Qulipta: Package Insert / Prescribing Info
Package insert / product label
Generic name: atogepant
Dosage form: tablet
Drug class: CGRP inhibitors
Medically reviewed by Drugs.com. Last updated on Jun 23, 2025.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Drug Interactions
- Use In Specific Populations
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
QULIPTA® (atogepant) tablets, for oral use
Initial U.S. Approval: 2021
Recent Major Changes
Indications and Usage for Qulipta
QULIPTA is a calcitonin gene-related peptide receptor antagonist indicated for the preventive treatment of migraine in adults. (1)
Qulipta Dosage and Administration
- QULIPTA is taken orally with or without food. (2.1)
- For episodic migraine, the recommended dosage is 10 mg, 30 mg, or 60 mg taken once daily. (2.1)
- For chronic migraine, the recommended dosage is 60 mg taken once daily. (2.1)
- Severe Renal Impairment or End-Stage Renal Disease (2.2, 8.6):
- Episodic migraine: 10 mg once daily.
- Chronic migraine: Avoid use.
- Episodic migraine: 10 mg once daily.
Dosage Forms and Strengths
Tablets: 10 mg, 30 mg, and 60 mg. (3)
Contraindications
Patients with a history of hypersensitivity to atogepant or to any of the components of QULIPTA. (4)
Warnings and Precautions
- Hypersensitivity Reactions: If a hypersensitivity reaction occurs, discontinue QULIPTA and initiate appropriate therapy. Severe hypersensitivity reactions have included anaphylaxis and dyspnea. These reactions can occur days after administration. (5.1)
- Hypertension: New-onset or worsening of pre-existing hypertension may occur. (5.2)
- Raynaud’s phenomenon: New-onset or worsening of pre-existing Raynaud’s phenomenon may occur. (5.3)
Adverse Reactions/Side Effects
The most common adverse reactions (at least 4% and greater than placebo) are nausea, constipation, and fatigue/somnolence. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact AbbVie at 1-800-678-1605 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Recommended dosage modifications:
- Strong CYP3A4 Inhibitors (2.2, 7.1):
○ Episodic or chronic migraine: 10 mg once daily
- Strong, Moderate, or Weak CYP3A4 Inducers (2.2, 7.2):
○ Episodic migraine: 30 mg or 60 mg once daily.
○ Chronic migraine: avoid use.
- OATP Inhibitors (2.2, 7.3):
○ Episodic migraine: 10 mg or 30 mg once daily.
○ Chronic migraine: 30 mg once daily.
Use In Specific Populations
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 6/2025
Full Prescribing Information
1. Indications and Usage for Qulipta
QULIPTA is indicated for the preventive treatment of migraine in adults.
2. Qulipta Dosage and Administration
2.1 Recommended Dosage
QULIPTA is taken orally with or without food.
Episodic Migraine
The recommended dosage of QULIPTA for episodic migraine is 10 mg, 30 mg, or 60 mg taken once daily.
Chronic Migraine
The recommended dosage of QULIPTA for chronic migraine is 60 mg taken once daily.
2.2 Dosage Modifications
Dosing modifications for concomitant use of specific drugs and for patients with renal impairment are provided in Table 1.
| Dosage Modifications | Recommended Once Daily
Dosage for Episodic Migraine | Usage and
Recommended Once Daily Dosage for Chronic Migraine |
| Concomitant Drug [see Drug Interactions (7)] | ||
| Strong CYP3A4 Inhibitors (7.1) | 10 mg | 10 mg |
| Strong, Moderate, or Weak CYP3A4 Inducers (7.2) | 30 mg or 60 mg | Avoid use |
| OATP Inhibitors (7.3) | 10 mg or 30 mg | 30 mg |
| Renal Impairment [see Use in Specific Populations (8)] | ||
| Severe Renal Impairment and End-Stage Renal Disease (CLcr <30 mL/min) (8.6) | 10 mg | Avoid use |
3. Dosage Forms and Strengths
QULIPTA 10 mg is supplied as white to off-white, round biconvex tablets debossed with “A” and “10” on one side.
QULIPTA 30 mg is supplied as white to off-white, oval biconvex tablets debossed with “A30” on one side.
QULIPTA 60 mg is supplied as white to off-white, oval biconvex tablets debossed with “A60” on one side.
4. Contraindications
QULIPTA is contraindicated in patients with a history of hypersensitivity to atogepant or any of the components of QULIPTA. Reactions have included anaphylaxis and dyspnea [see Warnings and Precautions (5.1)].
5. Warnings and Precautions
5.1 Hypersensitivity Reactions
Hypersensitivity reactions, including anaphylaxis, dyspnea, rash, pruritus, urticaria, and facial edema, have been reported with use of QULIPTA [see Adverse Reactions (6.2)]. Hypersensitivity reactions can occur days after administration. If a hypersensitivity reaction occurs, discontinue QULIPTA and institute appropriate therapy [see Contraindications (4)].
5.2 Hypertension
Development of hypertension and worsening of pre-existing hypertension have been reported following the use of CGRP antagonists, including QULIPTA, in the postmarketing setting. Some of the patients who developed new-onset hypertension had risk factors for hypertension. There were cases requiring initiation of pharmacological treatment for hypertension and, in some cases, hospitalization. Hypertension may occur at any time during treatment, but was most frequently reported within 7 days of therapy initiation. QULIPTA was discontinued in many of the reported cases.
Monitor patients treated with QULIPTA for new-onset hypertension, or worsening of pre-existing hypertension, and consider whether discontinuation of QULIPTA is warranted if evaluation fails to establish an alternative etiology or blood pressure is inadequately controlled.
5.3 Raynaud’s Phenomenon
Development of Raynaud’s phenomenon and recurrence or worsening of pre-existing Raynaud’s phenomenon have been reported in the postmarketing setting following the use of CGRP antagonists, including QULIPTA. In reported cases with small molecule CGRP antagonists, symptom onset occurred a median of 1.5 days following dosing. Many of the cases reported serious outcomes, including hospitalizations and disability, generally related to debilitating pain. In most reported cases, discontinuation of the CGRP antagonist resulted in resolution of symptoms.
QULIPTA should be discontinued if signs or symptoms of Raynaud’s phenomenon develop, and patients should be evaluated by a healthcare provider if symptoms do not resolve. Patients with a history of Raynaud’s phenomenon should be monitored for, and informed about the possibility of, worsening or recurrence of signs and symptoms.
6. Adverse Reactions/Side Effects
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Hypertension [see Warnings and Precautions (5.2)]
- Raynaud’s Phenomenon [see Warnings and Precautions (5.3)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of QULIPTA was evaluated in 2657 patients with migraine who received at least one dose of QULIPTA. Of these, 1225 patients were exposed to QULIPTA for at least 6 months, and 826 patients were exposed for 12 months.
In the 12-week, placebo-controlled clinical studies (Studies 1, 2, and 3), 314 patients received at least one dose of QULIPTA 10 mg once daily, 411 patients received at least one dose of QULIPTA 30 mg once daily, 678 patients received at least one dose of QULIPTA 60 mg once daily, and 663 patients received placebo [see Clinical Studies (14)]. Approximately 88% were female, 75% were White, 13% were Black, 10% were Asian, and 10% were of Hispanic or Latino ethnicity. The mean age at study entry was 41 years (range 18 to 74 years).
The most common adverse reactions (incidence at least 4% and greater than placebo) are nausea, constipation, and fatigue/somnolence.
Table 2 summarizes the adverse reactions that occurred during Studies 1, 2, and 3.
| Placebo
(N= 663) % | QULIPTA
10 mg (N=314) % | QULIPTA
30 mg (N=411) % | QULIPTA
60 mg (N=678) % |
|
| Nausea | 3 | 5 | 6 | 9 |
| Constipation | 2 | 6 | 6 | 8 |
| Fatigue/Somnolence | 4 | 4 | 4 | 5 |
| Decreased Appetite | <1 | 2 | 1 | 3 |
| Dizziness | 2 | 2 | 2 | 3 |
* 10 mg and 30 mg incidence from Studies 1 and 2; 60 mg pooled incidence from Studies 1, 2, and 3.
The adverse reactions that most commonly led to discontinuation of QULIPTA in these studies were nausea (0.6%), constipation (0.5%), and fatigue/somnolence (0.2%).
Liver Enzyme Elevations
In Study 1, Study 2, and Study 3, the rate of transaminase elevations over 3 times the upper limit of normal was similar between patients treated with QULIPTA (0.9%) and those treated with placebo (1.2%). However, there were cases with transaminase elevations over 3 times the upper limit of normal that were temporally associated with QULIPTA treatment; these were asymptomatic and resolved within 8 weeks of discontinuation. There were no cases of severe liver injury or jaundice.
Decreases in Body Weight
In Study 1, Study 2, and Study 3, the proportion of patients with a weight decrease of at least 7% at any point was 2.5% for placebo, 3.8% for QULIPTA 10 mg, 3.2% for QULIPTA 30 mg, and 5.3% for QULIPTA 60 mg.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of QULIPTA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency or establish a causal relationship to drug exposure.
Immune System Disorders: Hypersensitivity (e.g., anaphylaxis, dyspnea, rash, pruritus, urticaria, facial edema) [see Contraindications (4) and Warnings and Precautions (5.1)]
Vascular Disorders: Hypertension [see Warnings and Precautions (5.2)], Raynaud’s phenomenon [see Warnings and Precautions (5.3)]
Related/similar drugs
7. Drug Interactions
7.1 CYP3A4 Inhibitors
Coadministration of QULIPTA with itraconazole, a strong CYP3A4 inhibitor, resulted in a significant increase in exposure of atogepant in healthy subjects [see Clinical Pharmacology (12.3)]. The recommended dosage of QULIPTA with concomitant use of strong CYP3A4 inhibitors is 10 mg once daily [see Dosage and Administration (2.2)]. No dosage adjustment of QULIPTA is needed with concomitant use of moderate or weak CYP3A4 inhibitors.
7.2 CYP3A4 Inducers
Coadministration of QULIPTA with steady state rifampin, a strong CYP3A4 inducer, resulted in a significant decrease in exposure of atogepant in healthy subjects [see Clinical Pharmacology (12.3)]. Concomitant administration of QULIPTA with moderate inducers of CYP3A4 can also result in decreased exposure of atogepant. Coadministration of QULIPTA with steady-state topiramate, a weak CYP3A4 inducer, resulted in decreased exposure of atogepant in healthy subjects [see Clinical Pharmacology (12.3)].
For episodic migraine, the recommended dosage of QULIPTA with concomitant use of strong, moderate, or weak CYP3A4 inducers is 30 mg or 60 mg once daily [see Dosage and Administration (2.2)].
For chronic migraine, avoid concomitant use of strong, moderate, or weak CYP3A4 inducers with QULIPTA [see Dosage and Administration (2.2)].
7.3 OATP Inhibitors
Coadministration of QULIPTA with single dose rifampin, an OATP inhibitor, resulted in a significant increase in exposure of atogepant in healthy subjects [see Clinical Pharmacology (12.3)]. For episodic migraine, the recommended dosage of QULIPTA with concomitant use of OATP inhibitors is 10 mg or 30 mg once daily. For chronic migraine, the recommended dosage of QULIPTA with concomitant use of OATP inhibitors is 30 mg once daily [see Dosage and Administration (2.2)].
8. Use In Specific Populations
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors outcomes in women who become pregnant while taking QULIPTA. Patients should be encouraged to enroll by calling 1-833-277-0206 or visiting http://empresspregnancyregistry.com.
Risk Summary
There are no adequate data on the developmental risk associated with the use of QULIPTA in pregnant women. In animal studies, oral administration of atogepant during the period of organogenesis (rats and rabbits) or throughout pregnancy and lactation (rats) resulted in adverse developmental effects (decreased fetal and offspring body weight in rats; increased incidence of fetal structural variations in rabbits) at exposures greater than those used clinically [see Data].
In the U.S. general population, the estimated background risk of major birth defects and miscarriages in clinically recognized pregnancies is 2-4% and 15-20%, respectively. The estimated rate of major birth defects (2.2%-2.9%) and miscarriage (17%) among deliveries to women with migraine are similar to rates reported in women without migraine.
Clinical Considerations
Disease-Associated Maternal and/or Embryo/Fetal Risk
Published data have suggested that women with migraine may be at increased risk of preeclampsia and gestational hypertension during pregnancy.
Data
Animal Data
Oral administration of atogepant (0, 5, 15, 125, or 750 mg/kg/day) to pregnant rats during the period of organogenesis resulted in decreases in fetal body weight and in skeletal ossification at the two highest doses tested (125 and 750 mg/kg), which were not associated with maternal toxicity. At the no-effect dose (15 mg/kg/day) for adverse effects on embryofetal development, plasma exposure (AUC) was approximately 4 times that in humans at the maximum recommended human dose (MRHD) of 60 mg/day.
Oral administration of atogepant (0, 30, 90, or 130 mg/kg/day) to pregnant rabbits during the period of organogenesis resulted in an increase in fetal visceral and skeletal variations at the highest dose tested (130 mg/kg/day), which was associated with minimal maternal toxicity. At the no-effect dose (90 mg/kg/day) for adverse effects on embryofetal development, plasma exposure (AUC) was approximately 3 times that in humans at the MRHD.
Oral administration of atogepant (0, 15, 45, or 125 mg/kg/day) to rats throughout gestation and lactation resulted in decreased pup body weight at the highest dose tested (125 mg/kg/day), which persisted into adulthood. At the no-effect dose (45 mg/kg/day) for adverse effects on pre- and postnatal development, plasma exposure (AUC) was approximately 5 times that in humans at the MRHD.
8.2 Lactation
Risk Summary
Data from a lactation study in twelve healthy adult females indicate that atogepant is excreted in breast milk in low amounts. The estimated relative infant dose is approximately 0.19% of the maternal weight-adjusted dose, and the milk-to-plasma ratio is 0.08 (see Data). There are no data on the effects of atogepant on the breastfed infant or the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for QULIPTA and any potential adverse effects on the breastfed infant from QULIPTA or from the underlying maternal condition.
Data
A study was conducted in twelve healthy adult lactating females who were between 23 and 34 years of age and between 1 month and 6 months postpartum. Each subject was administered a single oral dose of atogepant 60 mg. Maternal plasma and breast milk were collected for 24 hours after dosing. Using a 150 mL/kg/day estimated infant milk intake, the mean estimated relative infant dose was approximately 0.19% of the maternal weight-adjusted dose. The mean milk-to-plasma ratio was 0.08. All subjects had detectable levels of atogepant in breast milk during the study; by 16 to 24 hours after dosing, 25% of females in the study had detectable levels of atogepant in breast milk. The mean cumulative amount of atogepant excreted in breast milk over 24 hours was less than 0.01 mg of a 60 mg dose.
8.5 Geriatric Use
Population pharmacokinetic modeling suggests no clinically significant pharmacokinetic differences between elderly and younger subjects. Clinical studies of QULIPTA did not include sufficient numbers of patients aged 65 years and over to determine whether they respond differently from younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
8.6 Renal Impairment
The renal route of elimination plays a minor role in the clearance of atogepant [see Clinical Pharmacology (12.3)]. For episodic migraine, in patients with severe renal impairment (CLcr 15-29 mL/min) and in patients with end-stage renal disease (ESRD) (CLcr <15 mL/min), the recommended dosage of QULIPTA is 10 mg once daily; in patients with ESRD undergoing intermittent dialysis, QULIPTA should preferably be taken after dialysis [see Dosage and Administration (2.2)]. For chronic migraine, avoid use of QULIPTA in patients with severe renal impairment and in patients with ESRD. No dose adjustment is recommended for patients with mild or moderate renal impairment.
11. Qulipta Description
The active ingredient of QULIPTA is atogepant, a calcitonin gene-related peptide (CGRP) receptor antagonist. The chemical name of atogepant is (3’S)-N-[(3S,5S,6R)-6-methyl-2-oxo-1-(2,2,2-trifluoroethyl)-5-(2,3,6-trifluorophenyl)piperidin-3-yl]-2’-oxo-1’,2’,5,7-tetrahydrospiro[cyclopenta[b]pyridine-6,3’-pyrrolo[2,3-b]pyridine]-3-carboxamide, and it has the following structural formula:
![The active ingredient of TRADENAME is atogepant, a calcitonin gene-related peptide (CGRP) receptor antagonist. The chemical name of atogepant is (S)-N-((3S,5S,6R)-6-methyl-2-oxo-1-(2,2,2-trifluoroethyl)-5-(2,3,6-trifluorophenyl)piperidin-3-yl)-2'-oxo-1',2',5,7-tetrahydrospiro[cyclopenta[b]pyridine-6,3'-pyrrolo[2,3-b]pyridine]-3-carboxamide and has the following structural formula:](https://www.drugs.com/pro/images/8c8ab8f4-32bd-497a-befa-70c8a51d8d52/qulipta-01.jpg)
The molecular formula is C29H23F6N5O3 and molecular weight is 603.5. Atogepant is a white to off-white powder. It is freely soluble in ethanol, soluble in methanol, sparingly soluble in acetone, slightly soluble in acetonitrile, and practically insoluble in water.
QULIPTA is available as tablets for oral administration containing 10 mg, 30 mg, or 60 mg atogepant. The inactive ingredients include colloidal silicon dioxide, croscarmellose sodium, mannitol, microcrystalline cellulose, polyvinylpyrrolidone vinyl acetate copolymer, sodium chloride, sodium stearyl fumarate, and vitamin E polyethylene glycol succinate.
12. Qulipta - Clinical Pharmacology
12.2 Pharmacodynamics
Cardiac Electrophysiology
At a dose 5 times the maximum recommended daily dose, QULIPTA does not prolong the QT interval to any clinically relevant extent.
12.3 Pharmacokinetics
Absorption
Following oral administration of QULIPTA, atogepant is absorbed with peak plasma concentrations at approximately 1 to 2 hours. Atogepant displays dose-proportional pharmacokinetics up to 170 mg per day (approximately 3 times the highest recommended dosage), with no accumulation.
Effect of Food
When QULIPTA was administered with a high-fat meal, the food effect was not significant (AUC and Cmax were reduced by approximately 18% and 22%, respectively, with no effect on median time to maximum atogepant plasma concentration). QULIPTA was administered without regard to food in clinical efficacy studies.
Distribution
Plasma protein binding of atogepant was not concentration-dependent in the range of 0.1 to 10 µM; the unbound fraction of atogepant was approximately 4.7% in human plasma. The mean apparent volume of distribution of atogepant (Vz/F) after oral administration is approximately 292 L.
Elimination
Metabolism
Atogepant is eliminated mainly through metabolism, primarily by CYP3A4. The parent compound (atogepant), and a glucuronide conjugate metabolite (M23) were the most prevalent circulating components in human plasma.
Excretion
The elimination half-life of atogepant is approximately 11 hours. The mean apparent oral clearance (CL/F) of atogepant is approximately 19 L/hr. Following single oral dose of 50 mg 14C-atogepant to healthy male subjects, 42% and 5% of the dose was recovered as unchanged atogepant in feces and urine, respectively.
Specific Populations
Patients with Renal Impairment
The renal route of elimination plays a minor role in the clearance of atogepant. Based on a population pharmacokinetic analysis, there is no significant difference in the pharmacokinetics of atogepant in patients with mild or moderate renal impairment (CLcr 30-89 mL/min) relative to those with normal renal function (CLcr >90 mL/min). Patients with severe renal impairment or end-stage renal disease (ESRD; CLcr <30 mL/min) have not been studied [see Dosage and Administration (2.2) and Use in Specific Populations (8.6)].
Patients with Hepatic Impairment
In patients with pre-existing mild (Child-Pugh Class A), moderate (Child-Pugh Class B), or severe (Child-Pugh Class C) hepatic impairment, the total atogepant exposure was increased by 24%, 15%, and 38%, respectively. Due to a potential for liver injury in patients with severe hepatic impairment, avoid use of QULIPTA in patients with severe hepatic impairment [see Use in Specific Populations (8.7)].
Other Specific Populations
Based on a population pharmacokinetic analysis, age, sex, race, and body weight did not have a significant effect on the pharmacokinetics (Cmax and AUC) of atogepant. Therefore, no dose adjustments are warranted based on these factors.
Drug Interactions
In Vitro Studies
Enzymes
In vitro, atogepant is not an inhibitor for CYPs 3A4, 1A2, 2B6, 2C8, 2C9, 2C19, or 2D6 at clinically relevant concentrations. Atogepant does not inhibit MAO-A or UGT1A1 at clinically relevant concentrations. Atogepant is not anticipated to be a clinically significant perpetrator of drug-drug interactions through CYP450s, MAO-A, or UGT1A1 inhibition.
Atogepant is not an inducer of CYP1A2, CYP2B6, or CYP3A4 at clinically relevant concentrations.
Transporters
Atogepant is a substrate of P-gp, BCRP, OATP1B1, OATP1B3, and OAT1. Dose adjustment for concomitant use of QULIPTA with inhibitors of OATP is recommended based on a clinical interaction study with a OATP inhibitor [see Dosage and Administration (2.2)].
Coadministration of atogepant with BCRP and/or P-gp inhibitors is not expected to increase the exposure of atogepant. Atogepant is not a substrate of OAT3, OCT2, or MATE1.
Atogepant is not an inhibitor of P-gp, BCRP, OAT1, OAT3, NTCP, BSEP, MRP3, or MRP4 at clinically relevant concentrations. Atogepant is a weak inhibitor of OATP1B1, OATP1B3, OCT1, and MATE1. No clinical drug interactions are expected for atogepant as a perpetrator with these transporters.
In Vivo Studies
CYP3A4 Inhibitors
Co-administration of QULIPTA with itraconazole, a strong CYP3A4 inhibitor, resulted in a clinically significant increase (Cmax by 2.15-fold and AUC by 5.5-fold) in the exposure of atogepant in healthy subjects [see Drug Interactions (7.1)].
Physiologically based pharmacokinetic (PBPK) modeling suggested co-administration of QULIPTA with moderate or weak CYP3A4 inhibitors increase atogepant AUC by 1.7- and 1.1-fold, respectively. The changes in atogepant exposure when coadministered with weak or moderate CYP3A4 inhibitors are not expected to be clinically significant.
CYP3A4 Inducers
Co-administration of QULIPTA with rifampin, a strong CYP3A4 inducer, decreased atogepant AUC by 60% and Cmax by 30% in healthy subjects [see Drug Interactions (7.2)]. No dedicated drug interaction studies were conducted to assess concomitant use with moderate CYP3A4 inducers. Moderate inducers of CYP3A4 can decrease atogepant exposure [see Drug Interactions (7.2)]. Co-administration of QULIPTA with topiramate, a weak inducer of CYP3A4, decreased atogepant mean steady-state AUC 0-τ by 25% and mean steady-state Cmax by 24% in healthy subjects [see Drug Interactions (7.2)].
BCRP/OATP/P-gp Inhibitors
Co-administration of QULIPTA with single dose rifampin, an OATP inhibitor, increased atogepant AUC by 2.85-fold and Cmax by 2.23-fold in healthy subjects [see Drug Interactions (7.3)].
Co-administration of QULIPTA with quinidine, a P-gp inhibitor, increased atogepant AUC by 26% and Cmax by 4% in healthy subjects. The changes in atogepant exposure when co-administered with P-gp inhibitors are not expected to be clinically significant.
PBPK modeling suggests that co-administration of QULIPTA with BCRP inhibitors increases atogepant exposure by 1.2-fold. This increase is not expected to be clinically significant.
Other Drug Interaction Evaluations
Co-administration of QULIPTA with oral contraceptive components ethinyl estradiol and levonorgestrel, famotidine, esomeprazole, acetaminophen, naproxen, sumatriptan, or ubrogepant did not result in significant pharmacokinetic interactions for either atogepant or co-administered drugs. Co-administration of QULIPTA with topiramate did not result in clinically significant changes in the pharmacokinetics of topiramate.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity
Atogepant was administered orally to mice (0, 5, 20, or 75 mg/kg/day in males; 0, 5, 30, 160 mg/kg/day in females) and rats (0, 10, 20, or 100 mg/kg in males; 0, 25, 65, or 200 mg/kg in females) for up to 2 years. There was no evidence of drug-related tumors in either species. Plasma exposures at the highest doses tested in mice and rats were approximately 8 and 20-35 times, respectively, that in humans at the maximum recommended human dose (MRHD) of 60 mg/day.
Mutagenicity
Atogepant was negative in in vitro (Ames, chromosomal aberration test in Chinese Hamster Ovary cells) and in vivo (rat bone marrow micronucleus) assays.
Impairment of Fertility
Oral administration of atogepant (0, 5, 20, or 125 mg/kg/day) to male and female rats prior to and during mating and continuing in females to Gestation Day 7 resulted in no adverse effects on fertility or reproductive performance. Plasma exposures (AUC) at the highest dose tested are approximately 15 times that in humans at the MRHD.
14. Clinical Studies
14.1 Episodic Migraine
The efficacy of QULIPTA for the preventive treatment of episodic migraine in adults was demonstrated in two randomized, multicenter, double-blind, placebo-controlled studies (Study 1 and Study 2). The studies enrolled patients with at least a 1-year history of migraine with or without aura, according to the International Classification of Headache Disorders (ICHD-3) diagnostic criteria.
In Study 1 (NCT03777059), 910 patients were randomized 1:1:1:1 to receive QULIPTA 10 mg (N = 222), QULIPTA 30 mg (N = 230), QULIPTA 60 mg (N = 235), or placebo (N = 223), once daily for 12 weeks. In Study 2 (NCT02848326), 652 patients were randomized 1:2:2:2 to receive QULIPTA 10 mg (N = 94), QULIPTA 30 mg (N = 185), QULIPTA 60 mg (N = 187), or placebo (N = 186), once daily for 12 weeks. In both studies, patients were allowed to use acute headache treatments (i.e., triptans, ergotamine derivatives, NSAIDs, acetaminophen, and opioids) as needed. The use of a concomitant medication that acts on the CGRP pathway was not permitted for either acute or preventive treatment of migraine. The studies excluded patients with myocardial infarction, stroke, or transient ischemic attacks within six months prior to screening.
Study 1
The primary efficacy endpoint was the change from baseline in mean monthly migraine days (MMD) across the 12-week treatment period. Secondary endpoints included the change from baseline in mean monthly headache days, the change from baseline in mean monthly acute medication use days, the proportion of patients achieving at least a 50% reduction from baseline in mean MMD (3-month average), the change from baseline in mean monthly Activity Impairment in Migraine-Diary (AIM-D) Performance of Daily Activities (PDA) domain scores, the change from baseline in mean monthly AIM-D Physical Impairment (PI) domain scores, across the 12-week treatment period, and the change from baseline at Week 12 for Migraine Specific Quality of Life Questionnaire version 2.1 (MSQ v2.1) Role Function-Restrictive (RFR) domain scores.
The AIM-D evaluates difficulty with performance of daily activities (PDA domain) and physical impairment (PI domain) due to migraine, with scores ranging from 0 to 100. Higher scores indicate greater impact of migraine, and reductions from baseline indicate improvement. The MSQ v2.1 Role Function-Restrictive (RFR) domain score assesses how often migraine impacts function related to daily social and work-related activities over the past 4 weeks, with scores ranging from 0 to 100. Higher scores indicate lesser impact of migraine on daily activities, and increases from baseline indicate improvement.
Patients had a mean age of 42 years (range 18 to 73 years), 89% were female, 83% were White, 14% were Black, and 9% were of Hispanic or Latino ethnicity. The mean migraine frequency at baseline was approximately 8 migraine days per month and was similar across treatment groups. A total of 805 (88%) patients completed the 12-week double-blind study period. Key efficacy results of Study 1 are summarized in Table 3.
| QULIPTA
10 mg N=214 | QULIPTA
30 mg N=223 | QULIPTA
60 mg N=222 | Placebo
N=214 |
|
| Monthly Migraine Days (MMD) across 12 weeks | ||||
| Baseline | 7.5 | 7.9 | 7.8 | 7.5 |
| Mean change from baseline | -3.7 | -3.9 | -4.2 | -2.5 |
| Difference from placebo | -1.2 | -1.4 | -1.7 | |
| p-value | <0.001 | <0.001 | <0.001 | |
| Monthly Headache Days across 12 weeks | ||||
| Baseline | 8.4 | 8.8 | 9.0 | 8.4 |
| Mean change from baseline | -3.9 | -4.0 | -4.2 | -2.5 |
| Difference from placebo | -1.4 | -1.5 | -1.7 | |
| p-value | <0.001 | <0.001 | <0.001 | |
| Monthly Acute Medication Use Days across 12 weeks | ||||
| Baseline | 6.6 | 6.7 | 6.9 | 6.5 |
| Mean change from baseline | -3.7 | -3.7 | -3.9 | -2.4 |
| Difference from placebo | -1.3 | -1.3 | -1.5 | |
| p-value | <0.001 | <0.001 | <0.001 | |
| ≥ 50% MMD Responders across 12 weeks | ||||
| % Responders | 56 | 59 | 61 | 29 |
| Difference from placebo (%) | 27 | 30 | 32 | |
| p-value | <0.001 | <0.001 | <0.001 | |
| MSQ v2.1 RFR Domain* at week 12 | ||||
| Baseline | 44.9 | 44.0 | 46.8 | 46.8 |
| Mean change from baseline | 30.4 | 30.5 | 31.3 | 20.5 |
| Difference from placebo | 9.9 | 10.1 | 10.8 | |
| p-value | <0.001 | <0.001 | <0.001 | |
| AIM-D PDA Domain** across 12 weeks | ||||
| Baseline | 15.5 | 16.9 | 15.9 | 15.2 |
| Mean change from baseline | -7.3 | -8.6 | -9.4 | -6.1 |
| Difference from placebo | -1.2 | -2.5 | -3.3 | |
| p-value | NS† | <0.001 | <0.001 | |
| AIM-D PI Domain*** across 12 weeks | ||||
| Baseline | 11.7 | 13.0 | 11.6 | 11.2 |
| Mean change from baseline | -5.1 | -6.0 | -6.5 | -4.0 |
| Difference from placebo | -1.1 | -2.0 | -2.5 | |
| p-value | NS† | 0.002 | <0.001 | |
| * Migraine Specific Quality of Life Questionnaire version 2.1 Role Function-Restrictive domain score ** Activity Impairment in Migraine-Diary Performance of Daily Activities domain score *** Activity Impairment in Migraine-Diary Physical Impairment domain score †Not statistically significant (NS) |
||||
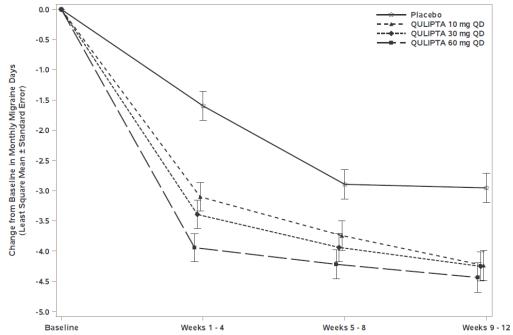
Figure 1 shows the mean change from baseline in MMD in Study 1. Patients treated with QULIPTA had greater mean decreases from baseline in MMD across the 12-week treatment period compared to patients who received placebo.
Figure 1: Change from Baseline in Monthly Migraine Days in Study 1
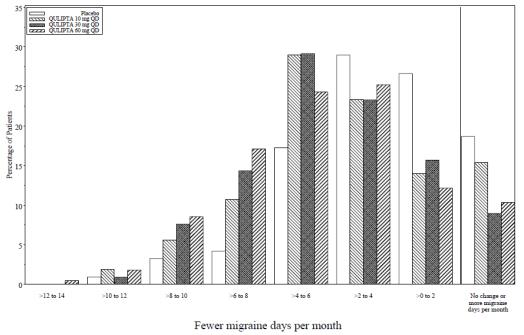
Figure 2 shows the distribution of change from baseline in mean MMD across the 12-week treatment period, in 2-day increments, by treatment group. A treatment benefit over placebo for all doses of QULIPTA is seen across a range of mean changes from baseline in MMD.
Figure 2: Distribution of Change from Baseline in Mean Monthly Migraine Days by Treatment Group in Study 1
Study 2
The primary efficacy endpoint was the change from baseline in mean monthly migraine days across the 12-week treatment period.
Patients had a mean age of 40 years (range: 18 to 74 years), 87% were female, 76% were White, 20% were Black, and 15% were of Hispanic or Latino ethnicity. The mean migraine frequency at baseline was approximately 8 migraine days per month. A total of 541 (83%) patients completed the 12-week double-blind study period.
In Study 2, there was a significantly greater reduction in mean monthly migraine days across the 12-week treatment period in all three QULIPTA treatment groups, compared with placebo, as summarized in Table 4.
| QULIPTA
10 mg N=92 | QULIPTA
30 mg N=182 | QULIPTA
60 mg N=177 | Placebo
N=178 |
|
| Monthly Migraine Days (MMD) across 12 weeks | ||||
| Baseline | 7.6 | 7.6 | 7.7 | 7.8 |
| Mean change from baseline | -4.0 | -3.8 | -3.6 | -2.8 |
| Difference from placebo | -1.1 | -0.9 | -0.7 | |
| p-value | 0.024 | 0.039 | 0.039 | |
| Monthly Headache Days across 12 weeks | ||||
| Baseline | 8.9 | 8.7 | 8.9 | 9.1 |
| Mean change from baseline | -4.3 | -4.2 | -3.9 | -2.9 |
| Difference from placebo | -1.4 | -1.2 | -0.9 | |
| p-value | 0.024 | 0.039 | 0.039 | |
Figure 3 shows the mean change from baseline in MMD in Study 2. Patients treated with QULIPTA had greater mean decreases from baseline in MMD across the 12-week treatment period compared to patients who received placebo.
Figure 3: Change from Baseline in Monthly Migraine Days in Study 2
Figure 4 shows the distribution of change from baseline in mean MMD across the 12-week treatment period, in 2-day increments, by treatment group. A treatment benefit over placebo for all doses of QULIPTA is seen across a range of mean changes from baseline in MMD.
Figure 4: Distribution of Change from Baseline in Mean Monthly Migraine Days by Treatment Group in Study 2
14.2 Chronic Migraine
Study 3
The efficacy of QULIPTA for the preventive treatment of chronic migraine in adults was demonstrated in a randomized, multicenter, double-blind, placebo-controlled study (Study 3). The study enrolled patients with at least a 1-year history of chronic migraine, according to the ICHD-3 diagnostic criteria.
Study 3 (NCT03855137) included randomization of patients to QULIPTA 60 mg once daily (N = 262) or placebo (N = 259) for 12 weeks. A subset of patients (11%) was allowed to use one concomitant migraine preventive medication. Patients were allowed to use acute headache treatments (i.e., triptans, ergotamine derivatives, NSAIDs, acetaminophen, and opioids) as needed. Patients with medication overuse headache also were enrolled. The use of a concomitant medication that acts on the CGRP pathway was not permitted for either acute or preventive treatment of migraine. The study excluded patients with myocardial infarction, stroke, or transient ischemic attacks within six months prior to screening.
The primary efficacy endpoint was the change from baseline in mean MMD across the 12-week treatment period. Secondary endpoints included the change from baseline in mean monthly headache days, the change from baseline in mean monthly acute medication use days, the proportion of patients achieving at least a 50% reduction from baseline in mean MMD (3-month average), the change from baseline in mean monthly AIM-D PDA domain scores, the change from baseline in mean monthly AIM-D PI domain scores, across the 12-week treatment period, and the change from baseline at Week 12 for MSQ v2.1 RFR domain scores.
Patients had a mean age of 42 years (range 18 to 74 years), 87% were female, 60% were White, 3% were Black, 36% were Asian, and 4% were of Hispanic or Latino ethnicity. The mean migraine frequency at baseline was approximately 19 migraine days per month and was similar across treatment groups. A total of 463 (89%) of these patients completed the 12-week double-blind study period.
Key efficacy results of Study 3 are summarized in Table 5.
| QULIPTA 60 mg QD
N=256 | Placebo
N=246 |
|
| Monthly Migraine Days (MMD) across 12 weeks | ||
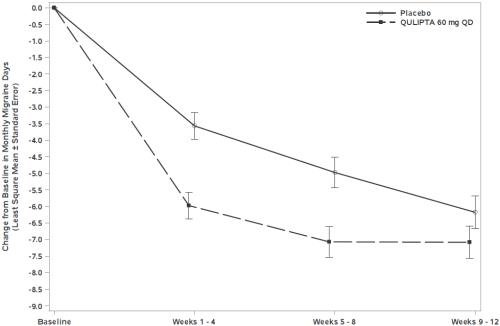
| Baseline | 19.2 | 18.9 |
| Mean change from baseline | -6.9 | -5.1 |
| Difference from placebo | -1.8 | |
| p-value | <0.001 | |
| Monthly Headache Days across 12 weeks | ||
| Baseline | 21.5 | 21.4 |
| Mean change from baseline | -7.0 | -5.1 |
| Difference from placebo | -1.9 | |
| p-value | <0.001 | |
| Monthly Acute Medication Use Days across 12 weeks | ||
| Baseline | 15.5 | 15.4 |
| Mean change from baseline | -6.2 | -4.1 |
| Difference from placebo | -2.1 | |
| p-value | <0.001 | |
| ≥ 50% MMD Responders across 12 weeks | ||
| % Responders | 41 | 26 |
| Difference from placebo (%) | 15 | |
| p-value | <0.001 | |
| MSQ v2.1 RFR Domain* at week 12 | ||
| Baseline | 43.4 | 43.9 |
| Mean change from baseline | 23.3 | 17.2 |
| Difference from placebo | 6.2 | |
| p-value | <0.001 | |
| AIM-D PDA Domain** across 12 weeks | ||
| Baseline | 31.2 | 29.5 |
| Mean change from baseline | -12.8 | -9.4 |
| Difference from placebo | -3.4 | |
| p-value | <0.001 | |
| AIM-D PI Domain*** across 12 weeks | ||
| Baseline | 27.1 | 25.2 |
| Mean change from baseline | -10.6 | -7.9 |
| Difference from placebo | -2.7 | |
| p-value | 0.003 | |
* Migraine Specific Quality of Life Questionnaire version 2.1 Role Function-Restrictive domain score
** Activity Impairment in Migraine-Diary Performance of Daily Activities domain score
*** Activity Impairment in Migraine-Diary Physical Impairment domain score
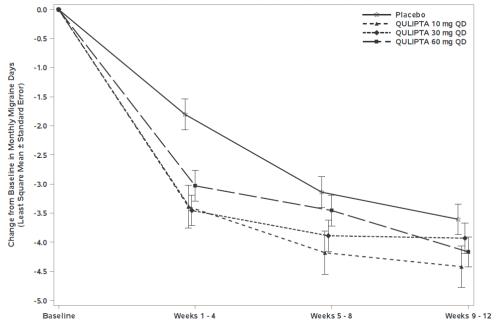
Figure 5 shows the mean change from baseline in MMD in Study 3. Patients treated with QULIPTA had greater mean decreases from baseline in MMD across the 12-week treatment period compared to patients who received placebo.
Figure 5: Change from Baseline in Monthly Migraine Days in Study 3
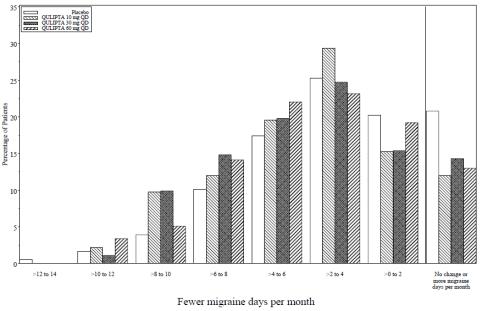
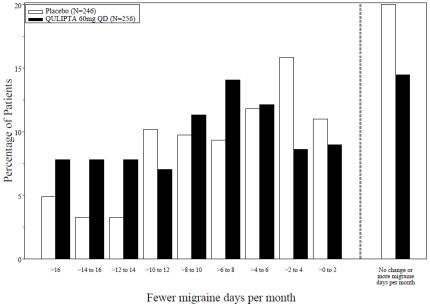
Figure 6 shows the distribution of change from baseline in mean MMD across the 12-week treatment period, in 2-day increments, by treatment group. A treatment benefit of QULIPTA over placebo is seen across a range of mean changes from baseline in MMD.
Figure 6: Distribution of Change from Baseline in Mean Monthly Migraine Days by Treatment Group in Study 3
16. How is Qulipta supplied
16.1 How Supplied
QULIPTA 10 mg is supplied as white to off-white, round biconvex tablets debossed with “A” and “10” on one side in the following packaging presentations:
- Bottle of 30, NDC: 0074-7095-30
QULIPTA 30 mg is supplied as white to off-white, oval biconvex tablets debossed with “A30” on one side in the following packaging presentations:
- Bottle of 30, NDC: 0074-7096-30
QULIPTA 60 mg is supplied as white to off-white, oval biconvex tablets debossed with “A60” on one side in the following packaging presentations:
- Bottle of 30, NDC: 0074-7094-30
17. Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Hypersensitivity Reactions
Inform patients about the signs and symptoms of hypersensitivity reactions and that these reactions can occur with QULIPTA. Advise patients to discontinue QULIPTA and seek immediate medical attention if they experience any symptoms of a hypersensitivity reaction [see Warnings and Precautions (5.1)].
Hypertension
Inform patients that hypertension can develop or pre-existing hypertension can worsen with QULIPTA, and that they should contact their healthcare providers if they experience elevation in their blood pressure [see Warnings and Precautions (5.2)].
Raynaud’s Phenomenon
Inform patients that Raynaud’s phenomenon can develop or worsen with QULIPTA. Advise patients to discontinue QULIPTA and contact their healthcare provider if they experience signs or symptoms of Raynaud’s phenomenon [see Warnings and Precautions (5.3)].
Drug Interactions
Inform patients that QULIPTA may interact with certain other drugs, and that dosage modifications of QULIPTA may be recommended when used with some other drugs. Advise patients to report to their healthcare provider the use of any other prescription medications, over-the-counter medications, herbal products, or grapefruit juice [see Dosage and Administration (2.2) and Drug Interactions (7.1, 7.2, 7.3)].
Pregnancy
Advise patients to notify their healthcare provider if they become pregnant during treatment or plan to become pregnant. Encourage pregnant patients to enroll in the registry that monitors pregnancy outcomes in women exposed to QULIPTA during pregnancy [see Use in Specific Populations (8.1)].
Lactation
Inform patients to notify their healthcare provider if they are breastfeeding or plan to breastfeed [see Use in Specific Populations (8.2)].
Manufactured for:
AbbVie Inc.
North Chicago, IL 60064
© 2025 AbbVie. All rights reserved.
QULIPTA and its design are trademarks of Allergan Pharmaceuticals International Limited, an AbbVie company.
20093629 June 2025
| PATIENT INFORMATION
QULIPTA® (kew-LIP-tah) (atogepant) tablets, for oral use |
|
What is QULIPTA?
|
|
Do not take QULIPTA if you:
|
|
Before you take QULIPTA tell your healthcare provider about all of your medical conditions, including if you:
|
|
| Keep a list of medicines you take to show to your healthcare provider or pharmacist when you get a new medicine. | |
How should I take QULIPTA?
|
|
| What are the possible side effects of QULIPTA?
QULIPTA can cause serious side effects, including:
|
|
|
• swelling of the face, lips, or tongue • trouble breathing • rash |
|
|
|
| The most common side effects of QULIPTA include: nausea, constipation, and fatigue/sleepiness. These are not all of the possible side effects of QULIPTA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store QULIPTA?
|
|
| General information about the safe and effective use of QULIPTA.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use QULIPTA for a condition for which it was not prescribed. Do not give QULIPTA to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about QULIPTA that is written for health professionals. |
|
| What are the ingredients in QULIPTA?
Active ingredient: atogepant Inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, mannitol, microcrystalline cellulose, polyvinylpyrrolidone vinyl acetate copolymer, sodium chloride, sodium stearyl fumarate, and vitamin E polyethylene glycol succinate. Manufactured for: AbbVie Inc. North Chicago, IL 60064 © 2025 AbbVie. All rights reserved. QULIPTA and its design are trademarks of Allergan Pharmaceuticals International Limited, an AbbVie company. |
|
This Patient Information has been approved by the U.S. Food and Drug Administration Revised: 6/2025
20093629
PRINCIPAL DISPLAY PANEL
NDC 0074-7095-30
Rx Only
QULIPTA®
(atogepant) tablets
10 mg
Contains 30 Tablets

| QULIPTA
atogepant tablet |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| QULIPTA
atogepant tablet |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| QULIPTA
atogepant tablet |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - AbbVie Inc. (078458370) |
Frequently asked questions
- How do Ubrelvy, Qulipta and Nurtec compare for migraines?
- What are the newest migraine medications in 2025?
- Does Qulipta cause weight loss?
- How long does Qulipta take to work?
- When is the best time of the day to take Qulipta?
- Does Qulipta (atogepant) cause hair loss?
- How well does Qulipta work for migraine?
- How does Qulipta work?
More about Qulipta (atogepant)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (190)
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Support group
- FDA approval history
- Drug class: CGRP inhibitors
- Breastfeeding
- En español