P P 8 Pill: white, three-sided, 7mm
The pill with imprint P P 8 (White, Three-sided, 7mm) has been identified as Hydromorphone Hydrochloride 8 mg and is used for Chronic Pain, Anesthetic Adjunct, Cough, and Pain. It belongs to the drug class Opioids (narcotic analgesics) and is classified as CSA Schedule 2 (High potential for abuse).
Images for P P 8
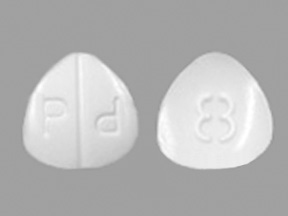
Hydromorphone Hydrochloride
- Imprint
- P P 8
- Strength
- 8 mg
- Color
- White
- Size
- 7.00 mm
- Shape
- Three-sided
- Availability
- Prescription only
- Drug Class
- Opioids (narcotic analgesics)
- Pregnancy Category
- C - Risk cannot be ruled out
- CSA Schedule
- 2 - High potential for abuse
- Labeler / Supplier
- Rhodes Pharmaceuticals
- Inactive Ingredients
-
lactose,
magnesium stearate,
sodium metabisulfite
Note: Inactive ingredients may vary.
Labelers / Repackagers
| NDC Code | Labeler / Repackager |
|---|---|
| 42858-0303 | Rhodes Pharmaceuticals |
| 42858-0338 | Rhodes Pharmaceuticals |
| 59011-0458 (Discontinued) | Purdue Pharma LP |
Related images for "P P 8"
More about hydromorphone
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (450)
- Drug images
- Latest FDA alerts (12)
- Side effects
- Dosage information
- Patient tips
- During pregnancy
- Support group
- Drug class: Opioids (narcotic analgesics)
- Breastfeeding
Patient resources
- Hydromorphone drug information
- Hydromorphone injection
- Hydromorphone rectal
- Hydromorphone Extended-Release Tablets
- Hydromorphone Liquid
Other brands
Dilaudid, Dilaudid-HP, Exalgo, Palladone
Professional resources
- Hydromorphone Hydrochloride monograph
- Hydromorphone (FDA)
- Hydromorphone Extended Release (FDA)
- Hydromorphone Injection (FDA)
- Hydromorphone Oral Solution (FDA)
Other brands
Dilaudid, Dilaudid-HP, Exalgo, Palladone
Related treatment guides
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.