Vermox: Package Insert / Prescribing Info
Package insert / product label
Generic name: mebendazole
Dosage form: tablet, chewable
Drug class: Anthelmintics
Medically reviewed by Drugs.com. Last updated on Mar 25, 2025.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Drug Interactions
- Use In Specific Populations
- Overdosage
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
VERMOX™ (mebendazole) chewable tablets for oral use
Initial U.S. Approval: 1974
Indications and Usage for Vermox
VERMOX™ is an anthelmintic indicated for the treatment of patients two years of age and older with gastrointestinal infections caused by:
- Ancylostoma duodenale (hookworm),
- Ascaris lumbricoides (roundworm),
- Enterobius vermicularis (pinworm),
- Necator americanus (hookworm), and
- Trichuris trichiura (whipworm), (1).
Vermox Dosage and Administration
Adults and Pediatrics: The tablet may be chewed, swallowed, or crushed and mixed with food (2).
| Pinworm (enterobiasis) | Whipworm (trichuriasis) | Roundworm (ascariasis) | Hookworm | |
|---|---|---|---|---|
| Dose | 1 tablet once | 1 tablet morning and evening for 3 consecutive days | 1 tablet morning and evening for 3 consecutive days | 1 tablet morning and evening for 3 consecutive days |
Dosage Forms and Strengths
- Chewable Tablet: 100 mg (3)
Contraindications
- Patients with a known hypersensitivity to the drug or its excipients (4).
Warnings and Precautions
- Risk of Convulsions: Convulsions in infants below the age of 1 year have been reported (5.1).
- Hematologic Effects: Neutropenia and agranulocytosis have been reported in patients receiving mebendazole at higher doses and for prolonged duration. Monitor blood counts in these patients (5.2)
- Metronidazole and Serious Skin Reactions: Stevens-Johnson syndrome/toxic epidermal necrolysis (SJS/TEN) have been reported with the concomitant use of mebendazole and metronidazole. Avoid concomitant use of mebendazole and metronidazole (5.3).
Adverse Reactions/Side Effects
- Adverse reactions reported in clinical trials were anorexia, abdominal pain, diarrhea, flatulence, nausea, vomiting and rash. (6.1).
To report SUSPECTED ADVERSE REACTIONS, contact Janssen Pharmaceuticals, Inc. at 1-800-JANSSEN (1-800-526-7736) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 6/2017
Full Prescribing Information
1. Indications and Usage for Vermox
VERMOX™ is indicated for the treatment of patients two years of age and older with gastrointestinal infections caused by Ancylostoma duodenale (hookworm), Ascaris lumbricoides (roundworm), Enterobius vermicularis (pinworm), Necator americanus (hookworm), and Trichuris trichiura (whipworm).
2. Vermox Dosage and Administration
The recommended dosage for VERMOX™ is described in Table 1 below. The same dosage schedule applies to adults and pediatric patients two years of age and older. The tablet may be chewed, swallowed, or crushed and mixed with food.
| Pinworm (enterobiasis) | Whipworm (trichuriasis) | Roundworm (ascariasis) | Hookworm | |
|---|---|---|---|---|
| Dose | 1 tablet Once | 1 tablet morning and evening for 3 consecutive days | 1 tablet morning and evening for 3 consecutive days | 1 tablet morning and evening for 3 consecutive days |
If the patient is not cured three weeks after treatment, a second course of treatment is advised. No special procedures, such as fasting or purging, are required.
3. Dosage Forms and Strengths
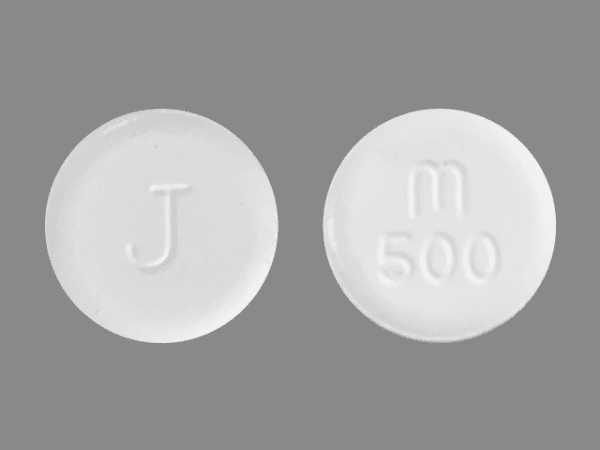
Chewable Tablet: 100 mg, round, flat radius-edged white to yellowish chewable tablet that is debossed with "M/100" on one side and "J" on the other side.
4. Contraindications
VERMOX™ is contraindicated in persons with a known hypersensitivity to the drug or its excipients.
5. Warnings and Precautions
5.1 Risk of Convulsions
Although VERMOX™ is approved for use in children two years of age and older, convulsions have been reported in infants below the age of 1 year during post-marketing experience with mebendazole, including VERMOX™ [see Adverse Reactions (6.2) and Use in Specific Populations (8.4)].
5.2 Hematologic Effects
Agranulocytosis and neutropenia have been reported with mebendazole use at higher doses and for more prolonged durations than is recommended for the treatment of soil-transmitted helminth infections. Monitor blood counts if VERMOX™ is used at higher doses or for prolonged duration.
6. Adverse Reactions/Side Effects
6.1 Clinical Studies
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of mebendazole was evaluated in 6276 subjects who participated in 39 clinical trials for treatment of single or mixed parasitic infections of the gastrointestinal tract. In these trials, the formulations, dosages and duration of mebendazole treatment varied. Adverse reactions reported in mebendazole-treated subjects from the 39 clinical trials are shown in Table 2 below.
| Adverse Reaction(s) |
|---|
|
| Gastrointestinal Disorders |
| Anorexia |
| Abdominal Pain |
| Diarrhea |
| Flatulence |
| Nausea |
| Vomiting |
| Skin and Subcutaneous Tissue Disorders |
| Rash |
6.2 Postmarketing Experience
The following adverse reactions have been identified in adult and pediatric patients postmarketing with mebendazole formulations and dosages other than the VERMOX™ 100 mg chewable tablet. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
| Adverse Reaction(s) | |
|---|---|
|
|
| Blood and Lymphatic System Disorders | Agranulocytosis, Neutropenia |
| Immune System Disorders | Hypersensitivity including anaphylactic reactions |
| Nervous System Disorders | Convulsions, Dizziness |
| Hepatobiliary Disorders | Hepatitis, Abnormal liver tests |
| Renal and Urinary Disorders | Glomerulonephritis |
| Skin and Subcutaneous Tissue Disorders | Toxic epidermal necrolysis, Stevens-Johnson syndrome, Exanthema, Angioedema, Urticaria, Alopecia |
Related/similar drugs
7. Drug Interactions
Concomitant use of mebendazole, including VERMOX™, and metronidazole should be avoided [see Warnings and Precautions (5.3)].
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
The available published literature on mebendazole use in pregnant women has not reported a clear association between mebendazole and a potential risk of major birth defects or miscarriages [see Data]. There are risks to the mother and fetus associated with untreated helminthic infection during pregnancy [see Clinical Considerations].
In animal reproduction studies, adverse developmental effects (i.e., skeletal malformations, soft tissue malformations, decreased pup weight, embryolethality) were observed when mebendazole was administered to pregnant rats during the period of organogenesis at single oral doses as low as 10 mg/kg (approximately 0.5-fold the total daily maximum recommended human dose [MRHD]). Maternal toxicity was present at the highest of these doses [see Data].
The estimated background risk of major birth defects and miscarriage for the indicated populations is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2–4% and 15–20%, respectively.
Data
Human Data
Several published studies, including prospective pregnancy registries, case-control, retrospective cohort, and randomized controlled studies, have reported no association between mebendazole use and a potential risk of major birth defects or miscarriage. Overall, these studies did not identify a specific pattern or frequency of major birth defects with mebendazole use. However, these studies cannot definitely establish the absence of any mebendazole-associated risk because of methodological limitations, including recall bias, confounding factors and, in some cases, small sample size or exclusion of first trimester mebendazole exposures.
Animal Data
Embryo-fetal developmental toxicity studies in rats revealed no adverse effects on dams or their progeny at doses up to 2.5 mg/kg/day on gestation days 6–15 (the period of organogenesis). Dosing at ≥10 mg/kg/day resulted in a lowered body weight gain and a decreased pregnancy rate. Maternal toxicity, including body weight loss in one animal and maternal death in 11 of 20 animals, was seen at 40 mg/kg/day. At 10 mg/kg/day, increased embryo-fetal resorption (100% were resorbed at 40 mg/kg/day), decreased pup weight and increased incidence of malformations (primarily skeletal) were observed. Mebendazole was also embryotoxic and teratogenic in pregnant rats at single oral doses during organogenesis as low as 10 mg/kg (approximately 0.5-fold the total daily MRHD, based on mg/m2).
In embryo-fetal developmental toxicity studies in mice dosed on gestation days 6–15, doses of 10 mg/kg/day and higher resulted in decreased body weight gain at 10 and 40 mg/kg/day and a higher mortality rate at 40 mg/kg/day. At doses of 10 mg/kg/day (approximately 0.2-fold the total daily MRHD, based on mg/m2) and higher, embryo-fetal resorption increased (100% at 40 mg/kg) and fetal malformations, including skeletal, cranial, and soft tissue anomalies, were present. Dosing of hamsters and rabbits did not result in embryotoxicity or teratogenicity at doses up to 40 mg/kg/day (1.6 to 3.9-fold the total daily MRHD, based on mg/m2).
In a peri- and post-natal toxicity study in rats, mebendazole did not adversely affect dams or their progeny at 20 mg/kg/day. At 40 mg/kg (1.9-fold the total daily MRHD, based on mg/m2), a reduction of the number of live pups was observed and there was no survival at weaning. No abnormalities were found on gross and radiographic examination of pups at birth.
8.2 Lactation
Risk Summary
Limited data from case reports demonstrate that a small amount of mebendazole is present in human milk following oral administration. There are no reports of effects on the breastfed infant, and the limited reports on the effects on milk production are inconsistent. The limited clinical data during lactation precludes a clear determination of the risk of VERMOX™ to a breastfed infant; therefore, developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for VERMOX™ and any potential adverse effects on the breastfed infant from VERMOX™ or from the underlying maternal condition.
8.4 Pediatric Use
The safety and effectiveness of VERMOX™ 100 mg chewable tablets has not been established in pediatric patients less than two years of age. Convulsions have been reported with mebendazole use in children less than one year of age [see Warnings and Precautions (5.1) and Adverse Reactions (6.2)].
10. Overdosage
In patients treated at dosages substantially higher than recommended or for prolonged periods of time, the following adverse reactions have been reported: alopecia, reversible transaminase elevations, hepatitis, agranulocytosis, neutropenia, and glomerulonephritis.
11. Vermox Description
VERMOX™ (mebendazole) is an orally administered, synthetic anthelmintic available as chewable tablets, each containing 100 mg of mebendazole. Inactive ingredients are: colloidal silicon dioxide, corn starch, hydrogenated vegetable oil, magnesium stearate, microcrystalline cellulose, sodium lauryl sulfate, sodium saccharin, sodium starch glycolate, talc, tetrarome orange, and FD&C yellow No.6.
Chemically, mebendazole is methyl 5-benzoylbenzimidazole-2-carbamate with a molecular formula of C16H13N3O3 and the following structural formula:
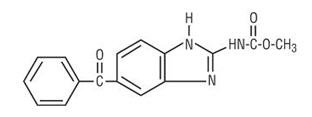
Mebendazole is a white to slightly yellow powder with a molecular weight of 295.29. It is less than 0.05% soluble in water, dilute mineral acid solutions, alcohol, ether and chloroform, but is soluble in formic acid.
12. Vermox - Clinical Pharmacology
12.1 Mechanism of Action
Mebendazole, a benzimidazole, is an anthelmintic drug [see Microbiology (12.4)].
12.3 Pharmacokinetics
Absorption
Following oral administration of mebendazole, the majority of the dose remains in the gastrointestinal tract where it exerts an anthelmintic effect locally. Following administration of 100 mg twice daily for three consecutive days, plasma concentrations of VERMOX™ (mebendazole) and its primary metabolite, the 2-amine hydrolyzed metabolite, do not exceed 0.03 mcg/mL and 0.09 mcg/mL, respectively. Dosing with a high fat meal increases the bioavailability of mebendazole, although the overall effect of food on the amount of drug remaining in the gastrointestinal tract is not expected to be substantial.
Distribution
The plasma protein binding of mebendazole is 90 to 95%. The volume of distribution is 1 to 2 L/kg, indicating that absorbed mebendazole penetrates areas outside the vascular space.
Metabolism
Orally administered mebendazole is extensively metabolized primarily by the liver. Plasma concentrations of its major metabolites (hydrolyzed and reduced forms of mebendazole) are higher than those of mebendazole. All metabolites are devoid of anthelmintic activity. Impaired hepatic function, impaired metabolism, or impaired biliary elimination may lead to higher plasma concentrations of mebendazole.
Excretion
Mebendazole, the conjugated forms of mebendazole, and its metabolites likely undergo some degree of enterohepatic recirculation. The apparent elimination half-life after an oral dose ranges from 3 to 6 hours in most patients. Less than 2% of orally administered mebendazole is excreted in urine and the remainder in the feces as unchanged drug or its metabolites.
12.4 Microbiology
Mechanism of Action
Mebendazole interferes with cellular tubulin formation in the helminth and causes ultrastructural degenerative changes in its intestine. As a result, its glucose uptake and the digestive and reproductive functions are disrupted, leading to immobilization, inhibition of egg production and death of the helminth.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In carcinogenicity tests of mebendazole in mice and rats, no carcinogenic effects were seen at doses as high as 40 mg/kg (one to two times the human dose, based on mg/m2) given daily over two years. No mutagenic activity was observed with mebendazole in a bacterial reverse gene mutation test. Mebendazole was mutagenic in the absence of S-9 when tested using a continuous (24 hour) treatment incubation period in the mouse lymphoma thymidine kinase assay. Mebendazole was aneugenic in vitro in mammalian somatic cells. In the in vivo mouse micronucleus assay, orally administered mebendazole induced an increased frequency of micronucleated polychromatic erythrocytes with evidence suggestive of aneugenicity. Doses up to 40 mg/kg in rats (2 times the total daily human dose, based on mg/m2), given to males for 60 days and to females for 14 days prior to gestation, had no effect upon fetuses and offspring.
14. Clinical Studies
Efficacy rates derived from various studies are shown in Table 4 below:
| Pinworm (enterobiasis) | Whipworm (trichuriasis) | Roundworm (ascariasis) | Hookworm | |
|---|---|---|---|---|
| Cure rates mean | 95% | 68% | 98% | 96% |
| Egg reduction mean | - | 93% | 99% | 99% |
16. How is Vermox supplied
VERMOX™ (mebendazole) is available as 100 mg, round, flat radius-edged white to yellowish chewable tablets that are debossed with "M/100" on one side and "J" on the other side. They are supplied as follows:
| Blister package of 12 tablets | NDC 50580-070-12 |
17. Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Advise patients that:
- Taking VERMOX™ and metronidazole together may cause serious skin reactions and should be avoided. [see Warning and Precautions (5.3)]
- VERMOX™ can be taken with or without food. [see Dosage and Administration (2)]
Product of Belgium
Manufactured by:
Janssen Pharmaceutica NV
Beerse, Belgium
Manufactured for:
Janssen Pharmaceuticals, Inc.
Titusville, New Jersey 08560
© 2016 Janssen Pharmaceutical Companies
| This Patient Information has been approved by the U.S. Food and Drug Administration | Revised: 06/2017 | ||
| PATIENT INFORMATION
VERMOX™ (ver-MOX) (mebendazole) chewable tablets, for oral use |
|||
| What is VERMOX?
VERMOX is a prescription medicine used to treat adults and children 2 years of age and older with intestinal worm infections caused by pinworm, whipworm, roundworm, or hookworm. |
|||
| Who should not take VERMOX?
Do not take VERMOX if you are allergic to mebendazole or any of the ingredients in VERMOX. See the end of this leaflet for a complete list of ingredients in VERMOX. |
|||
Before you take VERMOX, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Using VERMOX with certain other medicines can change the way these medicines act, causing serious side effects. Know the medicines you take. Keep a list of them to show to your healthcare provider or pharmacist when you get a new medicine. |
|||
How should I take VERMOX?
|
|||
| What should I avoid while taking VERMOX?
Do not take VERMOX with metronidazole (a medicine used to treat bacterial and protozoan infections) as serious skin reactions called Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN) can happen. |
|||
| What are the possible side effects of VERMOX?
VERMOX may cause serious side effects, including:
|
|||
|
|
||
| The most common side effects of VERMOX include: | |||
|
|
||
| Tell your healthcare provider if you have any side effect that bothers you or does not go away. These are not all the possible side effects of VERMOX. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
How should I store VERMOX?
Keep VERMOX and all medicines out of the reach of children. |
|||
| General information about the safe and effective use of VERMOX.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use VERMOX for a condition for which it was not prescribed. Do not give VERMOX to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about VERMOX that is written for health professionals. |
|||
| What are the ingredients in VERMOX?
Active ingredient: mebendazole Inactive ingredients: colloidal silicon dioxide, corn starch, hydrogenated vegetable oil, magnesium stearate, microcrystalline cellulose, sodium lauryl sulfate, sodium saccharin, sodium starch glycolate, talc, tetrarome orange, and FD&C yellow No.6. Manufactured by: Janssen Pharmaceutica NV, Beerse, Belgium Manufactured for: Janssen Pharmaceuticals, Inc., Titusville, New Jersey 08560 © 2016 Janssen Pharmaceutical Companies For more information, call 1-800-JANSSEN (1-800-526-7736). |
|||
| VERMOX
mebendazole tablet, chewable |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| Labeler - Johnson & Johnson Consumer Inc, McNeil Consumer Healthcare Division (878046358) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Janssen Pharmaceutica NV | 370005019 | API MANUFACTURE(50580-070) , MANUFACTURE(50580-070) | |
More about Vermox (mebendazole)
- Check interactions
- Compare alternatives
- Reviews (1)
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Drug class: anthelmintics
- Breastfeeding