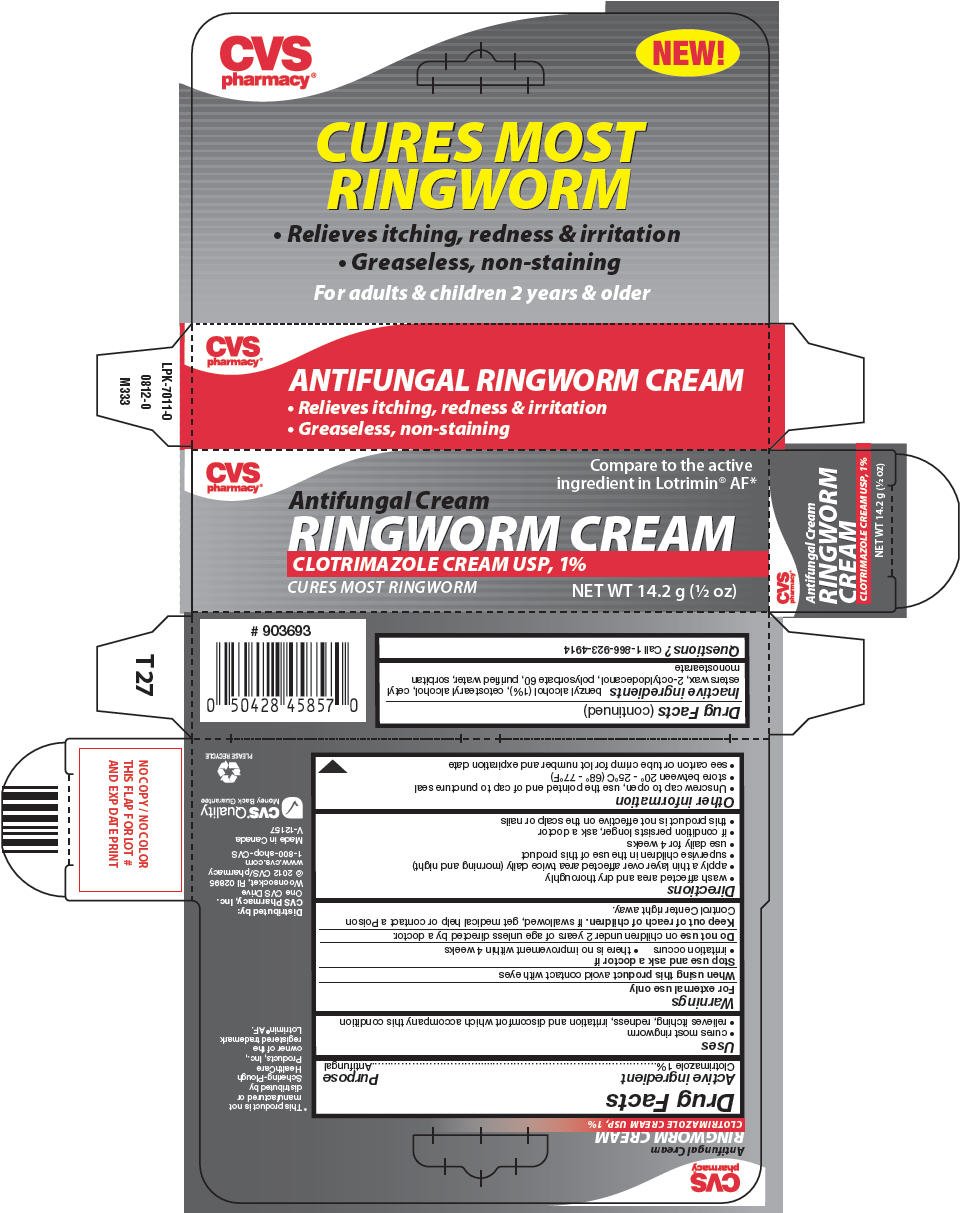
CVS Antifungal Ringworm
Dosage form: cream
Ingredients: Clotrimazole 1g in 100g
Labeler: CVS Pharmacy
NDC code: 59779-514
Medically reviewed by Drugs.com. Last updated on Dec 18, 2024.
Antifungal Ringworm
Drug Facts
Clotrimazole 1%
Antifungal
- cures most ringworm
- relieves itching, redness, irritation and discomfort which accompany this condition
For external use only
When using this product avoid contact with eyes
- irritation occurs
- there is no improvement within 4 weeks
Do not use on children under 2 years of age unless directed by a doctor.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
- wash affected area and dry thoroughly
- apply a thin layer over affected area twice daily (morning and night)
- supervise children in the use of this product
- use daily for 4 weeks
- if condition persists longer, ask a doctor
- this product is not effective on the scalp or nails
- Unscrew cap to open, use the pointed end of cap to puncture seal
- store between 20° - 25°C (68° - 77°F)
- see carton or tube crimp for lot number and expiration date
benzyl alcohol (1%), cetostearyl alcohol, cetyl esters wax, 2-octyldodecanol, polysorbate 60, purified water, sorbitan monostearate
Call 1-866-923-4914
Distributed by:
CVS Pharmacy, Inc.
One CVS Drive
Woonsocket, RI 02895
CVS
pharmacy®
Compare to the active
ingredient in Lotrimin® AF*
Antifungal Cream
RINGWORM CREAM
CLOTRIMAZOLE CREAM USP, 1%
CURES MOST RINGWORM
NET WT 14.2 g (½ oz)
| CVS
ANTIFUNGAL RINGWORM
clotrimazole cream |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - CVS Pharmacy (062312574) |
| Registrant - Taro Pharmaceuticals U.S.A., Inc (145186370) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Taro Pharmaceuticals Inc. | 206263295 | MANUFACTURE(59779-514) | |
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
