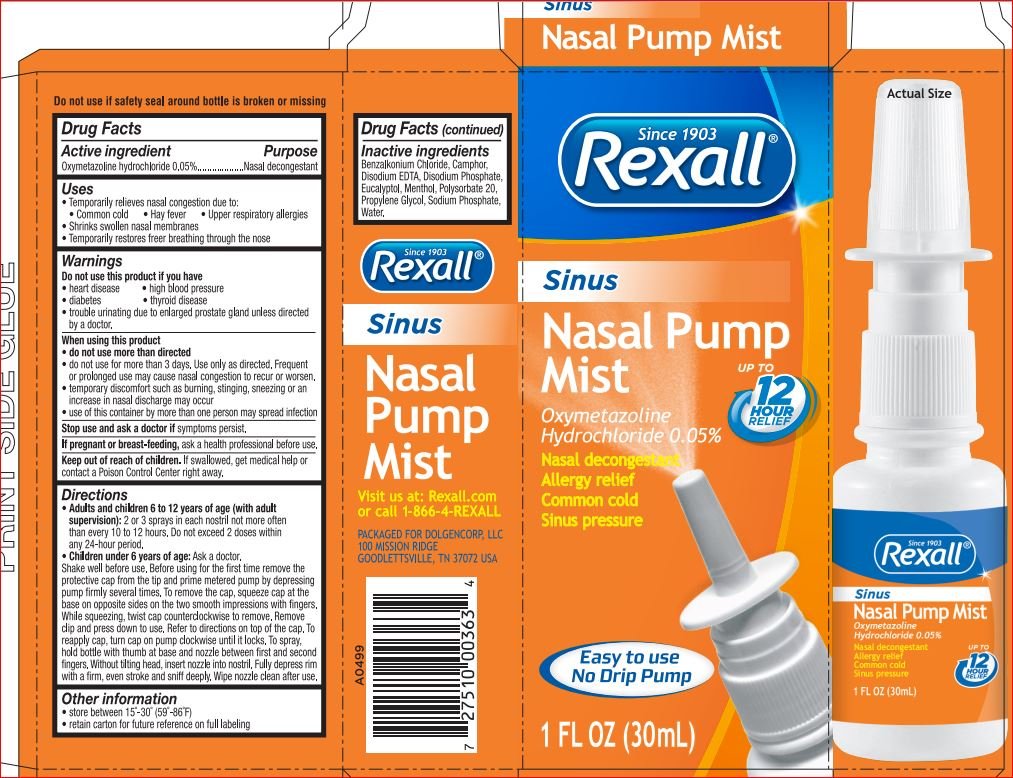
Rexall Sinus Nasal Pump Mist
Dosage form: liquid
Ingredients: Oxymetazoline Hydrochloride 0.05g in 100mL
Labeler: Dollar General
NDC code: 55910-778
Medically reviewed by Drugs.com. Last updated on Sep 24, 2024.
Oxymetazoline Hydrochloride 0.05% Nesal Decongestant
temporarily relief nasal decongestion due to:
- common cold
- hay fever
- upper respiratory allergies
- shrinks swollen membranes
- temporarily restores freer breathing through the nose
Do not use this product if person has heart disease, high blood pressure, thyroid disease, diabetes, or if adult has difficulty in urination due to enlargement of the prostate gland unless directed by a doctor.
When using this product
Do not exceed recommended dosage.
This product may cause temporary discomfort such as burning, stinging, sneezing, or an increase in nasal discharge.
The use of this container by more than one person may spread infection
Stop use and ask doctor if
Symptoms persist
If pregnant or breast-feeding, ask a health professional before use
Directions. Adults and children 6 to under 12 years of age (with adult supervision): 2 or 3 sprays in each nostril not more often than every 10 to 12 hours. Do not exceed 2 doses in any 24-hour period. Children under 6 years of age: consult a doctor
shake well before each use. Before using for the first time remove the protective cap from the tip and prime mitered pump by depressing pump firmly several times. To remove the cap, squeeze cap at the base on opposite sides on the two smooth impressions with fingers. While squeezing, twist cap counterclockwise to remove. Remove clip and press down to use. Refer to directions on top of each cap. To reapply cap, turn cap on pump clockwise until it locks. To spray, hold bottle with thumb at base and nozzle between first and second flingers. Without tilting head, insert nozzle into nostril. Fully depress rim with a firm, even stroke and sniff deeply. Wipe nozzle clean after use.
Benzalkonium Chloride
Camphor
Disodium EDTA
Disodium Phosphate
Eucalyptol
Menthol
Polysorbate 20
Propylene Glycol
Sodium Phosphate
Water
| REXALL SINUS NASAL PUMP MIST
oxymetazoline hydrochloride 0.05% liquid |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| Labeler - Dollar General (068331990) |
| Registrant - Product Quest Mfg (927768135) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Product Quest Mfg | 927768135 | manufacture(55910-778), label(55910-778) | |
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
