Triple Antibiotic Ointment: Package Insert / Prescribing Info
Package insert / product label
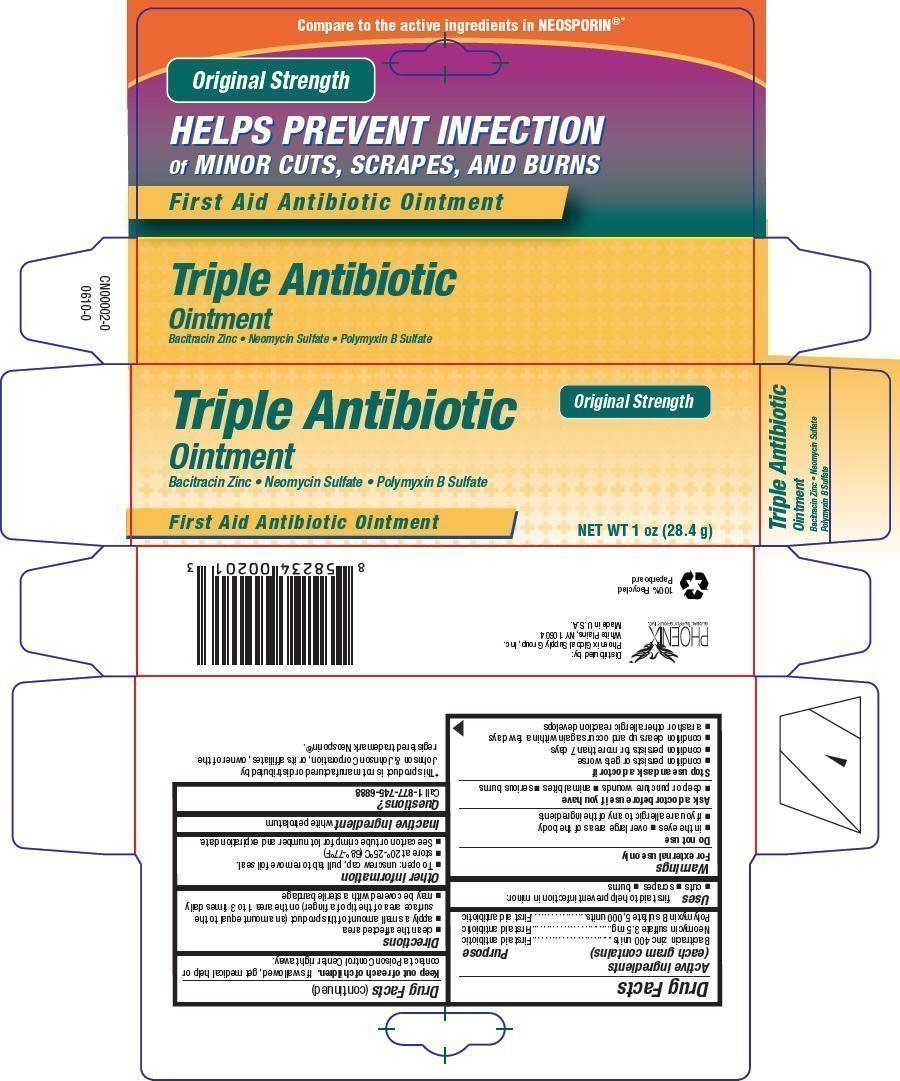
Generic name: bacitracin zinc, neomycin sulfate, and polymyxin b sulfate
Dosage form: ointment
Drug class: Topical antibiotics
Medically reviewed by Drugs.com. Last updated on Mar 25, 2025.
Active ingredients
(each gram contains) | Purpose |
| Bacitracin zinc 400 units | First aid antibiotic |
| Neomycin sulfate 3.5 mg | First aid antibiotic |
| Polymyxin B sulfate 5,000 units | First aid antibiotic |
Indications and Usage for Triple Antibiotic Ointment
first aid to help prevent infection in minor:
Warnings
For external use only
Do not use
- in the eyes
- over large areas of the body
- if you are allergic to any of the ingredients
Ask a doctor before use if you have
- deep or puncture wounds
- animal bites
- serious burns
Stop use and ask a doctor if
- condition persists or gets worse
- condition persists for more than 7 days
- condition clears up and occurs again within a few days
- a rash or other allergic reaction develops
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Triple Antibiotic Ointment Dosage and Administration
- clean the affected area
- apply a small amount of this product (an amount equal to the surface area of the tip of a finger) on the area 1 to 3 times daily
- may be covered with a sterile bandage
Storage and Handling
- To open: unscrew cap, pull tab to remove foil seal.
- store at 20°-25°C (68°-77°F)
- See carton or tube crimp for lot number and expiration date.
Related/similar drugs
Inactive ingredient
white petrolatum
Questions?
Call 1-877-745-6888
Distributed by:
Phoenix Global Supply Group, Inc.
White Plains, NY 10604
PRINCIPAL DISPLAY PANEL - 28.4 g Tube Carton
Triple Antibiotic
Ointment
Bacitracin Zinc • Neomycin Sulfate • Polymyxin B Sulfate
First Aid Antibiotic Ointment
Original Strength
NET WT 1 oz (28.4 g)
TRIPLE ANTIBIOTIC
bacitracin zinc, neomycin sulfate, and polymyxin b sulfate ointment |
|
|
|
|
|
|
|
|
|
|
|
|
More about Triple Antibiotic (bacitracin / neomycin / polymyxin b topical)
Patient resources
Related treatment guides
Medical Disclaimer