Homatropine Ophthalmic Solution: Package Insert / Prescribing Info
Package insert / product label
Generic name: homatropine hydrobromide
Dosage form: ophthalmic solution
Drug class: Mydriatics
Medically reviewed by Drugs.com. Last updated on Mar 3, 2025.
On This Page
Homatropine Ophthalmic Solution Description
Homatropine hydrobromide is an anticholinergic prepared as a sterile topical ophthalmic solution. Chemical Name: Benzeneacetic acid, a-hydroxy-,8-methyl-8-azabicyclo [3.2.1]-oct-3-yl ester, hydrobromide, endo-(±)-.
The active ingredient is represented by the chemical structure,
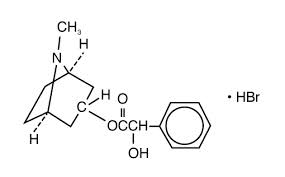
Each mL containes: Active: Homatropine Hydrobromide 5.0%. Preservative: Benzalkonium Chloride 0.005%.
Inactive: Boric Acid, Edetate Disodium, Potassium Chloride, Water for Injection. Boric Acid or Sodium Carbonate may be added to adjust the pH.
Indications and Usage for Homatropine Ophthalmic Solution
A moderately long-acting mydriatic and cycloplegic for cycloplegic refraction and in the treatment of inflammatory conditions of the uveal tract. For pre and postoperative states when mydriasis is required. Use as an optical aid in some
cases of axial lens opacities.
Homatropine Ophthalmic Solution - Clinical Pharmacology
This anticholinergic preparation blocks the responses of the sphincter muscle of the iris and the accommodative muscle of the ciliary body to cholinergic stimulation, producing pupillary dilation (mydriasis) and paralysis of accommodation (cycloplegia).
Contraindications
Contraindicated in persons with primary glaucoma or a tendency toward glaucoma, e.g. narrow anterior chamber angle, and in those persons showing hypersensitivity to any component of this preparation.
PREGNANCY SECTION
Pregnancy Category C. Animal reproduction studies have not been conducted with homatropine hydrobromine. It is also not known whether homatropine hydrobromide can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Homamtropine hydrobromide should be given to a pregnant woman ony if clearly needed.
Warnings
For topical use only - not for injection. Risk-benefit should be considered when the following medical problems exist: keratoconus (homatropine may produce fixed dilated pupil); Down's syndrome, children with brain damage and the elderly (increased susceptibility). In infants and small children, use with extreme caution. Excessive use in pediatric patients or certain individual with a history of susceptibility to belladonna alkaloids may produce systemic symptoms of homatropine poisoning (see overdosage section).
NURSING MOTHERS SECTION
It is not known whether this drug is excreted in human milk. Because many drgs are excreted in human milk,
caution should be exercised when homatropine hydrobromide is administered to a nursing woman.
PEDIATRIC USE SECTION
Homatropine should not be used during the first three months of life due to a possible association between the
cycloplegia produced and the development of amblyopia.
Warnings and Precautions
To avoid excessive systemic absorption, the lacrimal sac should be compressed by digital pressure for two to three minutes after installation. To avoid inducing angle closure glaucoma, an estimation of the depth of the angle of the anterior chamber should be made. Excessive topical use of this drug can potentially lead to a confusion state characterized by delirium, agitation, and rarely coma. This state is more apt to occur in the pediatric and geriatric age groups. The specific anti-dote for this systemic anticholinergic syndrome is inject able physostigmine salicylate.
Adverse Reactions/Side Effects
Transient symptoms of stinging and burning may occur. Prolonged use may produce local irritation characterized by follicular conjunctivitis, vascular congestion, edema, exudates, and an eczematoid dermatitis. Thirst dryness of mouth, eye irritation not present before therapy, or increased sensitivity of eyes to light may occur.
To report SUSPECTED ADVERSE REACTIONS, contact Altaire Pharmaceuticals, Inc. at (631)-722-5988.
Related/similar drugs
Overdosage
When signs and syptoms of homatropine toxicity develop (see adverse reactin section), Physostigmine
should be administered parenterally (for dosage refer to Goodman & Gilman or other pharmacology reference).
In infants and pediatric patients, the body surfaces must be kept moist.
Homatropine Ophthalmic Solution Dosage and Administration
For refraction, instill one or two drops topically in the eye(s). May be repeated in five or ten minutes if necessary. For uveitis, instill one or two drops topically up to every three to four hours. Individuals with heavily pigmented irides may require larger doses.
CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY SECTION
There have been no long-term studies done using homatropine hydrobmide in animals to evaluate
carcinogenic potential.
USER SAFETY WARNINGS SECTION
Patient should be advised not to drive or engage in other hazardous activities while pupils are dilated. Patient may experience sensitivity to light and should protect eyes in bright illumination during dilation. Parents should be warned not to get this preparation in their child’s mouth and to wash their own hands and the child's hands following administration. Do not touch dropper tip to any surfaces, as this may contaminate the solution.
| HOMATROPINE HYDROBROMIDE
homatropine hydrobromide ophthalmic solution |
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
| Labeler - OCuSOFT, Inc. (174939207) |
More about homatropine ophthalmic
- Check interactions
- Compare alternatives
- Side effects
- Dosage information
- During pregnancy
- Drug class: mydriatics
- Breastfeeding
- En español

