DentiPatch: Package Insert / Prescribing Info
Package insert / product label
Generic name: lidocaine
Dosage form: Transoral Delivery System
Drug classes: Group I antiarrhythmics, Local injectable anesthetics
Medically reviewed by Drugs.com. Last updated on Mar 25, 2025.
On This Page
DentiPatch Description
The DentiPatch® system contains a local anesthetic agent to be applied topically to the oral cavity. See INDICATIONS for specific uses.
Lidocaine is chemically designated as acetamide, 2-(diethylamino)-N-(2,6-dimethylphenyl)-, and has the following structural formula:
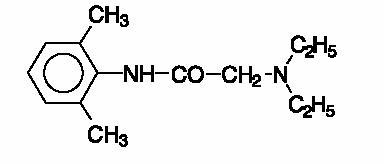
The molecular formula of lidocaine is C14H22N2O. The molecular weight is 234.34.
Each 2 cm2 patch contains lidocaine base as the active ingredient in the amount of 46.1 mg. Non-active ingredients include: karaya gum, glycerin, dipropylene glycol, lecithin, propylene glycol, aspartame, spearmint flavor, polyester film laminate and polyester-rayon fabric.
Each unit is sealed in a paper polyethylene-foil pouch.
DentiPatch - Clinical Pharmacology
Mechanism of action:
The DentiPatch® system is applied to the buccal mucosa to provide topical anesthesia by releasing lidocaine. Lidocaine stabilizes the neuronal membrane by inhibiting the ionic fluxes required for the initiation and conduction of impulses, thereby effecting local anesthetic action.
Onset and duration of action:
The DentiPatch® system acts on intact mucous membranes to produce local anesthesia.
DentiPatch® (Lidocaine Transoral Delivery System)
Anesthesia occurs usually within 2.5 minutes of application, is present for the duration of a 15 minute application period, and persists for approximately 30 minutes following removal.
Hemodynamics:
Excessive blood levels may cause changes in cardiac output, total peripheral resistance, and mean arterial pressure. These changes may be attributable to a direct depressant effect of the local anesthetic agent on various components of the cardiovascular system.
Pharmacokinetics and Metabolism:
Absorption
While systemic availability is not an objective when lidocaine is administered for topical anesthesia, limited absorption occurs following application of the patches to the buccal mucosa. The rate of absorption and percentage of dose absorbed depends upon several variables, including the concentration and total dose administered, the duration of exposure and the vascularity of the tissues at the site of application.
Although the total content of lidocaine base contained in each 2 cm2 system is 46.1 mg, the total amount of drug absorbed during 15 minutes of application is limited as drug delivery is confined to a fixed surface area. Corresponding blood levels of lidocaine following application are less than 0.1 μg/ml. Assuming the toxic range of lidocaine is approximately 5 μg/ml, the maximum plasma concentration achieved from this patch is, therefore, approximately 1/100th of this value.
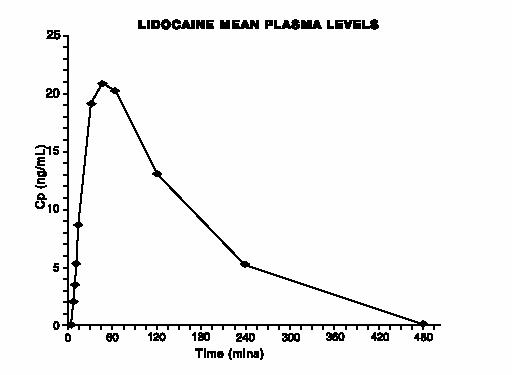
The figure shows the mean lidocaine plasma concentration following application of the 46.1 mg patch for 15 minutes to a group of normal male volunteers.
In this study, blood levels of lidocaine were compared following application of the patch and 5% Xylocaine® ointment. The maximum plasma concentration following the 46.1 mg patch was approximately 1/7 of those achieved by the ointment.
Another study compared the lidocaine plasma levels following applications of the patch, a 50 mg film of 5% topical ointment and an intravenous control in a cross-over design. The maximum plasma concentration following the 46.1 mg patch was approximately 1/5 of that achieved from the intravenous dose. The relevant mean pharmacokinetic parameters from this study are summarized in the table below. In addition, the table also indicates the range of AUC's obtained in two independent studies.
| PHARMACOKINETIC PARAMETERS | ||||||
|
Cmax = Maximum observed plasma concentration. Tmax = Time to maximum plasma concentration. Apparent dose = Calculated dose (estimated from the AUC0-α values and the known IV dose). t1/2=Apparent terminal elimination half-life. |
||||||
| Lidocaine content (mg) | Cmax (ng/mL) | Tmax (min) | Apparent Dose (mg) | t1/2 min | AUC0-α (ng•min/mL) Study 3005 Study 3006 |
|
| 46.1 | 16.5 (±7.9) | 28.6 (±12.9) | 1.55 (±0.77) | 102 (±25) | 2110 (±930) | 3679 (±1432) |
The apparent dose of lidocaine averaged 1.55 (± 0.77) mg from the 46.1 mg dosage. This compares to an apparent dose of 3.77 (±2.71) mg from the 5% topical ointment and 4.79 (±0.79) mg from the intravenous drug. In this study, the half-life of elimination of lidocaine following the patch was approximately 2 hours compared to a mean half-life of 112 (±20) minutes following the IV drug.
Metabolism and Excretion
Lidocaine is metabolized rapidly by the liver and metabolites and unchanged drug are excreted by the kidney.
Biotransformation includes oxidative N-dealkylation, ring hydroxylation, cleavage of the amide linkage, and conjugation (the hepatic ratio for lidocaine has been reported between 62% and 81% in man). Sequential oxidative N-dealkylation of lidocaine by the cytochrome P-450 system in hepatic microsomes produce two major metabolites, mono-ethylglyciniexylidide (MEGX) and glycine xylidide (GX), both of which have pharma-cologic activity.
Greater than 98% of an administered lidocaine dose can be recovered in the urine as metabolites and parent compound. Approximately 2% of an administered dose is excreted as intact drug in the urine over 24 hours. The primary metabolite found in the urine was 4-hydroxy 2, 6-xylidine, which comprised 73% of the dose in man following an oral dose of 3.0 mg/kg, MEGX and GX are found in small quantities: 4.0% and 2.5%, respectively. Other metabolic products recovered in the urine in amounts of less than 1.0% of an administered dose include 3-hydroxylidocaine, 3-hydroxyMEGX, and 2.6-xylidine. The metabolite 2,6-xylidine has unknown pharmacologic activity but has been demonstrated to be carcinogenic in rats. (See Carcinogenesis subsection of PRECAUTIONS).
Lidocaine crosses the blood-brain and placental barriers, presumably by passive diffusion. Factors such as acidosis and the use of CNS stimulants and depressants affect the CNS levels of lidocaine required to produce overt systemic effects. Objective adverse manifestations become increasingly apparent with increasing venous plasma levels above 6.0 μg free base per mL.
The extent of lidocaine protein binding is variable and is dependent upon the method of sample collection, lidocaine concentration, pH of sample and binding technique utilized. Under controlled conditions and using equilibrium dialysis techniques, the percentage of unbound lidocaine in serum and plasma has been reported to range from 21% to 39% (mean 28-30%). The major binding site of lidocaine in plasma is alpha,-1 acid glycoprotein.
Special Populations
Geriatric
No special studies have been conducted in this age group.
Pediatric
No special studies have been conducted in the pediatric age group.
Gender
No significant differences in absorption, etc. have been found in males and females. In a group of 30 volunteers (16 female, 14 male) receiving the 46.1 mg patch, the observed maximum plasma concentrations were 27.2 (±15.2) ng/mL at 45 (±12.5) minutes following application. Mean maximum concentrations were 31.5 (±17.4) ng/mL in females and 22.2 (±10.6) ng/mL in males.
Race
No specific studies were conducted comparing the pharmacokinetics in different races.
Renal Insufficiency
Renal dysfunction does not affect lidocaine kinetics but may increase the accumulation of metabolites.
Hepatic Insufficiency
Because of the rapid rate at which lidocaine is metabolized, any condition that affects liver function may alter lidocaine kinetics. The half-life may be prolonged two-fold or more in patients with liver dysfunction.
Drug-Drug Interactions
Lidocaine is metabolized by cytochrome P450 3A4-7 and 2D6. Inhibitors of these enzymes by H2 antagonists, antibiotics or some antiepileptics may elevate systemic lidocaine levels resulting in a prolonged effect.
Clinical Studies
The DentiPatch® system was studied in 275 volunteers who underwent needle insertion during four clinical studies. Application of the 46.1 mg patch for 15 minutes to the maxillary mucosae provided significantly more buccal anesthesia than a corresponding placebo at 5, 10, and 15 minutes.
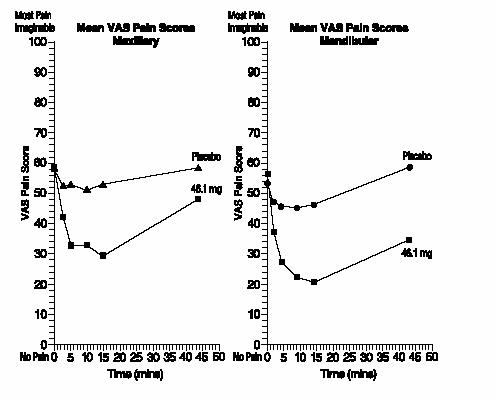
Similar findings were also observed following application of the patch to placement sites on both the maxillary and mandibular buccal mucosae. In one large, multi-center trial consisting of 100 subjects, differences in responsiveness to the 46.1 mg patch compared to placebo were assessed following application of the patches for 15-minute periods to each of these placement sites. Assessments were made at 2.5, 5, 10, and 15 minutes during application and again at 45 minutes.
For the mandibular placement sites, statistically significant decreases in pain scores from baseline were observed at 2.5, 5, 10, 15, and 45 minutes post-application. For each time point, the decrease in pain score for the 46.1 mg lidocaine group was statistically significantly greater than placebo. For the maxillary placement sites, statistically significant decreases in the pain scores were observed at 5, 10, 15, and 45 minutes for the 46.1 mg group, but was not statistically significantly different from placebo at 2.5 minutes.
The mean change in visual analog scores (VAS) from baseline at these two sites for the 46.1 mg group and placebo are shown for each time point in the figures below.
Mean Change In VAS Scores
The mean plasma concentrations of lidocaine which were observed following application of the active dosage at these time points showed little difference between mandibular and maxillary placement sites.
Indications and Usage for DentiPatch
The DentiPatch® system is indicated for the production of mild topical anesthesia of the accessible mucous membranes of the mouth prior to superficial dental procedures. It may also reduce the pain associated with injections of local anesthetic into the gingiva.
Contraindications
Lidocaine is contraindicated in patients with a known history of hypersensitivity to local anesthetics of the amide type or to other components of the DentiPatch® system.
Warnings
IN ORDER TO MANAGE POSSIBLE ADVERSE REACTIONS, RESUSCITATIVE EQUIPMENT, OXYGEN AND OTHER RESUSCITATIVE DRUGS MUST BE IMMEDIATELY AVAILABLE WHEN LOCAL ANESTHETIC AGENTS, SUCH AS LIDOCAINE, ARE ADMINISTERED TO MUCOUS MEMBRANES.
The DentiPatch® system should be used with extreme caution if there is sepsis or extremely traumatized mucosa in the area of application, since under such conditions there is the potential for rapid systemic absorption.
Precautions
General
The safety and effectiveness of lidocaine depend on proper dosage, correct technique, adequate precautions, and readiness for emergencies. Resuscitative equipment, oxygen, and other resuscitative drugs should be available for immediate use. (See WARNINGS and ADVERSE REACTIONS.) The lowest dosage that results in effective anesthesia should be used to avoid high plasma levels and serious adverse effects. Repeated doses of lidocaine may cause significant increases in blood levels with each repeated dose because of slow accumulation of the drug or its metabolites. Tolerance varies with the status of the patient. Debilitated, elderly patients, acutely ill patients, and children should be given reduced doses commensurate with their age and physical status. Lidocaine should also be used with caution in patients with severe shock or heart block.
The DentiPatch® system should be used with caution in patients with known drug sensitivities. Patients allergic to para-aminobenzoic acid derivatives (procaine, tetracaine, benzocaine, etc.) have not shown cross sensitivity to lidocaine.
Many drugs used during the conduct of anesthesia are considered potential triggering agents for familial malignant hyperthermia. Since it is not known whether amide-type local anesthetics may trigger this reaction and since the need for supplemental general anesthesia cannot be predicted in advance, it is suggested that a standard protocol for management should be available. Early unexplained signs of tachycardia, tachypnea, labile blood pressure and metabolic acidosis may precede temperature elevation. Successful outcome is dependent on early diagnosis, prompt discontinuance of the suspect triggering agent(s) and institution of treatment, including oxygen therapy, indicated supportive measures and dantrolene (consult dantrolene sodium intravenous package insert before using).
Information for patients
Use of the DentiPatch® system is frequently associated with mild local redness and infrequently with the development over a day or so of more severe local reactions. While these reactions are expected to resolve spontaneously, the concerned patient should be encouraged to report them to the practitioner.
Carcinogenesis, mutagenesis, impairment of fertility
Carcinogenesis: Long term study in animals to evaluate the carcinogenic potential of lidocaine has not been conducted.
A two-year oral toxicity study of 2,6-xylidine, a metabolite of lidocaine, conducted in both male and female rats, has shown that daily doses of 900 mg/m2 (150 mg/kg) resulted in carcinomas and adenomas of the nasal cavity. With daily doses of 300 mg/m2 (50 mg/kg), the increase in incidence of nasal carcinomas and/or adenomas in each sex of the rat were not statistically greater than the control group. In the low dose group of 90 mg/m2 (15 mg/kg) and control, no nasal tumors were observed. A rhabdomyosarcoma, a rare tumor, was observed in the nasal cavity of both male and female rats at the high dose of 900 mg/m2 (150 mg/kg). In addition, the compound caused subcutaneous fibromas and/or fibrosarcomas in both male and female rats and neoplastic nodules of the liver in the female rats with a significantly positive trend test; pairwise comparisons using Fisher's Exact Test showed significance only at the high dose of 900 mg/m2 (150 mg/kg).
Mutagenesis: The mutagenic potential of lidocaine HCI has been tested in the Ames Salmonella/mammalian microsome test and by analysis of structural chromosome aberrations in human lymphocytes in vitro, and by the mouse micronucleus test in vivo. There was no indication in these three tests of any mutagenic effects.
The mutagenicity of 2,6-xylidine, a metabolite of lidocaine, has been studied in different tests with mixed results. The compound was found to be weakly mutagenic in the Ames test only under metabolic activation conditions. In addition, 2,6-xylidine was observed to be mutagenic at the thymidine kinase locus, with or without activation, and induced chromosome aberrations and sister chromatid exchanges at concentrations at which the drug precipitated out of the solution (1.2 mg/mL). No evidence of genotoxicity was found in the in vivo assays measuring unscheduled DNA synthesis in rat hepatocytes, chromosome damage in polychromatic erythrocytes or preferential killing of DNA repair-deficient bacteria in liver, lung, kidney, testes and blood extracts from mice. However, covalent binding studies of DNA from liver and ethmoid turbinates in rats indicate that 2,6-xylidine may be genotoxic under certain conditions in vivo.
Use in Pregnancy
Teratogenic Effects. Pregnancy Category B. Reproduction studies have been performed in rats at doses up to 6.6 times the human dose and have revealed no evidence of harm to the fetus caused by lidocaine. There are, however, no adequate and well-controlled studies in pregnant women. Animal reproduction studies are not always predictive of human response. General consideration should be given to this fact before administering lidocaine to women of childbearing potential, especially during early pregnancy when maximum organogenesis takes place.
Nursing mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when lidocaine is administered to a nursing woman.
Adverse Reactions/Side Effects
Localized Reactions: During controlled clinical trials with this dosage form, the area of patch application was evaluated for oral irritation following removal. In the majority of instances, no irritation was observed. Minimal to moderate redness was reported, however, in fewer than 15% of the applications of the patch and placebo.
General Adverse Events: In controlled clinical trials, the percentage of subjects reporting adverse events was similar in the two treatment groups consisting of the 46.1 mg and placebo patches. Overall, the most frequently occurring adverse experiences irrespective of causality were taste perversion, stomatitis (including erythema and other types of mucosal reactions), headache and gingivitis.
The incidence of drug-related (definite, probably/highly probably) events was low in both treatment groups, however.
Other adverse experiences reported following the administration of lidocaine are similar in nature to those observed with other amide local anesthetic agents. These adverse experiences are, in general, dose-related and may result from high plasma levels caused by excessive dosage or rapid absorption, or may result from a hypersensitivity, idiosyncrasy or diminished tolerance on the part of the patient. Serious adverse experiences are generally systemic in nature. The following types are the most common reported.
Central nervous system: CNS manifestations are excitatory and/or depressant and may be characterized by lightheaded-ness, nervousness, apprehension, euphoria, confusion, dizziness, drowsiness, tinnitus, blurred or double vision, vomiting, sensations of heat, cold or numbness, twitching, tremors, convulsions, unconsciousness, respiratory depression and arrest. The excitatory manifestations may be very brief or may not occur at all, in which case the first manifestation of toxicity may be drowsiness merging into unconsciousness and respiratory arrest.
Drowsiness following the administration of lidocaine is usually an early sign of a high blood level of the drug and may occur as a consequence of rapid absorption:
Cardiovascular system:Cardiovascular manifestations are usually depressant and are characterized by bradycardia, hypotension, and cardiovascular collapse, which may lead to cardiac arrest.
Allergic: Allergic reactions are characterized by cutaneous lesions, urticaria, edema or anaphylactoid reactions. Allergic reactions may occur as a result of sensitivity either to the local anesthetic agent or to other ingredients in the formulation. Allergic reactions as a result of sensitivity to lidocaine are extremely rare and, if they occur, should be managed by conventional means. The detection of sensitivity by skin testing is of doubtful value.
Related/similar drugs
Overdosage
Acute emergencies from local anesthetics are generally related to high plasma levels encountered during therapeutic use of local anesthetics. (See ADVERSE REACTIONS, WARNINGS, and PRECAUTIONS.)
Management of local anesthetic emergencies: The first consideration is prevention, best accomplished by careful and constant monitoring of cardiovascular and respiratory vital signs and the patient's state of consciousness after each local anesthetic administration. At the first sign of change, oxygen should be administered.
The first step in the management of convulsions consists of immediate attention to the maintenance of a patent airway and assisted or controlled ventilation with oxygen and a delivery system capable of permitting immediate positive airway pressure by mask. Immediately after the institution of these ventilatory measures, the adequacy of the circulation should be evaluated, keeping in mind that drugs used to treat convulsions sometimes depress the circulation when administered intravenously. Should convulsions persist despite adequate respiratory support, and if the status of the circulation permits, small increments of an ultra-short acting barbiturate (such as thiopental or thiamylal) or a benzodiazepine (such as diazepam) may be administered intravenously. The clinician should be familiar, prior to use of local anesthetics, with these anticonvulsant drugs. Supportive treatment of circulatory depression may require administration of intravenous fluids and, when appropriate, a vasopressor as directed by the clinical situation (e.g., ephedrine).
If not treated immediately, both convulsions and cardiovascular depression can result in hypoxia, acidosis, bradycardia, arrhythmias and cardiac arrest. If cardiac arrest should occur, standard cardiopulmonary resuscitative measures should be instituted.
Dialysis is of negligible value in the treatment of acute overdosage with lidocaine.
DentiPatch Dosage and Administration
When the DentiPatch® system is used concomitantly with other products containing lidocaine, the total dose contributed by all formulations must be kept in mind.
Isolate the procedure area with cotton rolls and use suction as appropriate. Dry the tissue with air or gauze. Remove the DentiPatch® system from its packaging and peel off the clear protective liner. Immediately apply the DentiPatch® system using firm pressure. Allow the patch to remain in place until the desired anesthetic effect is produced but not for longer than 15 minutes. Experience in children is inadequate to recommend a pediatric dose at this time.
How is DentiPatch supplied
DentiPatch® system (46.1 mg/unit) - each 2.0 cm2 system contains 46.1 mg of lidocaine USP.
Dispenser carton of 50 systems NDC 57616-041-12
Do not store above 25°C.
Keep out of the reach of children.
May 1999
Package Insert
Noven Parmaceuticals, Inc.
100956-1
Manufactured by:
Noven Pharmaceuticals, Inc.
Miami, Florida 33186
| DENTIPATCH
lidocaine patch |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| Labeler - Noven Parmaceuticals, Inc. |
More about DentiPatch (lidocaine)
- Check interactions
- Compare alternatives
- Latest FDA alerts (10)
- Side effects
- Dosage information
- During pregnancy
- Drug class: group I antiarrhythmics
- Breastfeeding
