaglinide levels and creatinine clearance.
Metformin:
In patients with decreased renal function, the plasma and blood half-life of metformin is prolonged and the renal clearance is decreased [see CONTRAINDICATIONS (4) and WARNINGS AND PRECAUTIONS (5.1)].
Hepatic Impairment
Repaglinide:
A single-dose, open-label study was conducted in 12 healthy subjects and 12 patients with chronic liver disease (CLD) classified by Child-Pugh scale and caffeine clearance. Patients with moderate to severe impairment of liver function had higher and more prolonged serum concentrations of both total and unbound repaglinide than healthy subjects (AUChealthy: 91.6 ng/mL*hr; AUCcld patients:368.9 ng/mL*hr; Cmax, healthy: 46.7 ng/mL; Cmax, CLD patients: 105.4 ng/mL). AUC was statistically correlated with caffeine clearance. No difference in glucose profiles was observed across patient groups.
Metformin:
No pharmacokinetics studies with metformin have been conducted in patients with hepatic impairment.
Geriatric Patients
Healthy volunteers treated with repaglinide 2 mg before each of 3 meals, showed no significant differences in repaglinide pharmacokinetics between the group of patients <65 years of age and those ≥65 years of age.
Limited data from controlled pharmacokinetic studies of metformin in healthy elderly subjects suggest that total plasma clearance is decreased, the half-life is prolonged, and Cmax is increased, compared to healthy young subjects. From these data, it appears that the change in metformin pharmacokinetics with aging is primarily accounted for by a change in renal function [see WARNINGS AND PRECAUTIONS (5.1)].
Drug Interactions:
| Study Drug*
| Metformin AUC
| Metformin Cmax
|
| Cimetidine
| 40% ↑ | 60% ↑ |
| Furosemide
| 15% ↑ | 22% ↑ |
| Nifedipine
| 9% ↑ | 20% ↑ |
| Propranolol-metformin
| 10% ↓ | 6% ↓ |
| Ibuprofen-metformin
| 5% ↑ | 7% ↑ |
| Study Drug
| Dosing
| Repaglinide Dosing*
| Repaglinide
|
|
| AUC
| Cmax
|
|||
| Clarithromycin†
| 250 mg BID for 4 days | 40% ↑ | 67% ↑ |
|
| Clopidogrel†
| 300 mg (Day 1) 75 mg QD (Day 2 to 3) | 0.25 mg (Day 1 and 3) | (day1) 5.1 fold ↑ (3.9 to 6.6) (day 3) 3.9 fold ↑ (2.9 to 5.3) | 2.5 fold ↑ (1.8 to 3.5) 2.0 fold ↑ (1.3 to 3.1) |
| Cyclosporine
| 100 mg (2 doses 12 hours apart) | 2.5 fold ↑ | 1.8 fold ↑ |
|
| Deferasirox†
| 30 mg/kg QD for 4 days | 0.5 mg | 2.3 fold ↑ | 62% ↑ |
| Fenofibrate
| 200 mg QD for 5 days | 0% | 0% |
|
| Gemfibrozil†
| 600 mg BID for 3 days | 8.1 fold ↑ | 2.4 fold ↑ |
|
| Itraconazole†
| 100 mg BID for 3 days | 1.4 fold↑ | 1.5 fold ↑ |
|
| Gemfibrozil+Itraconazole† Co-administration
| Gem: 600 mg BID for 3 days Itra: 100 mg BID for 3 days | 19 fold ↑ | 2.8 fold ↑ |
|
| Ketoconazole
| 200 mg QD for 4 days | 2 mg | 15% ↑ | 16% ↑ |
| Levonorgestrel/ethinyl Estradiol
| (0.15 mg/0.03mg) Combination tablet QD for 21 days | 2 mg | 0% | 20% ↑ |
| Nifedipine†
| 10 mg TID for 4 days | 2 mg | 0% | 0% |
| Rifampin†
| 600 mg QD for 6 to 7 days | 4 mg | 32 to 80% ↓ | 17 to 79% ↓ |
| Simvastatin
| 20 mg QD for 4 days | 2 mg | 0% | 26% ↑ |
| Trimethoprim†
| 160 mg BID for 2 days 160 mg QD for 1 day | 61% ↑ | 41% ↑ |
|
| Other Drugs
| AUC
| Cmax
|
| Furosemide1
| 12% ↓ | 31%↓ |
| Ethinyl Estradiol2
| 20% ↑ | 20% ↑ |
| Fenofibrate
| 0% | 18% ↑ |
| 1 When administered with metformin 2 Co-administration of a combination tablet (0.15 mg levonorgestrel/0.03 mg ethinyl estradiol) once daily for 21 days with 2 mg repaglinide administered TID (days 1 to 4) and a single dose on day 5. ↓ indicates decrease ↑ indicates increase |
||
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Repaglinide and Metformin Hydrochloride Tablets
No animal studies have been conducted with the combined products in repaglinide and metformin hydrochloride tablets to evaluate carcinogenesis, mutagenesis and impairment of fertility. The following data are based on findings in studies performed with the individual components.
Repaglinide
In a 104-week carcinogenicity study in rats at doses up to 120 mg/kg/day, the incidences of benign adenomas of the thyroid and liver were increased in male rats. The higher incidences of thyroid and liver tumors in male rats were not seen at lower dose of 30 mg/kg/day and 60 mg/kg/day respectively (which are over 15 and 30 times, respectively, clinical exposures on a mg/m2 basis).
In a 104-week carcinogenicity study in mice at doses up to 500 mg/kg/day, no evidence of carcinogenicity was found in mice (which is approximately 125 times clinical exposure on a mg/m2 basis).
Repaglinide was non-genotoxic in a battery of in vivo and in vitro studies: Bacterial mutagenesis (Ames test), in vitro forward cell mutation assay in V79 cells (HGPRT), in vitro chromosomal aberration assay in human lymphocytes, unscheduled and replicating DNA synthesis in rat liver, and in vivo mouse and rat micronucleus tests.
In a rat fertility study, repaglinide was administered to male and female rats at doses up to 300 and 80 mg/kg/day, respectively. No adverse effects on fertility were observed (which are over 40 times clinical exposure on a mg/m2 basis).
Metformin
In a 104-week carcinogenicity study in rats at doses up to 900 mg/kg/day, the incidences of benign stromal uterine polyps were increased in female rats at 900 mg/kg/day (which is approximately four times the maximal recommended human daily dose of 2000 mg of metformin component of repaglinide and metformin hydrochloride tablets on a mg/m2 basis).
In a 91-week carcinogenicity study in mice at doses up to 1500 mg/kg/day, no evidence of carcinogenicity was found in mice (which is approximately four times the maximal recommended human daily dose of 2000 mg of metformin component of repaglinide and metformin hydrochloride tablets on a mg/m2 basis).
There was no evidence of a mutagenic potential of metformin alone in the following in vitro tests: Ames test (S. typhimurium), gene mutation test (mouse lymphoma cells), or chromosomal aberrations test (human lymphocytes). Results in the in vivo mouse micronucleus test were also negative.
In a rat fertility study, metformin was administered to male and female rats at doses up to 600 mg/kg/day. No adverse effects on fertility were observed (which is approximately three times the maximal recommended human daily dose of 2000 mg of metformin component of repaglinide and metformin hydrochloride tablets on a mg/m2basis).
14. Clinical Studies
There have been no clinical efficacy studies conducted with repaglinide and metformin hydrochloride tablets; however, bioequivalence of repaglinide and metformin hydrochloride tablets to repaglinide and metformin co-administered as individual tablets was demonstrated in healthy subjects.
14.1 Repaglinide as Add-on Combination Therapy With Metformin
A total of 83 patients with type 2 diabetes and inadequate glycemic control on metformin monotherapy were randomized to repaglinide as add-on therapy to metformin, repaglinide monotherapy, or continued treatment with metformin monotherapy. The repaglinide dosage was titrated for 4 to 8 weeks, followed by a 3-month maintenance period. Combination therapy with repaglinide and metformin resulted in a statistically significant improvement in HbA1c and fasting plasma glucose (FPG) compared to repaglinide or metformin monotherapy (Table 8). In this study where metformin dosage was kept constant, the combination therapy of repaglinide and metformin showed dose-sparing effects with respect to repaglinide. The improvement in HbA1c and FPG of the combination group was achieved at a lower daily repaglinide dosage than in the repaglinide monotherapy group.
|
| Repaglinide in Combination with Metformin
| Repaglinide monotherapy
| Metformin monotherapy
|
| N
| 27 | 28 | 27 |
| Median Final Dose (mg/day)
| 6 (repaglinide) 1500 (metformin) | 12 | 1500 |
| HbA1c (%)
| |||
| Baseline
| 8.3 | 8.6 | 8.6 |
| Change from baseline
| -1.4* | -0.4 | -0.3 |
| Fasting plasma glucose (mg/dL)
| |||
| Baseline
| 184 | 174 | 194 |
| Change from baseline
| -39* | +9 | -5 |
| 1 based on intent-to-treat analysis
* p< 0.05, for pairwise comparisons with repaglinide and metformin monotherapy. |
|||
16. How is Repaglinide and Metformin Tablets supplied
Repaglinide and metformin hydrochloride tablets are supplied as biconvex tablets available in 1 mg/500 mg (yellow) and 2 mg/500 mg (pink) strengths. 1 mg/500 mg tablets are debossed with 'V41' on one side and 'LU' on the other side and 2 mg/500 mg tablets are debossed with 'V42' on one side and 'LU' on the other side. The tablets are colored to indicate strength.
| 1 mg repaglinide/500 mg metformin HCl tablets (yellow) | Bottles of 100 | NDC 68180-490-01 |
| Bottles of 500 | NDC 68180-490-02 |
|
| Bottles of 1000 | NDC 68180-490-03 |
|
| 2 mg repaglinide/500 mg metformin HCl tablets (pink) | Bottles of 100 | NDC 68180-491-01 |
| Bottles of 500 | NDC 68180-491-02 |
|
| Bottles of 1000 | NDC 68180-491-03 |
Store at 25°C (77° F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Protect from moisture. Keep bottles tightly closed.
Dispense in tight containers with safety closures.
17. Patient Counseling Information
Explain the risks of lactic acidosis due to the metformin component, its symptoms and conditions that predispose to its development, as noted in WARNINGS AND PRECAUTIONS (5.1). Advise patients to discontinue repaglinide and metformin hydrochloride tablets immediately and to promptly notify their healthcare provider if unexplained hyperventilation, myalgia, malaise, unusual somnolence, dizziness, slow or irregular heartbeat, sensation of feeling cold (especially in the extremities), or other nonspecific symptoms occur. Gastrointestinal symptoms are common during initiation of metformin treatment and may occur during initiation of repaglinide and metformin hydrochloride tablets therapy; however, patients should consult their physician if they develop unexplained symptoms. Although gastrointestinal symptoms that occur after stabilization are unlikely to be drug related, such an occurrence of symptoms should be evaluated to determine if it may be due to lactic acidosis or other serious disease.
Counsel patients against excessive alcohol intake while receiving repaglinide and metformin hydrochloride tablets.
Inform patients about the importance of regular testing of renal function and hematological parameters when receiving treatment with repaglinide and metformin hydrochloride tablets.
Instruct patients to inform their doctor that they are taking repaglinide and metformin hydrochloride tablets prior to any surgical or radiological procedure, as temporary discontinuation of repaglinide and metformin hydrochloride tablets may be required until renal function has been confirmed to be normal [see WARNINGS AND PRECAUTIONS (5.1)].
Hypoglycemia
Inform patients that repaglinide and metformin hydrochloride tablets can cause hypoglycemia and instruct patients and their caregivers on self-management procedures including glucose monitoring and management of hypoglycemia. Inform patients that their ability to concentrate and react may be impaired as a result of hypoglycemia. In patients at higher risk for hypoglycemia and patients who have reduced symptomatic awareness of hypoglycemia, increased frequency of blood glucose monitoring is recommended [see WARNINGS AND PRECAUTIONS (5.2)].
Administration
Instruct patients to take repaglinide and metformin hydrochloride tablets within 30 minutes before meals. Instruct patients to skip their dose of repaglinide and metformin hydrochloride tablets when a meal is skipped.
Drug Interactions
Discuss potential drug interactions with patients and inform them of potential drug-drug interactions with repaglinide and metformin hydrochloride tablets. [see DRUG INTERACTIONS (7)].
Manufactured for:
Lupin Pharmaceuticals, Inc.
Baltimore, Maryland 21202
United States
Manufactured by:
Lupin Limited
Goa 403 722
INDIA
Revised: March 2018 ID#: 254694
| REPAGLINIDE AND METFORMIN HYDROCHLORIDE
repaglinide and metformin hydrochloride tablet |
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| REPAGLINIDE AND METFORMIN HYDROCHLORIDE
repaglinide and metformin hydrochloride tablet |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Lupin Pharmaceuticals, Inc. (089153071) |
| Registrant - LUPIN LIMITED (675923163) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LUPIN LIMITED | 677600414 | MANUFACTURE(68180-490, 68180-491) , PACK(68180-490, 68180-491) | |
More about metformin / repaglinide
- Check interactions
- Compare alternatives
- Imprints, shape & color data
- Side effects
- Dosage information
- During pregnancy
- Drug class: antidiabetic combinations


