3M Avagard
Dosage form: lotion
Ingredients: Chlorhexidine Gluconate 8.3mg in 1mL, Alcohol 506.3mg in 1mL
Labeler: 3M Company
NDC code: 17518-051
Medically reviewed by Drugs.com. Last updated on Jun 20, 2025.
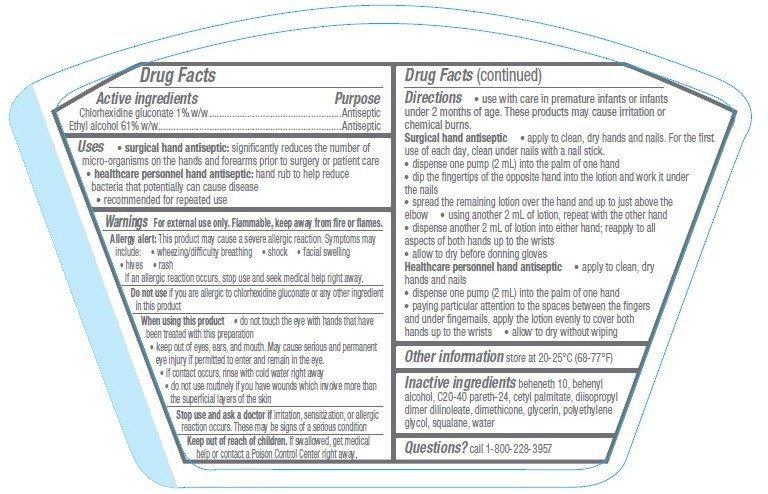
Drug Facts
Chlorhexidine gluconate 1% w/w
Ethyl alcohol 61% w/w
Antiseptic
Antiseptic
- surgical hand antiseptic: significantly reduces the number of micro-organisms on the hands and forearms prior to surgery or patient care
- healthcare personnel hand antiseptic: hand rub to help reduce bacteria that potentially can cause disease
- recommended for repeated use
For external use only. Flammable, keep away from fire or flames.
Allergy alert: This product may cause a severe allergic reaction. Symptoms may include:
- wheezing/difficulty breathing
- shock
- facial swelling
- hives
- rash
If an allergic reaction occurs, stop use and seek medical help right away.
Do not use if you are allergic to chlorhexidine gluconate or any other ingredient in this product
- do not touch the eye with hands that have been treated with this preparation
- keep out of eyes, ears, and mouth. May cause serious and permanent eye injury if permitted to enter and remain in the eye.
- if contact occurs, rinse with cold water right away
- do not use routinely if you have wounds which involve more than the superficial layers of the skin
Stop use and ask a doctor if irritation, sensitization, or allergic reaction occurs. These may be signs of a serious condition
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
- use with care in premature infants or infants under 2 months of age. These products may cause irritation or chemical burns.
Surgical hand antiseptic
- apply to clean, dry hands and nails. For the first use of each day, clean under nails with a nail stick.
- dispense one pump (2 mL) into the palm of one hand
- dip the fingertips of the opposite hand into the lotion and work it under the nails
- spread the remaining lotion over the hand and up to just above the elbow
- using another 2 mL of lotion, repeat with the other hand
- dispense another 2 mL of lotion into either hand; reapply to all aspects of both hands up to the wrists
- allow to dry before donning gloves
Healthcare personnel hand antiseptic
- apply to clean, dry hands and nails
- dispense one pump (2 mL) into the palm of one hand
- paying particular attention to the spaces between the fingers and under fingernails, apply the lotion evenly to cover both hands up to the wrists
- allow to dry without wiping
store at 20-25°C (68-77°F)
beheneth 10, behenyl alcohol, C20-40 pareth-24, cetyl palmitate, diisopropyl dimer dilinoleate, dimethicone, glycerin, polyethylene glycol, squalane, water
Call 1-800-228-3957
3M
NDC 17518-051-01
Avagard™
(Chlorhexidine Gluconate 1% Solution and Ethyl Alcohol 61% w/w)
Surgical and Healthcare Personnel Hand Antiseptic āwith āMoisturizers
Peel to read Drug Facts Panel before use
Made in U.S.A. for 3M Health Care
2510 Conway Ave., St. Paul, MN 55144
1-800-228-3957
www.3M.com/infectionprevention
© 2017, 3M. All rights reserved. The shape and colors of the bottle and wall bracket are trademarks of 3M.
Patent: 3M.com/Patents
MAL10110039XC
Flammable, keep away from fire or flame, heat, sparks and sources of static discharge.
LOT 0000000000
EXP 0000-00-00
16 fl oz • 500 mL
REF 9200

| 3M AVAGARD
chlorhexidine gluconate and alcohol lotion |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - 3M Company (006173082) |
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
