Astero Gel: Package Insert / Prescribing Info
Package insert / product label
Generic name: lidocaine
Dosage form: gel
Drug class: Topical anesthetics
Medically reviewed by Drugs.com. Last updated on Feb 27, 2025.
On This Page
1. Indications and Usage for Astero Gel
Astero® is indicated for associated pain, painful wounds and wound healing in either open and closed injuries or conditions. Conditions of pain include topical pain, postsurgical pain and pain associated w/various types of closed or open wounds. Conditions of closed wounds include soft tissue and bony injuries caused by contusions, hematomas, crush injuries and sprains/strains due to torsion, traction, compression and/or blunt trauma.
2. Astero Gel Dosage and Administration
Each pump of the Astero® bottle (30mL Airless Metered Dose bottle) (NDC:35781-2500-3)(90mL Airless Metered Dose bottle) (NDC:35781-2500-9) will deliver 0.25 mL of Astero® (10 mg Lidocaine Hydrochloride USP), enough to cover a 2-inch by 2-inch area of skin. A single application should not exceed 4 pumps of the M-DOSE bottle, equal to 1 gram of Astero®, (40 mg of Lidocaine Hydrochloride USP).
No more than 12 pumps of the M-DOSE bottle, approximately 3 grams of Astero® (120 mg Lidocaine Hydrochloride USP,) should be administered in any one day. Although the incidence of adverse effects with Astero® is quite low, caution should be exercised, particularly when employing large amounts, since the incidence of adverse effects is directly proportional to the total dose of local anesthetic agent administered.
2.1 Dosage for children: The recommended dose of Astero® varies as a function of age and weight. For children less than ten years who have a normal lean body mass and a normal lean body development, the maximum dose may be determined by the application of one of the standard pediatric drug formulas (e.g., Clark’s rule). For example, a child of five years weighing 50 lbs., the dose of lidocaine should not exceed 75-100 mg (approximately 1.9 to 2.5 grams of Astero®) when calculated according to Clark’s rule. In general, the maximum amount of lidocaine administered should not exceed 4.5 mg/kg (2.0 mg/lb) of body weight of the child. Do not use on children under 2 unless directed by a physician.
2.2 Administration: Apply as directed. Do not exceed 12 pumps in a twenty-four-hour (24-hour) period. One pump covers an area of 2 x2 inches.
3. Dosage Forms and Strengths
Astero® is a topical medicated hydrogel wound dressing. Each gram of Astero® contains 4% Lidocaine Hydrochloride USP (40mg).
4. Contraindications
Lidocaine Hydrochloride USP is contraindicated in patients with a known history of hypersensitivity to local anesthetics of the amide type or to other components of Astero®.
5. Warnings
FOR EXTERNAL USE ONLY. NOT FOR OPHTHALMIC USE. EXCESSIVE DOSAGE, OR SHORT INTERVALS BETWEEN DOSES, CAN RESULT IN HIGH PLASMA LEVELS OF LIDOCAINE AND SERIOUS ADVERSE EFFECTS, PATIENTS SHOULD BE INSTRUCTED TO STRICTLY ADHERE TO THE RECOMMENDED DOSAGE AND ADMINISTRATION GUIDELINES AS SET FORTH IN THIS PACKAGE INSERT. THE MANAGEMENT OF SERIOUS ADVERSE REACTIONS MAY REQUIRE THE USE OF RESUSCITATIVE EQUIPMENT, OXYGEN, AND OTHER RESUSCITATIVE DRUGS.
If irritation or sensitivity occurs or infection appears, discontinue use and institute appropriate therapy. Astero® should be used with caution in ill, elderly, debilitated patients and children who may be more sensitive to the systemic effects of Lidocaine Hydrochloride USP. In case of accidental ingestion get medical help or contact Poison Control Center right away.
6. Adverse Reactions/Side Effects
Adverse experiences following the administration of Lidocaine Hydrochloride USP are similar in nature to those observed with other amide local anesthetic agents. These adverse experiences are, in general, dose-related and may result from high plasma levels caused by excessive dosage or rapid absorption, or may result from a hypersensitivity, idiosyncrasy or diminished tolerance on the part of the patient. Serious adverse experiences are generally systemic in nature. The following types are those most commonly reported:
6.1 Central nervous system
CNS manifestations of Lidocaine toxicity are excitatory and/or depressant and may be characterized by lightheadedness, nervousness, apprehension, euphoria, confusion, dizziness, drowsiness, tinnitus, blurred or double vision, vomiting, sensations of heat, cold or numbness, twitching, tremors, convulsions, unconsciousness, respiratory depression and arrest. The excitatory manifestations may be very brief or may not occur at all, in which case the first manifestation of toxicity may be drowsiness merging into unconsciousness and respiratory arrest. Drowsiness following the administration of Lidocaine Hydrochloride USP is usually an early sign of a high blood level of the drug and may occur as a consequence of rapid absorption.
6.2 Cardiovascular system
Cardiovascular manifestations of Lidocaine toxicity are usually depressant and are characterized by bradycardia, hypotension, and cardiovascular collapse, which may lead to cardiac arrest.
6.3 Allergic
Allergic reactions are characterized by cutaneous lesions, urticaria, edema or anaphylactoid reactions. Allergic reactions may occur as a result of sensitivity either to the local anesthetic agent or to other components in the formulation. Allergic reactions as a result of sensitivity to Lidocaine Hydrochloride USP are extremely rare and, if they occur, should be managed by conventional means. The detection of sensitivity by skin testing is of doubtful value.
Related/similar drugs
7. Drug Interactions
7.1 Serious interactions:
Antiarrhythmic Drugs: Astero® should be used with caution in patients receiving Class I antiarrhythmic drugs (such as tocainide and mexiletine) since the toxic effects are additive and potentially synergistic. Bupivacaine liposome: Lidocaine Hydrochloride USP increases toxicity of Bupivacaine by increasing the free (unencapsulated) bupivacaine. Dofetilide: Lidocaine Hydrochloride USP increases effects of dofetilide through pharmacodynamic synergism. Lomitapide: Lidocaine Hydrochloride USP increases levels of lomitapide by affecting hepatic/intestinal enzymes CYP3A4 metabolism.
7.2 General interactions:
Drugs metabolized via CYP3A4 enzyme: (ex. Antipsychotics, SSRIs, TCAs, many chemotherapeutics, calcium channel blockers, benzodiazepines) Lidocaine Hydrochloride USP may increase serum levels of many drugs metabolized by hepatic / intestinal CYP3A4 enzymes. Drugs that affect hepatic CYP1A2 enzyme: (ex. Quinolone antibiotics, cimetidine, barbiturates, benzodiazepines, erythromycin) May increase serum Lidocaine Hydrochloride USP levels by decreasing Lidocaine Hydrochloride USP metabolism by CYP1A2 enzyme.
8. Use In Specific Populations
8. Use in Specific Populations:
8.1 Use in Pregnancy:
Teratogenic Effects of Lidocaine Hydrochloride USP. Pregnancy Category B. Reproduction studies have been performed in rats at doses up to 6.6 times the human dose and have revealed no evidence of harm to the fetus caused by Lidocaine Hydrochloride USP. There are, however, no adequate and well-controlled studies in pregnant women. Animal reproduction studies are not always predictive of human response. General consideration should be given to this fact before administering Lidocaine Hydrochloride USP to women of childbearing potential, especially during early pregnancy when maximum organogenesis takes place.
8.2 Labor and Delivery:
Lidocaine Hydrochloride USP is not contraindicated in labor and delivery. Should Astero® be used concomitantly with other products containing Lidocaine Hydrochloride USP, the total dose contributed by all formulations must be kept in mind.
8.3 Nursing Mothers:
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Lidocaine Hydrochloride USP is administered to a nursing woman.
8.4 Pediatric use:
Dosage in children should be reduced, commensurate with age, body weight and physical condition. Caution must be taken to avoid over dosage when applying Astero® to large areas of injured or abraded skin, since the systemic absorption of Lidocaine Hydrochloride USP may be increased under such conditions. Do not use on children under 2 unless directed by a physician.
9 Over Dosage:
Acute emergencies from local anesthetics are generally related to high plasma levels encountered during therapeutic use of local anesthetics. (see ADVERSE REACTIONS, WARNINGS, and PRECAUTIONS).––
9.1 Management of local anesthetic emergencies:
The first consideration is prevention, best accomplished by careful and constant monitoring of cardiovascular and respiratory vital signs and the patient’s state of consciousness after each local anesthetic administration. At the first sign of change, oxygen should be administered. The first step in the management of convulsions consists of immediate attention to the maintenance of a patent airway and assisted or controlled ventilation with oxygen and a delivery system capable of permitting immediate positive airway pressure by mask.
Immediately after the institution of these ventilatory measures, the adequacy of the circulation should be evaluated, keeping in mind that drugs used to treat convulsions sometimes depress the circulation when administered intravenously. Should convulsions persist despite adequate respiratory support, and if the status of the circulation permits, small increments of an ultra-short acting barbiturate (such as thiopental or thiamylal) or a benzodiazepine (such as diazepam) may be administered intravenously. The clinician should be familiar, prior to use of local anesthetics, with these anticonvulsant drugs. Supportive treatment of circulatory depression may require administration of intravenous fluids and, when appropriate, a vasopressor as directed by the clinical situation (e.g., ephedrine).
If not treated immediately, both convulsions and cardiovascular depression can result in hypoxia, acidosis, bradycardia, arrhythmias and cardiac arrest. If cardiac arrest should occur, standard cardiopulmonary resuscitative measures should be instituted.
Dialysis is of negligible value in the treatment of acute over dosage with Lidocaine Hydrochloride USP. The oral LD50 of Lidocaine HCI USP in non-fasted female rats is 459 (346-773) mg/kg (as the salt) and 214 (159-324) mg/kg (as the salt) in fasted female rats.
10. Astero Gel Description
A soothing hydrogel wound dressing that promotes a moist wound environment that is ideal for the healing process
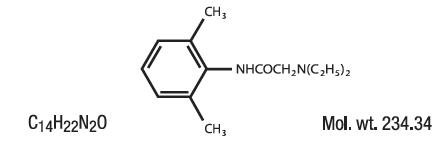
ASTERO contains Lidocaine HCl USP 4%, which is chemically designated as acetamide, 2-(diethylamino)-N-(2,6-dimethylphenyl)-, and has the following structural formula:
11. Astero Gel - Clinical Pharmacology
11.1 Mechanism of action:
Astero, when applied to wounds, provides for analgesia by the release of lidocaine from the gel into the epidermal and dermal layers of the skin and by the accumulation of lidocaine in the vicinity of dermal pain receptors and nerve endings. Astero® is a hydrated polymer (Hydrogel) wound dressing containing 4% w/w Lidocaine HCl USP. By providing moisture to the wound, Astero® creates a moist healing environment, which promotes granulation, epithelialization, and autolytic debridement. The high water content of hydrogel dressings cools the wound, producing pain relief that can last up to 6 hours. Dressing-change discomfort is also reduced because Astero® doesn’t adhere to the wound surface.
Astero® releases Lidocaine Hydrochloride USP from the Neutral (pH 7-7.2) hydrogel to stabilize the neuronal membrane by inhibiting the ionic fluxes required for initiation and conduction of impulses, thereby effecting local anesthetic action.
11.2 Onset of anesthesia:
Astero® effects local, topical anesthesia. The onset of action is 3-5 minutes.
11.3 Hemodynamics:
Excessive blood levels may cause changes in cardiac output, total peripheral resistance, and mean arterial pressure. These changes may be attributable to a direct depressant effect of the local anesthetic agent on various components of the cardiovascular system.
11.4 Pharmacokinetics and metabolism:
Lidocaine Hydrochloride USP may be absorbed following topical administration to mucous membranes or open wounds, its rate and extent of absorption depending upon the specific site of application, duration of exposure, concentration, and total dosage. Lidocaine Hydrochloride USP is also well-absorbed from the gastrointestinal tract, but little intact drug appears in the circulation because of biotransformation in the liver. Lidocaine Hydrochloride USP is metabolized rapidly by the liver, and metabolites and unchanged drug are excreted by the kidneys. Biotransformation includes oxidative N-dealkylation, ring hydroxylation, cleavage of the amide linkage, and conjugation. N-dealkylation, a major pathway of biotransformation, yields the metabolites monoethylglycinexylidide and glycinexylidide. The pharmacological/toxicological actions of these metabolites are similar to, but less potent than, those of Lidocaine Hydrochloride USP. Approximately 90% of Lidocaine Hydrochloride USP administered is excreted in the form of various metabolites, and less than 10% is excreted unchanged. The primary metabolite in urine is a conjugate of 4-hydroxy-2,6-dimethylaniline. The plasma binding of Lidocaine Hydrochloride USP is dependent on drug concentration, and the fraction bound decreases with increasing concentration. At concentrations of 1 to 4μg of free base per mL, 60 to 80 percent of Lidocaine Hydrochloride USP is protein bound. Binding is also dependent on the plasma concentration of the alpha-l-acid glycoprotein. Lidocaine Hydrochloride USP crosses the blood-brain and placental barriers, presumably by passive diffusion. Studies of Lidocaine Hydrochloride USP metabolism following intravenous bolus injections have shown that the elimination half-life of this agent is typically 1.5 to 2.0 hours. Because of the rapid rate at which Lidocaine Hydrochloride USP is metabolized, any condition that affects liver function may alter Lidocaine Hydrochloride USP kinetics. The half-life may be prolonged two-fold or more in patients with liver dysfunction. Renal dysfunction does not affect Lidocaine Hydrochloride USP kinetics but may increase the accumulation of metabolites.
Factors such as acidosis and the use of CNS stimulants and depressants affect the CNS levels of Lidocaine Hydrochloride USP required to produce overt systemic effects. Objective adverse manifestations become increasingly apparent with increasing venous plasma levels above 6.0μg free base per mL. In the rhesus monkey arterial blood levels of 18-21μg/mL have been shown to be threshold for convulsive activity.
12. Nonclinical Toxicology
Studies of Lidocaine Hydrochloride USP in animals to evaluate the carcinogenic and mutagenic potential or the effect on fertility have not been conducted.
13. How is Astero Gel supplied
ASTERO® (Lidocaine HCl USP 4%)
1.0 oz (28.5g) 30mL Airless Pump - NDC 35781-2500-3
3.04 oz (86.0) 90mL Airless Pump - NDC 35781-2500-9
Gensco Pharma
8550 NW 33rd St Suite 200
Doral, FL 33122
14. Storage and Handling
STORE AND DISPOSE OF THIS AND ALL MEDICATIONS OUT OF REACH OF CHILDREN AND PETS. All prescriptions using this product shall be pursuant to state statutes as applicable. This product may be administered only under a physician’s supervision. There are no implied or explicit claims on the therapeutic equivalence. Store at 25ºC (77ºF); excursions permitted to 15º-30ºC (59º-86º F). See USP Controlled Room Temperature. Protect from freezing.
| ASTERO
dressing, wound, drug |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Gensco Laboratories, LLC (831042325) |
More about Astero (lidocaine topical)
- Check interactions
- Compare alternatives
- Latest FDA alerts (8)
- Side effects
- Dosage information
- During pregnancy
- Drug class: topical anesthetics
- Breastfeeding
Professional resources
Other brands
Lidocaine Viscous, Lidocan Patch, ZTlido, Xylocaine Jelly, ... +34 more

