Marquis (Canada)
This treatment applies to the following species: Company: Boehringer Ingelheim Animal Health
Company: Boehringer Ingelheim Animal Health
15% w/w Ponazuril Antiprotozoal Oral Paste for Horses
VETERINARY USE ONLY
DIN 02250128
INDICATION: Marquis® is indicated as an aid in reducing the clinical severity of Equine Protozoal Myeloencephalitis (EPM) caused by Sarcocystis neurona in horses; it is expected that approximately two horses out of three will respond favourably to treatment.
Description
Marquis® (15% w/w ponazuril) Antiprotozoal Oral Paste for Horses is supplied in ready-to-use syringes containing 127 g of paste. Each gram of paste contains 150 mg of ponazuril (15% w/w). Marquis® is designed to be delivered as an orally administered paste.
Each syringe barrel of Marquis® contains enough paste to treat one 545kg (1200 lb) horse for seven days, at a dose rate of 5 mg/kg body weight. The plunger contains a dosage ring calibrated for a dose rate of 5 mg/kg body weight and marked for horse weight from 273 kg (600 lb) to 545 kg (1200 lb) at 91 kg (200 lb) increments. The syringe barrels are packaged in units of four with four plungers. This package provides sufficient paste to treat one 545 kg (1200 lb) horse for 28 days at a dose rate of 5 mg/kg.
Ponazuril is an anticoccidial (antiprotozoal) compound with activity against several genera of the phylum Apicomplexa.
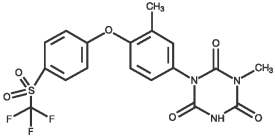
CHEMICAL NOMENCLATURE AND STRUCTURE: Ponazuril 1,3,5-Triazine-2,4,6(1H,3H,5H)-trione,1-methyl-3-[3-methyl-4-[4-[(trifluoromethyl)sulfonyl] phenoxy]phenyl]-(9CI)
Clinical Pharmacology
The activity of ponazuril has been demonstrated in several Apicomplexans1-6. Lindsay, Dubey and Kennedy7 showed that the concentration of ponazuril necessary to kill Sarcocystis neurona was 0.1 to 1.0 µg/mL. Furr and Kennedy8 evaluated the pharmacokinetics of ponazuril in serum and CSF in normal horses treated daily at 5 mg/kg for 28 days. The time to peak serum concentration (Tmax) was 18.20 (± 5.9) days and the maximum serum concentration (Cmax) was 5.59 (±0.92) µg/mL. The terminal elimination half-life for serum (calculated using Day 28 to 42 data) was 4.50 (± 0.57) days. In CSF, Tmax was 15.40 (± 7.9) days and Cmax was 0.21 (± 0.072) µg/mL.EFFECTIVENESS SUMMARY: A field study was conducted at six sites with seven investigators across the United States.9 The study was conducted using historical controls. In this study, each animal’s response to treatment was compared to its pre-treatment values.
The following standardized neurologic scale was used to grade the horses:
Grade 0 - Normal, no deficit detected
Grade 1 - Deficit just detected at normal gait
Grade 2 - Deficit easily detected and is exaggerated by backing, turning, swaying, loin pressure or neck extension
Grade 3 - Deficit very prominent on walking, turning, loin pressure or neck extension
Grade 4 - Stumbling, tripping and falling down spontaneously
Grade 5 - Recumbent, unable to rise
Improvement was defined as a decrease of at least one grade.
Naturally-occurring, clinical cases of EPM, characterized by signalment and laboratory diagnosis, were randomly allotted to one of two treatment doses (5 or 10 mg of ponazuril/kg/day for a period of 28 days), then evaluated for clinical changes through 118 days. Acceptance into the study was based on the results from a standardized neurological examination including radiography, serum S. neurona IgG level determination by Western Blot (WB), and a positive cerebrospinal fluid (CSF) for S. neurona IgG level by WB.
Response to treatment was determined by the investigator to be acceptable when a clinical improvement of at least one grade occurred by no later than 3 months after treatment, regardless of whether the CSF by WB was positive or negative.
In summary, at a daily dosage of 5 mg of ponazuril per kg of body weight, administered during 28 consecutive days, 31 of 46 horses (67.4%) improved by day 118, while 15 did not improve or continued to deteriorate. At a daily dosage of 10 mg of ponazuril per kg of body weight, administered during 28 consecutive days, 36 of 53 horses (67.9%) improved by Day 118, while 17 did not improve or continued to deteriorate.
Warnings
Not for use in horses intended for food. Keep this product, as well as any drug, out of the reach of children.Marquis Caution
Prior to treatment, EPM should be distinguished from other diseases that may cause ataxia in horses. Injuries or lameness may also complicate the evaluation of an animal with EPM. In most instances, ataxia due to EPM is asymmetrical and affects the hind limbs.
Clinicians should recognize that clearance of the parasite by ponazuril may not completely resolve the clinical signs attributed to the natural progression of the disease. The prognosis for animals treated for EPM may be dependent upon the severity of disease and the duration of the infection prior to treatment.
The reproductive safety of ponazuril has not been established in equines. Consequently, do not administer this product to breeding mares or stallions.
The administration of this product is restricted to horses in which Equine Protozoal Myeloencephalitis has been confirmed.
Adverse Reactions
In the field study, eight animals were noted to have unusual daily observations. Two horses exhibited blisters on the nose and mouth at some point in the field study, three animals showed a skin rash or hives for up to 18 days, one animal had loose stools throughout the treatment period, one had a mild colic on one day and one animal had a seizure while on medication. The association of these reactions to treatment was not established.ANIMAL SAFETY SUMMARY: Marquis® was administered to 24 adult horses (12 males and 12 females) in a target animal safety study. Three groups of 8 horses each received 0, 10, or 30 mg/kg (water as control, 2X and 6X for 5 mg/kg dose). Horses were dosed after feeding. One half of each group was treated for 28 days and the other half for 56 days followed by necropsy upon termination of treatment. There were several instances of loose feces in all animals in the study irrespective of treatment, sporadic inappetence and one horse at 10 mg/kg (2X) lost weight while on test. Loose feces were treatment related. Histopathological findings included moderate edema in the uterine epithelium of three of the four females in the 6X group (two treated for 28 days and one for 56 days).
DOSAGE: Marquis® Antiprotozoal Paste is to be administered orally, at a daily dose of 5 mg of ponazuril per kg of body weight, for a period of 28 consecutive days.
ADMINISTRATION:
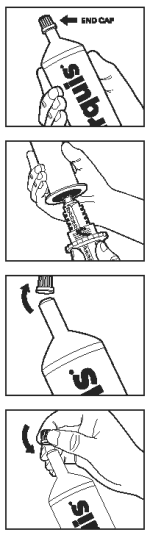
Paste syringe assembly:
Before administration, the syringe barrel and plunger require assembly. Ensure plunger is clean and dry.
Step 1. End cap must be on syringe barrel when inserting plunger.
Step 2. Carefully insert a plunger into the base of syringe barrel until it snaps into place, then remove end cap and gently apply pressure to the plunger until paste is seen at the tip of the syringe barrel.
Step 3. Return end cap to tip of paste syringe.
Assembling
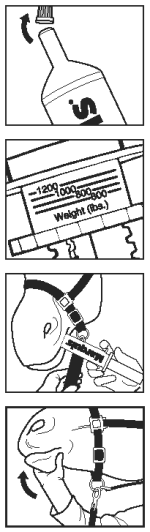
Administering Marquis® (ponazuril) to the horse:
Note: The paste syringe is a multi-dose package. Ensure that the correct dose is administered with each use.
Step 1. Remove end cap and gently apply pressure to the plunger until paste is seen at the tip of the syringe barrel. Return end cap to tip of paste syringe.
Step 2. Determine weight of horse and ensure the horse’s mouth contains no feed.
Step 3. To measure dose, dosage ring collar and barrel collar should be flush. Hold plunger and rotate dosage ring with the other hand to the weight of the horse.
Step 4. Remove end cap from tip of syringe barrel.
Step 5. The selected dose of paste should be deposited onto the back and top of the horse’s tongue. Introduce tip of paste syringe into the side of the horse’s mouth at the space between the front (incisor) and back (molar) teeth. Deposit paste on the horse’s tongue by depressing the plunger of the syringe as far as the dosage ring permits. Remove tip of syringe from horse’s mouth.
Step 6. To aid swallowing of paste, immediately raise horse’s head for a few seconds after dosing.
Step 7. Clean the tip of the syringe with a clean disposable towel and return end cap to the tip of the syringe barrel.
Step 8. For the next daily dose, repeat steps 1-7.
Administering
Storage
Store at a controlled room temperature of 15-30° C.How Supplied
Carton contains four (4) X 127 g syringe applicators and four syringe plungers.References
1. Mehlhorn, H., Ortmann-Falkenstein, G., Haberkorn, A.: (1984) The effects of the sym. Trianzinons on developmental stages of Eimeria tenella, E. maxima and E. acervulina: a light and electron microscopical study. Zeitschr Parsitenk 70: 173-182.
2. Bohrmann, R.: (1991) Treatment with toltrazuril in a natural outbreak of coccidiosis in calves. Dtsch. Tierarzl. Wschr. 98: 343-345.
3. Stafford, K.J., West, D.M., Vermunt, J.J., Pomroy, W., Adlington, B.A., Calder, S.M.: (1994) The effect of repeated doses of toltrazuril on coccidial oocyst output and weight gain in infected lambs. NZ Vet J 42(3): 117-119.
4. Haberkorn, A.G., Stoltefuss, D.I.J.: (1987) Studies on the activity spectrum of toltrazuril, a new anti-coccidial compound. Vet. Med. Review 1: 22-32.
5. Koudela, B., Vodstricilova, M., Klimes, B., Vladik, P., Vitovec, J.: (1991) Application of the anticoccidiosis drug toltrazuril in the coccidiosis of neonatal pigs. Veterinami medicina (Praha) 36: 657-663.
6. Benoit, E., Buronfosse, T., Delatour, P.: (1994) Effect of Cytochrome P-450 1A induction on enantioselective metabolism and pharmacokinetics of an aryltrifluoromethyl sulfide in the rat. Chirality 6(5): 372-377.
7. Lindsay, D.S., Dubey, J.P., Kennedy, T.J.: (2000) Determination of the activity of ponazuril against Sarcocystis neurona in cell cultures. Vet Parasit 92: 165-169.
8. Furr, M., Kennedy, T.: Pharmacokinetics of ponazuril in horses. Study 150-717 Bayer Corporation.
9. Furr, M., Andrews, F., MacKay, R., Reed, S., Bernard, W., Bain, F., Byars, D., Kennedy, T.: Treatment of equine protozoal myeloencephalitis (EPM) with various doses of ponazuril. Study Report 150-664 Bayer Corporation.
Boehringer Ingelheim Animal Health Canada Inc., 5180 South Service Road, Burlington ON L7L 5H4
MARQUIS® is a registered trademark of Boehringer Ingelheim Vetmedica GmbH, used under license.
86830167
B2R2
2050-8697-02
CPN: 1182152.2
5180 SOUTH SERVICE ROAD, BURLINGTON, ON, L7L 5H4
| Customer Care No.: | 1-800-567-1885 | |
| Technical Services No.: | 1-877-565-5501 | |
| Website: | www.boehringer-ingelheim.ca |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". Animalytix assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the Animalytix service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-08-27
