Hetlioz: Package Insert / Prescribing Info
Package insert / product label
Generic name: tasimelteon
Dosage form: capsule
Drug class: Miscellaneous anxiolytics, sedatives and hypnotics
Medically reviewed by Drugs.com. Last updated on Dec 16, 2024.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Drug Interactions
- Use In Specific Populations
- Drug Abuse and Dependence
- Overdosage
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
HETLIOZ® (tasimelteon) capsules, for oral use
HETLIOZ LQ™ (tasimelteon) oral suspension
Initial U.S. Approval: 2014
Recent Major Changes
Indications and Usage for Hetlioz
HETLIOZ is a melatonin receptor agonist.
- HETLIOZ capsules are indicated for the treatment of:
- Non-24-Hour Sleep-Wake Disorder (Non-24) in adults (1)
- Nighttime sleep disturbances in Smith-Magenis Syndrome (SMS) in patients 16 years of age and older (1)
HETLIOZ LQ oral suspension is indicated for the treatment of:
- Nighttime sleep disturbances in SMS in pediatric patients 3 years to 15 years of age (1)
Hetlioz Dosage and Administration
| Indicated
Population | Dosage
Form | Body
Weight | Recommended
Dosage |
| Non-24 (2.2) | |||
| Adults | Capsules | Not applicable | 20 mg one hour prior to bedtime |
| Nighttime sleep disturbances in SMS (2.3) | |||
| Patients 16 years of age and older | Capsules | Not applicable | 20 mg one hour prior to bedtime |
| Pediatric Patients 3 to 15 years of age | Oral Suspension | ≤ 28 kg | 0.7 mg/kg one hour before bedtime |
| ≥ 28 kg | 20 mg one hour prior to bedtime |
||
Contraindications
None (4)
Warnings and Precautions
May cause somnolence: After taking HETLIOZ, patients should limit their activity to preparing for going to bed, because HETLIOZ can impair the performance of activities requiring complete mental alertness (5.1)
Adverse Reactions/Side Effects
The most common adverse reactions (incidence >5% and at least twice as high on HETLIOZ than on placebo) were headache, increased alanine aminotransferase, nightmares or unusual dreams, and upper respiratory or urinary tract infection (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Vanda Pharmaceuticals Inc. at 1-844-438-5469 or www.hetlioz.com or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
- Strong CYP1A2 inhibitors (e.g., fluvoxamine): Avoid use of HETLIOZ in combination with strong CYP1A2 inhibitors because of increased exposure (7.1, 12.3)
- Strong CYP3A4 inducers (e.g., rifampin): Avoid use of HETLIOZ in combination with rifampin or other CYP3A4 inducers, because of decreased exposure (7.2, 12.3)
Use In Specific Populations
Hepatic impairment: HETLIOZ has not been studied in patients with severe hepatic impairment and is not recommended in these patients (8.6)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 12/2020
Full Prescribing Information
1. Indications and Usage for Hetlioz
1.1 Non-24-Hour Sleep-Wake Disorder (Non-24)
- HETLIOZ capsules are indicated for the treatment of Non-24 in adults.
1.2 Nighttime Sleep Disturbances in Smith-Magenis Syndrome (SMS)
- HETLIOZ capsules are indicated for the treatment of nighttime sleep disturbances in SMS in patients 16 years of age and older.
- HETLIOZ LQ oral suspension is indicated for the treatment of nighttime sleep disturbances in SMS in pediatric patients 3 to 15 years of age.
2. Hetlioz Dosage and Administration
2.1 Non-Interchangeability between HETLIOZ Capsules and HETLIOZ LQ Oral Suspension
HETLIOZ capsules and HETLIOZ LQ oral suspension are not substitutable [see Clinical Pharmacology (12.3)].
2.2 Recommended Dosage for HETLIOZ Capsules for Non-24
Adults
The recommended dosage of HETLIOZ capsules in adults is 20 mg one hour before bedtime, at the same time every night.
Because of individual differences in circadian rhythms, drug effect may not occur for weeks or months.
2.3 Recommended Dosage for HETLIOZ Capsules and HETLIOZ LQ Oral Suspension for Nighttime Sleep Disturbances in SMS
Patients 16 years of Age and Older
The recommended dosage of HETLIOZ capsules in patients 16 years and older is 20 mg one hour before bedtime, at the same time every night.
Pediatric Patients 3 Years to 15 Years of Age
The recommended dosage of HETLIOZ LQ oral suspension in pediatric patients 3 years to 15 years of age is based on body weight (Table 1). Administer HETLIOZ one hour before bedtime, at the same time every night.
| Body Weight | Daily Dose (oral suspension) |
| ≤28 kg | 0.7 mg/kg one hour before bedtime |
| >28 kg | 20 mg one hour before bedtime |
2.4 Important Administration Information
Administer HETLIOZ capsules and HETLIOZ LQ oral suspension without food [see Clinical Pharmacology (12.3)].
If a patient is unable to take HETLIOZ at approximately the same time on a given night, they should skip that dose and take the next dose as scheduled.
HETLIOZ LQ Oral Suspension
See "Instructions for Use" for complete administration instructions.
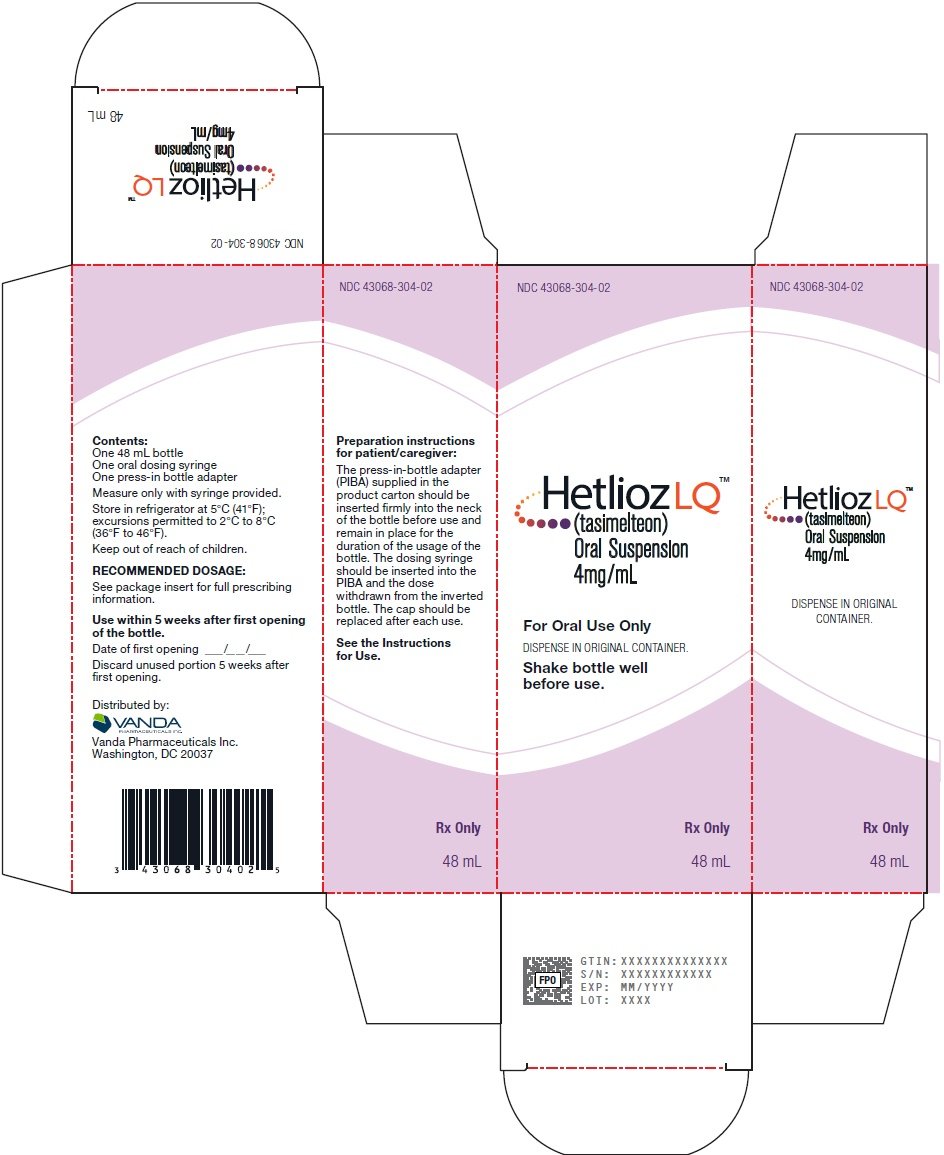
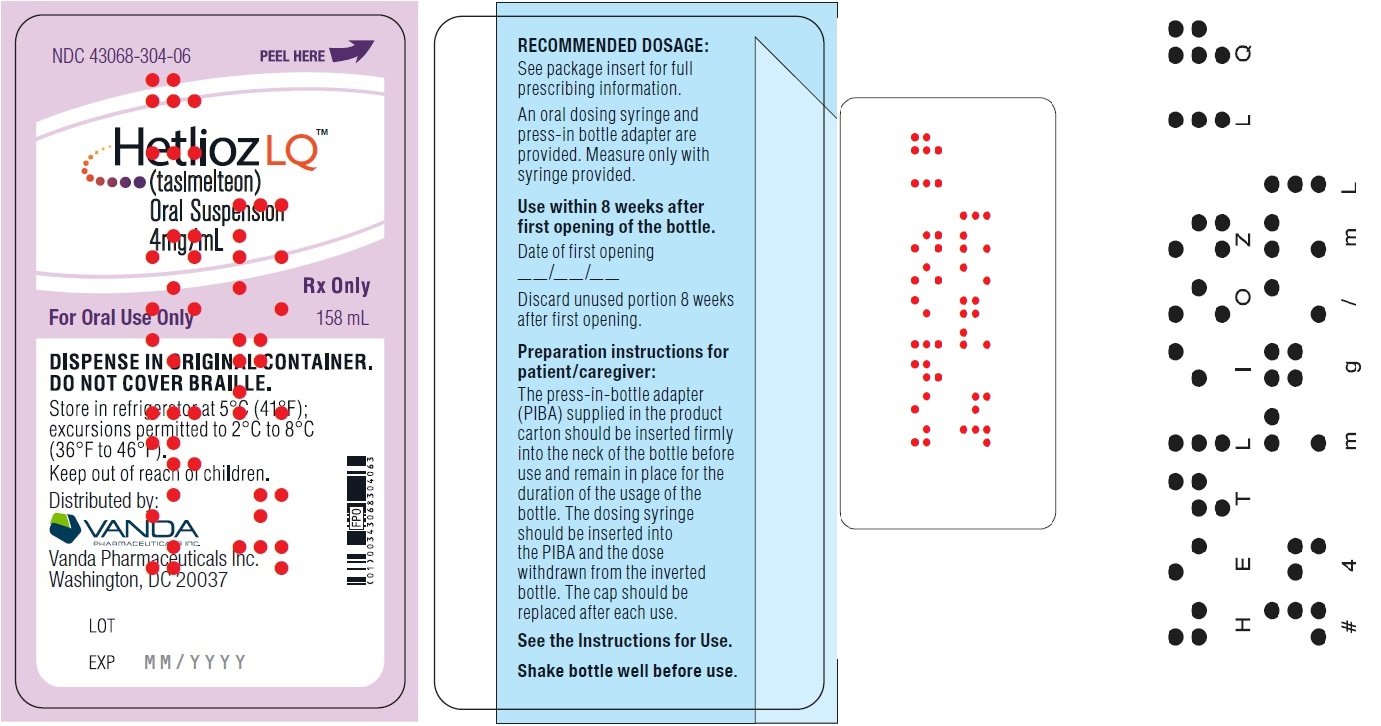
Shake HETLIOZ LQ oral suspension well for at least 30 seconds before every administration. Remove seal and insert press-in bottle adapter (included in the package) into the neck of the bottle until a tight seal is made. Turn the bottle upside down and withdraw the prescribed amount of HETLIOZ LQ oral suspension from the bottle. Leave the press-in bottle adapter in place on bottle neck and replace cap on bottle. Store refrigerated. After opening, discard after 5 weeks (for the 48 mL bottle) and after 8 weeks (for the 158 mL bottle).
3. Dosage Forms and Strengths
Capsules: 20 mg size 1 dark blue opaque, hard gelatin capsules printed with “VANDA 20 mg” in white.
Oral suspension: 4 mg/mL white to slightly yellow opaque suspension in 48 mL or 158 mL bottles.
6. Adverse Reactions/Side Effects
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
More than 2080 subjects have been treated with at least one dose of HETLIOZ, of which more than 380 have been treated for > 26 weeks and more than 170 have been treated for > 1 year.
Non-24-Hour Sleep-Wake Disorder (Non-24)
A 26-week, parallel-arm placebo-controlled study (Study 1) evaluated HETLIOZ (n=42) compared to placebo (n=42) in patients with Non-24. A randomized-withdrawal, placebo- controlled study of 8 weeks duration (Study 2) also evaluated HETLIOZ (n=10), compared to placebo (n=10), in patients with Non-24.
In placebo-controlled studies, 6% of patients exposed to HETLIOZ discontinued treatment due to an adverse event, compared with 4% of patients who received placebo.
Table 2 shows the incidence of adverse reactions from Study 1.
|
*Adverse reactions with an incidence > 5% and at least twice as high on HETLIOZ than on placebo are displayed. |
||
| Table 2: Adverse Reactions in Study 1 | ||
| HETLIOZ
N=42 | Placebo
N=42 |
|
| Headache | 17 % | 7 % |
| Alanine aminotransferase increased | 10 % | 5 % |
| Nightmare/abnormal dreams | 10 % | 0 % |
| Upper respiratory tract infection | 7 % | 0 % |
| Urinary tract infection | 7 % | 2 % |
Nighttime Sleep Disturbances in Smith-Magenis Syndrome (SMS)
A 9-week, double-blind, randomized, placebo-controlled, two-period crossover study evaluated HETLIOZ (capsules and oral suspension; n=25) compared to placebo (n=26) in the treatment of nighttime sleep disturbances in patients with Smith-Magenis Syndrome. Pediatric patients (n=11, age 3 to 15 years) received HETLIOZ LQ oral suspension, and patients ≥16 years of age (n=14) received HETLIOZ capsules.
Adverse reactions were similar in patients treated for Non-24 and patients with Smith-Magenis syndrome treated for nighttime sleep disturbances. Adverse reactions were also similar in pediatric patients (3 years to 15 years) who received HETLIOZ LQ oral suspension, and patients ≥16 years of age who received HETLIOZ capsules.
Related/similar drugs
7. Drug Interactions
7.1 Strong CYP1A2 Inhibitors (e.g., fluvoxamine)
Avoid use of HETLIOZ in combination with fluvoxamine or other strong CYP1A2 inhibitors because of a potentially large increase in tasimelteon exposure and greater risk of adverse reactions [see Clinical Pharmacology (12.3)].
7.2 Strong CYP3A4 Inducers (e.g., rifampin)
Avoid use of HETLIOZ in combination with rifampin or other CYP3A4 inducers because of a potentially large decrease in tasimelteon exposure with reduced efficacy [see Clinical Pharmacology (12.3)].
7.3 Beta-adrenergic Receptor Antagonists (e.g., acebutolol, metoprolol)
Beta-adrenergic receptor antagonists have been shown to reduce the production of melatonin via specific inhibition of beta-1 adrenergic receptors. Nighttime administration of beta-adrenergic receptor antagonists may reduce the efficacy of HETLIOZ.
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
Available postmarketing case reports with HETLIOZ use in pregnant women are not sufficient to evaluate drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. In pregnant rats, no embryofetal developmental toxicity was observed at exposures of 50 mg/kg/day, or up to 24 times higher than the human exposure at the maximum recommended human dose (MRHD) (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated populations are unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
In pregnant rats administered tasimelteon at oral doses of 5, 50, or 500 mg/kg/day during the period of organogenesis, there were no effects on embryofetal development. The highest dose tested is approximately 240 times the MRHD of 20 mg/day, based on mg/m2 body surface area.
In pregnant rabbits administered tasimelteon at oral doses of 5, 30, or 200 mg/kg/day during the period of organogenesis, embryolethality and embryofetal toxicity (reduced fetal body weight and delayed ossification) were observed at the highest dose tested. The highest dose is approximately 200 times the MRHD.
Oral administration of tasimelteon at 50, 150, or 450 mg/kg/day to rats throughout organogenesis resulted in persistent reductions in body weight, delayed sexual maturation, and physical development, and neurobehavioral impairment in offspring at the highest dose tested which is approximately 220 times the MRHD based on mg/m2 body surface area. Reduced body weight in offspring was also observed at the mid-dose. The no effect dose (NOEL), (50 mg/kg/day) is approximately 25 times the MRHD based on mg/m2 body surface area.
8.2 Lactation
Risk Summary
There are no data on the presence of tasimelteon or its metabolites in human or animal milk, the effects on the breastfed infant, or the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for HETLIOZ and any potential adverse effects on the breastfed infant from HETLIOZ or from the underlying maternal condition.
8.4 Pediatric Use
Safety and effectiveness of HETLIOZ for the treatment of Non-24 in pediatric patients have not been established.
Safety and effectiveness of HETLIOZ LQ oral suspension for the treatment of nighttime sleep disturbances in Smith-Magenis Syndrome (SMS) have been established in pediatric patients 3 years and older. Use is based on a placebo-controlled crossover study of pediatric and adult patients [see Clinical Studies (14.2)].
Safety and effectiveness of HETLIOZ for the treatment of nighttime sleep disturbances in SMS have not been established in patients younger than 3 years old.
Juvenile Animal Toxicity Data
Juvenile rats received oral doses of tasimelteon at 50, 150, or 450 mg/kg from weaning (day 21) through adulthood (day 90). These doses are approximately 12 to 108 times the maximum recommended human dose (MRHD) of 20 mg based on a mg/m2 body surface area. Toxicity was observed mainly at the highest dose and included mortality (females only), tremors, unsteady gait, decrease in growth and development compared to controls. The former reflected as decreases in bone growth, bone mineral content, bone ossification, and a delay in attainment of sexual maturation. Tasimelteon had no effect on fertility, reproduction, or learning and memory. The No Observed Adverse Effect Level (NOAEL) is 150 mg/kg/day, which is approximately 178 times the MRHD based on AUC.
8.5 Geriatric Use
The risk of adverse reactions may be greater in elderly (>65 years) patients than younger patients because exposure to tasimelteon is increased by approximately 2-fold compared with younger patients.
8.6 Hepatic Impairment
Dose adjustment is not necessary in patients with mild or moderate hepatic impairment. HETLIOZ has not been studied in patients with severe hepatic impairment (Child-Pugh Class C). Therefore, HETLIOZ is not recommended for use in patients with severe hepatic impairment [see Clinical Pharmacology (12.3)].
8.7 Smokers
Smoking causes induction of CYP1A2 levels. The exposure of tasimelteon in smokers was lower than in non-smokers and therefore the efficacy of HETLIOZ may be reduced in smokers [see Clinical pharmacology (12.3)].
9. Drug Abuse and Dependence
9.1 Controlled Substance
Tasimelteon is not a controlled substance under the Controlled Substances Act.
10. Overdosage
There is limited premarketing clinical experience with the effects of an overdosage of HETLIOZ.
As with the management of any overdose, general symptomatic and supportive measures should be used, along with immediate gastric lavage where appropriate. Intravenous fluids should be administered as needed. Respiration, pulse, blood pressure, and other appropriate vital signs should be monitored, and general supportive measures employed.
While hemodialysis was effective at clearing HETLIOZ and the majority of its major metabolites in patients with renal impairment, it is not known if hemodialysis will effectively reduce exposure in the case of overdose.
As with the management of any overdose, the possibility of multiple drug ingestion should be considered. Contact a poison control center for current information on the management of overdose.
11. Hetlioz Description
HETLIOZ (contains tasimelteon) a melatonin receptor agonist, chemically designated as (1R, 2R)-N-[2-(2,3-dihydrobenzofuran-4-yl)cyclopropylmethyl]propanamide, containing two chiral centers. The molecular formula is C15H19NO2, and the molecular weight is 245.32. The structural formula is:
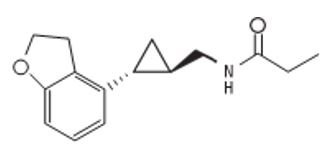
Tasimelteon is a white to off-white crystalline powder. It is very slightly soluble in cyclohexane, slightly soluble in water and 0.1 N hydrochloric acid, and freely soluble or very soluble in methanol, 95% ethanol, acetonitrile, isopropanol, polyethylene glycol 300, propylene glycol and ethyl acetate.
HETLIOZ capsules are intended for oral administration. Each capsule contains 20 mg of tasimelteon and the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, lactose anhydrous, magnesium stearate, and microcrystalline cellulose. Each hard gelatin capsule consists of FD&C Blue #1, FD&C Red #3, and FD&C Yellow #6, gelatin, and titanium dioxide.
HETLIOZ LQ oral suspension contains 4 mg of tasimelteon per mL of suspension and the following inactive ingredients: ascorbic acid, cherry flavor, mannitol, microcrystalline cellulose/carboxymethylcellulose sodium, polysorbate 80, sodium benzoate, sodium chloride, sucrose, sucralose, and water.
12. Hetlioz - Clinical Pharmacology
12.1 Mechanism of Action
The mechanism by which tasimelteon exerts its therapeutic effect in patients with Non-24 or nighttime sleep disturbances in SMS is unclear. However, tasimelteon is an agonist at melatonin MT1 and MT2 receptors which are thought to be involved in the control of circadian rhythms.
12.2 Pharmacodynamics
Tasimelteon is an agonist at MT1 and MT2 receptors with greater affinity for the MT2 as compared to the MT1 receptor (Ki = 0.304 nM and 0.07 nM, respectively). The major metabolites of tasimelteon have less than one-tenth of the binding affinity of the parent molecule for both the MT1 and MT2 receptors.
12.3 Pharmacokinetics
The pharmacokinetics of tasimelteon is linear over doses ranging from 3 to 300 mg (0.15 to 15 times the recommended daily dosage). The pharmacokinetics of tasimelteon and its metabolites did not change with repeated daily dosing.
Absorption
The absolute oral bioavailability is 38.3%. The peak concentration (Tmax) of tasimelteon capsule occurred approximately 0.5 to 3 hours after fasted oral administration.The Tmax of tasimelteon suspension occurred approximately 15 to 30 minutes after fasted oral administration.
The pharmacokinetic profile of oral suspension has not been directly compared to capsules; therefore, capsules are the only dosage form recommended for use in adults.
Effect of food
When administered with a high-fat meal, the Cmax of tasimelteon was 44% lower than when given in a fasted state, and the median Tmax was delayed by approximately 1.75 hours. Therefore, HETLIOZ should be taken without food.
Distribution
The apparent oral volume of distribution of tasimelteon at steady state in young healthy subjects is approximately 59 - 126 L. At therapeutic concentrations, tasimelteon is about 90% bound to proteins.
Metabolism
Tasimelteon is extensively metabolized. Metabolism of tasimelteon consists primarily of oxidation at multiple sites and oxidative dealkylation resulting in opening of the dihydrofuran ring followed by further oxidation to give a carboxylic acid. CYP1A2 and CYP3A4 are the major isozymes involved in the metabolism of tasimelteon.
Phenolic glucuronidation is the major phase II metabolic route.
Major metabolites had 13-fold or less activity at melatonin receptors compared to tasimelteon.
Elimination
Following oral administration of radiolabeled tasimelteon, 80% of total radioactivity was excreted in urine and approximately 4% in feces, resulting in a mean recovery of 84%. Less than 1% of the dose was excreted in urine as the parent compound.
The observed mean elimination half-life for tasimelteon is 1.3 ± 0.4 hours. The mean terminal elimination half-life ± standard deviation of the main metabolites ranges from 1.3 ± 0.5 to 3.7 ± 2.2.
Repeated once daily dosing with HETLIOZ does not result in changes in pharmacokinetic parameters or significant accumulation of tasimelteon.
Studies in Specific Populations
-
Elderly
In elderly subjects, tasimelteon exposure increased by approximately two-fold compared with non-elderly adults.
-
Pediatric Patients
Pharmacokinetic information in pediatric patients are available only for the oral suspension formulation. Body weight was found to have significant effect on the pharmacokinetics. The increase in body weight was associated with increase in tasimelteon clearance up to 28 kg. The average dose normalized Cmax and AUCinf at the recommended dose was 231 ng/mL and 310 ng.h/mL. No data are available in patients less than 3 years old.
-
Gender
The mean overall exposure of tasimelteon was approximately 20-30% greater in female than in male subjects.
-
Race
The effect of race on exposure of HETLIOZ was not evaluated.
-
Hepatic Impairment
The pharmacokinetic profile of a 20 mg dose of HETLIOZ was compared among eight subjects with mild hepatic impairment (Child-Pugh Score ≥5 and ≤6 points), eight subjects with moderate hepatic impairment (Child-Pugh Score ≥7 and ≤9 points), and 13 healthy matched controls. Tasimelteon exposure was increased less than two-fold in subjects with moderate hepatic impairment. Therefore, no dose adjustment is needed in patients with mild or moderate hepatic impairment. HETLIOZ has not been studied in patients with severe hepatic impairment (Child-Pugh Class C) and is not recommended in these patients.
-
Renal Impairment
The pharmacokinetic profile of a 20 mg dose of HETLIOZ was compared among eight subjects with severe renal impairment (estimated glomerular filtration rate [eGFR] ≤ 29 mL/min/1.73m2), eight subjects with end-stage renal disease (ESRD) (GFR < 15 mL/min/1.73m2) requiring hemodialysis, and sixteen healthy matched controls. There was no apparent relationship between tasimelteon CL/F and renal function, as measured by either estimated creatinine clearance or eGFR. Subjects with severe renal impairment had a 30% lower clearance, and clearance in subjects with ESRD was comparable to that of healthy subjects. No dose adjustment is necessary for patients with renal impairment.
-
Smokers (smoking is a moderate CYP1A2 inducer)
Tasimelteon exposure decreased by approximately 40% in smokers, compared to non- smokers [see Use in Specific Populations (8.7)].
Drug Interaction Studies
- No potential drug interactions were identified in in vitro studies with CYP inducers or inhibitors of CYP1A1, CYP1A2, CYP2B6, CYP2C9/2C19, CYP2E1, CYP2D6, and transporters including P-glycoprotein, OATP1B1, OATP1B3, OCT2, OAT1, and OAT3.
- Effect of Other Drugs on HETLIOZ
- Drugs that inhibit CYP1A2 and CYP3A4 are expected to alter the metabolism of tasimelteon.
-
Fluvoxamine (strong CYP1A2 inhibitor): the AUC0-inf and Cmax of tasimelteon increased by 7-fold and 2-fold, respectively, when co-administered with fluvoxamine 50 mg (after 6 days of fluvoxamine 50 mg per day) [see Drug Interactions (7.1)].
-
Ketoconazole (strong CYP3A4 inhibitor): tasimelteon exposure increased by approximately 50% when co-administered with ketoconazole 400 mg (after 5 days of ketoconazole 400 mg per day) [see Drug Interactions (7.2)].
- Rifampin (strong CYP3A4 and moderate CYP2C19 inducer): the exposure of tasimelteon decreased by approximately 90% when co-administered with rifampin 600 mg (after 11 days of rifampin 600 mg per day). Efficacy may be reduced when HETLIOZ is used in combination with strong CYP3A4 inducers, such as rifampin [see Drug Interactions (7.2)].
-
Fluvoxamine (strong CYP1A2 inhibitor): the AUC0-inf and Cmax of tasimelteon increased by 7-fold and 2-fold, respectively, when co-administered with fluvoxamine 50 mg (after 6 days of fluvoxamine 50 mg per day) [see Drug Interactions (7.1)].
-
Effect of HETLIOZ on Other Drugs-
Midazolam (CYP3A4 substrate): Administration of HETLIOZ 20 mg once a day for 14 days did not produce any significant changes in the Tmax, Cmax, or AUC of midazolam or 1-OH midazolam. This indicates there is no induction of CYP3A4 by tasimelteon at this dose.
- Rosiglitazone (CYP2C8 substrate): Administration of HETLIOZ 20 mg once a day for 16 days did not produce any clinically significant changes in the Tmax, Cmax, or AUC of rosiglitazone after oral administration of 4 mg. This indicates that there is no induction of CYP2C8 by tasimelteon at this dose.
-
Midazolam (CYP3A4 substrate): Administration of HETLIOZ 20 mg once a day for 14 days did not produce any significant changes in the Tmax, Cmax, or AUC of midazolam or 1-OH midazolam. This indicates there is no induction of CYP3A4 by tasimelteon at this dose.
-
Effect of Alcohol on HETLIOZ- In a study of 28 healthy volunteers, a single dose of ethanol (0.6 g/kg for women and 0.7 g/kg for men) was co-administered with a 20 mg dose of HETLIOZ. There was a trend for an additive effect of HETLIOZ and ethanol on some psychomotor tests.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Tasimelteon was administered orally for up to two years to mice (30, 100, and 300 mg/kg/day) and rats (20, 100, and 250 mg/kg/day). No evidence of carcinogenic potential was observed in mice; the highest dose tested is approximately 75 times the maximum recommended human dose (MRHD) of 20 mg/day, based on a mg/m2body surface area. In rats, the incidence of liver tumors was increased in males (adenoma and carcinoma) and females (adenoma) at 100 and 250 mg/kg/day; the incidence of tumors of the uterus (endometrial adenocarcinoma) and uterus and cervix (squamous cell carcinoma) were increased at 250 mg/kg/day. There was no increase in tumors at the lowest dose tested in rats, which is approximately 10 times the MRHD based on a mg/m2body surface area.
Mutagenesis
Tasimelteon was negative in an in vitro bacterial reverse mutation (Ames) assay, an in vitro cytogenetics assay in primary human lymphocytes, and an in vivo micronucleus assay in rats.
Impairment of Fertility
When male and female rats were given tasimelteon at oral doses of 5, 50, or 500 mg/kg/day prior to and throughout mating and continuing in females to gestation day 7, estrus cycle disruption and decreased fertility were observed at all but the lowest dose tested. The no-effect dose for effects on female reproduction (5 mg/kg/day) is approximately 2 times the MRHD based on a mg/m2 body surface area.
14. Clinical Studies
14.1 Non-24-Hour Sleep-Wake Disorder (Non-24)
The effectiveness of HETLIOZ in the treatment of Non-24-Hour Sleep-Wake Disorder (Non-24) was established in two randomized double-masked, placebo-controlled, multicenter, parallel-group studies (Studies 1 and 2) in totally blind patients with Non-24.
In study 1, 84 patients with Non-24 (median age 54 years) were randomized to receive HETLIOZ 20 mg or placebo, one hour prior to bedtime, at the same time every night for up to 6 months.
Study 2 was a randomized withdrawal trial in 20 patients with Non-24 (median age 55 years) that was designed to evaluate the maintenance of efficacy of HETLIOZ after 12-weeks. Patients were treated for approximately 12 weeks with HETLIOZ 20 mg one hour prior to bedtime, at the same time every night. Patients in whom the calculated time of peak melatonin level (melatonin acrophase) occurred at approximately the same time of day (in contrast to the expected daily delay) during the run-in phase were randomized to receive placebo or continue treatment with HETLIOZ 20 mg for 8 weeks.
Study 1 and Study 2 evaluated the duration and timing of nighttime sleep and daytime naps via patient-recorded diaries. During Study 1, patient diaries were recorded for an average of 88 days during screening, and 133 days during randomization. During Study 2, patient diaries were recorded for an average of 57 days during the run-in phase, and 59 days during the randomized-withdrawal phase.
Because symptoms of nighttime sleep disruption and daytime sleepiness are cyclical in patients with Non-24, with severity varying according to the state of alignment of the individual patient’s circadian rhythm with the 24-hour day (least severe when fully aligned, most severe when 12 hours out of alignment), efficacy endpoints for nighttime total sleep time and daytime nap duration were based on the 25% of nights with the least nighttime sleep, and the 25% of days with the most daytime nap time. In Study 1, patients in the HETLIOZ group had, at baseline, an average 195 minutes of nighttime sleep and 137 minutes of daytime nap time on the 25% of most symptomatic nights and days, respectively. Treatment with HETLIOZ resulted in a significant improvement, compared with placebo, for both of these endpoints in Study 1 and Study 2 (see Table 3).
| Table 3: Effects of HETLIOZ 20 MG on Nighttime Sleep Time and Daytime Nap Time in Study 1 and Study 2 | ||||
| Study 1 | Study 2 | |||
| Change from Baseline | HETLIOZ
20 MG N=42 | Placebo
N=42 | HETLIOZ
20 MG N=10 | Placebo
N=10 |
| Nighttime sleep time on 25% most symptomatic nights (minutes) | 50 | 22 | -7 | -74 |
| Daytime nap time on 25% most symptomatic days (minutes) | -49 | -22 | -9 | 50 |
A responder analysis of patients with both ≥ 45 minutes increase in nighttime sleep and ≥ 45 minutes decrease in daytime nap time was conducted in Study 1: 29% (n=12) of patients treated with HETLIOZ, compared with 12% (n=5) of patients treated with placebo met the responder criteria.
14.2 Nighttime Sleep Disturbances in Smith-Magenis Syndrome (SMS)
The effectiveness of HETLIOZ in the treatment of nighttime sleep disturbances in Smith-Magenis Syndrome (SMS) was established in a 9-week, double-blind, placebo- controlled cross-over study in adults and pediatric patients with SMS (Study 3; NCT 02231008). Patients 16 years of age and older received HETLIOZ 20 mg capsules, and pediatric patients 3 years to 15 years of age received a weight-based dose of oral suspension.
Study 3 had two 4-week periods, separated by a 1-week washout interval. Patients were randomized to a treatment sequence of HELTIOZ in the first period and placebo in the second period, or placebo in the first period and HETLIOZ in the second period. Patients were to take the study drug one hour prior to bedtime.
The primary endpoints in Study 3 were nighttime total sleep time and nighttime sleep quality from a parent/guardian-recorded diary. Nighttime total sleep time was reported as a time unit in hours and minutes. Nighttime sleep quality was rated as follows: 5 = excellent; 4 = good; 3 = average; 2 = fair; 1 = poor. The efficacy comparisons for nighttime sleep quality and total sleep time were based on the 50% of nights with the worst sleep quality and the 50% of nights with the least nighttime sleep in each 4-week period. In accordance with the cross-over design, the efficacy comparisons were within patient.
A total of 25 patients were randomized in Study 3. During screening, the mean quality score of the 50% of nights with the worst sleep quality was 2.1, and the total sleep time of 50% of nights with the least nighttime sleep was 6.4 hours.
Compared to placebo, treatment with HETLIOZ resulted in a statistically significant improvement in the 50% worst nights’ sleep quality. Although improvement on the 50% worst total nighttime sleep time numerically favored HETLIOZ treatment, the difference was not statistically significant (Table 4).
| SD: standard deviation; SE: standard error; LS Mean: least-squares mean; CI: confidence interval unadjusted for multiplicity.
aLS Means are the model-based averages based on the 50% worst days per 4-week period. bDifference (drug minus placebo) in least-squares means. *Endpoint on which HETLIOZ was statistically significant different from placebo after controlling for multiple comparisons. |
|||
| Primary Efficacy Measures | Treatment
Group | LS Meana (SE)
| Placebo-subtracted
Differenceb (95% CI) |
| Average of 50% Worst Daily Nighttime Sleep Quality* | HETLIOZ (n=25) | 2.8 (0.15) | 0.4 (0.1, 0.7) |
| Placebo (n=25) | 2.4 (0.15) | -- | |
| Average of 50% Worst Daily Nighttime Total Sleep Time, hours | HETLIOZ (n=25) | 7.0 (0.26) | 0.3 (-0.0, 0.6) |
| Placebo (n=25) | 6.7 (0.26) | -- | |
16. How is Hetlioz supplied
HETLIOZ Capsules
20 mg capsules are available as size 1, dark blue opaque, hard gelatin capsules printed with “VANDA 20 mg” in white, containing 20 mg of tasimelteon per capsule.
- NDC 43068-220-01 Bottles of 30
HETLIOZ LQ Oral Suspension
4 mg/mL white to slightly yellow opaque suspension. Each bottle has a child resistant cap and packaged in a carton. Each carton contains a bottle of HETLIOZ LQ oral suspension, a 5 mL oral dosing syringe and a press-in bottle adapter.
- NDC 43068-304-02 Bottles of 48 mL
- NDC 43068-304-06 Bottles of 158 mL
Storage and Handling
HETLIOZ Capsules
Store HETLIOZ capsules at controlled room temperature, 20°C to25°C (68° to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [See USP Controlled Room Temperature]. Protect from exposure to light and moisture.
HETLIOZ LQ Oral Suspension
Store HETLIOZ LQ Oral suspension at refrigerated temperature 5°C (41°F); excursions permitted to 2°C to 8°C (36°F t o 46°F).
17. Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling for HETLIOZ LQ oral suspension, if appropriate (Instructions for Use).
- Advise patients to limit their activities to preparing for going to bed after taking HETLIOZ capsules or HETLIOZ LQ oral suspension because HETLIOZ can potentially impair the performance of activities requiring complete mental alertness [see Warnings and Precautions (5.1)].
- Administration Information for HETLIOZ capsules and HETLIOZ LQ oral suspension [see Dosage and Administration (2.1, 2.2, 2.3, 2.4)].
- Advise patients to take HETLIOZ without food.
- Advise patients to take HETLIOZ before bedtime at the same time every night.
- Advise patients to skip the dose that night if they cannot take HETLIOZ at approximately the same time on a given night.
- Advise patients to swallow HETLIOZ capsules whole.
- HETLIOZ LQ oral suspension:
Advise patients to shake the bottle for at least 30 seconds before each administration; use the press-in bottle adapter included in the package; leave the adapter in place on the bottle neck; replace the cap; and refrigerate after each use. After opening, discard after 5 weeks (for the 48 mL bottle) and after 8 weeks (for the 158 mL bottle).
- Non-24 (HETLIOZ capsules)
Advise patients that because of individual differences in circadian rhythms, daily use for several weeks or months may be necessary before benefit from HETLIOZ is observed [see Dosage and Administration (2.2)].
Distributed by:
Vanda Pharmaceuticals Inc.
Washington, D.C. 20037 USA
www.hetlioz.com
Vanda, HETLIOZ, and HETLIOZ LQ are trademarks or registered trademarks of Vanda Pharmaceuticals Inc. in the United States and other countries.
|
INSTRUCTIONS FOR USE
HETLIOZ LQ™ (HeT-lee-ōz eL-Cue) |
|
Read this Instructions for Use before you give HETLIOZ LQ to your child and each time you get a refill. There may be new information. This leaflet does not take the place of talking to your healthcare provider about your medical condition or treatment.
|
|
|
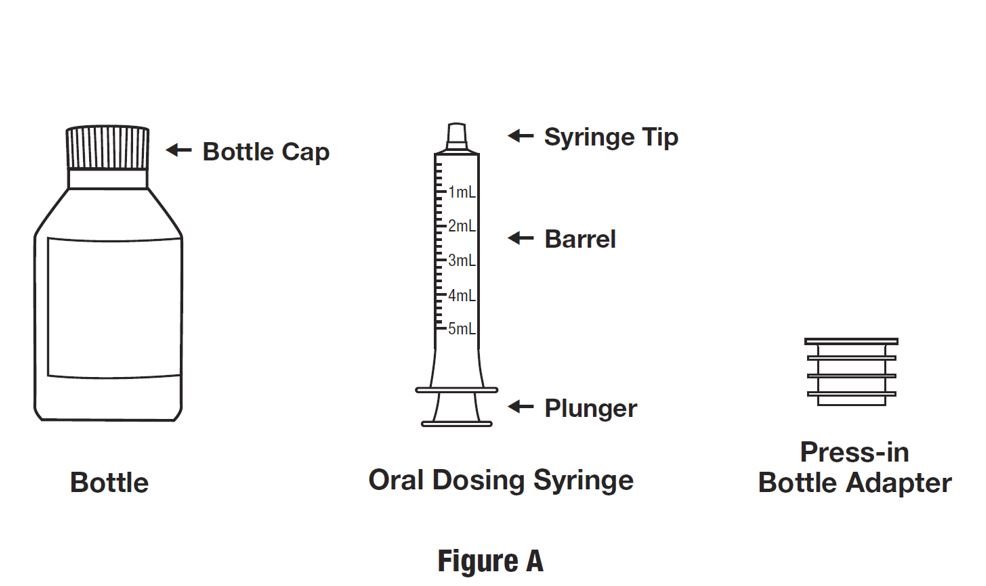
Each HETLIOZ LQ carton includes:
(see Figure A)
|
|
|
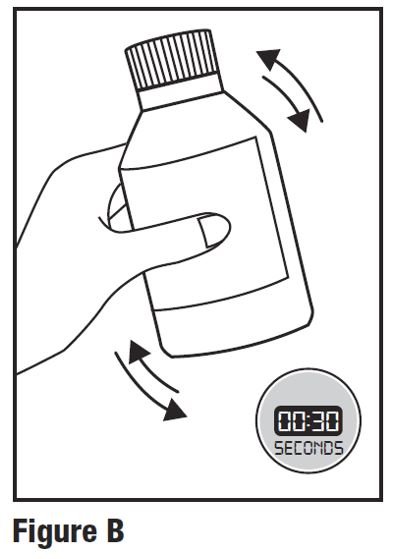
Preparing a dose of HETLIOZ LQ: Step 1. Remove the HETLIOZ LQ bottle, bottle adapter, and oral dosing syringe from the carton. Shake the bottle well for at least 30 seconds before each use (see Figure B).
|
|
|
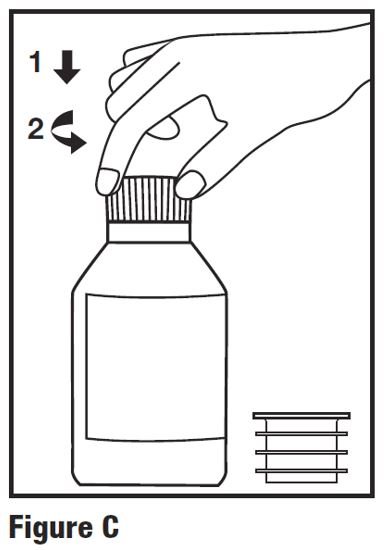
Step 2. Press down on the child-resistant cap and twist the cap counterclockwise to open bottle (see Figure C).
|
|
|
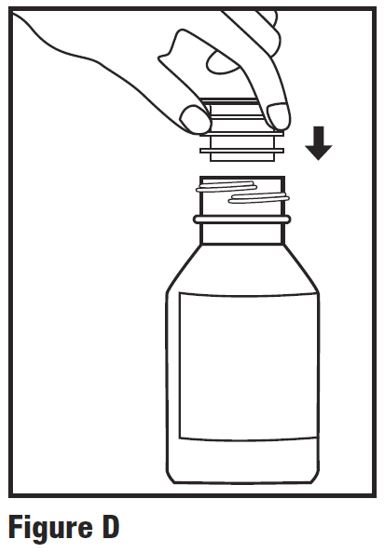
Step 3. First time use only: Remove seal from bottle and insert press-in bottle adapter into the bottle. Press on the bottle adapter until it is even with the top of the bottle (see Figure D).
|
|
|
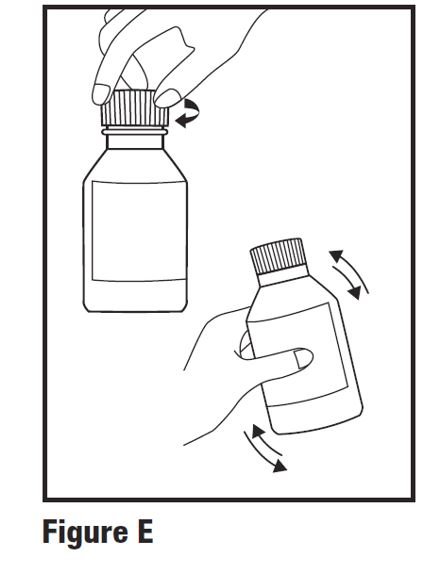
Step 4. First time use of bottle only: Replace child-resistant cap tightly by turning clockwise and shake well again for at least 30 seconds (see Figure E).
|
|
|
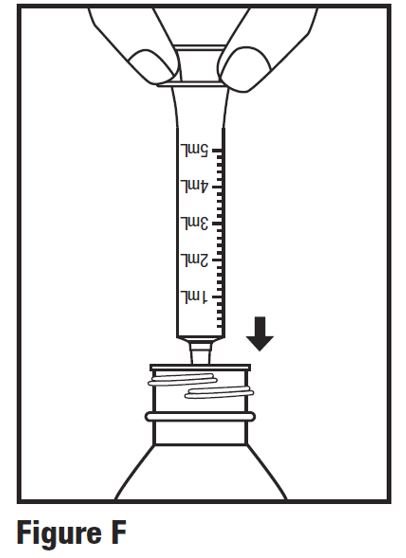
Step 5. Press down on the child-resistant cap and twist the cap counterclockwise to open bottle. Push plunger of oral dosing syringe completely down. Insert the oral dosing syringe into the opening of the press-in bottle adapter as far as it will go (see Figure F).
|
|
|
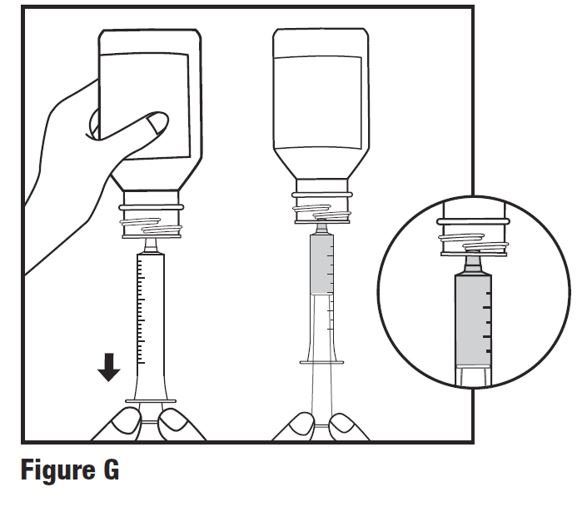
Step 6. With the oral dosing syringe in the bottle adapter, carefully turn the bottle upside down. Slowly pull back on the plunger to withdraw the prescribed amount of HETLIOZ LQ from the bottle. Line up the end of the plunger with the mL (milliliter) marking for your child’s prescribed dose of HETLIOZ LQ (see Figure G).
|
|
|
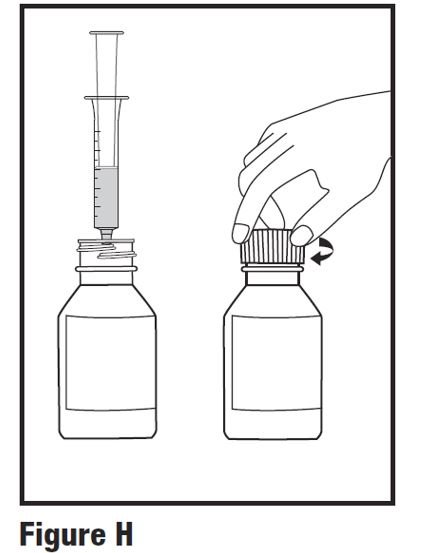
Step 7. Leave the oral dosing syringe in the bottle adapter and turn the bottle upright. Carefully remove the oral dosing syringe from the bottle adapter. Replace the child-resistant cap securely (see Figure H).
|
|
|
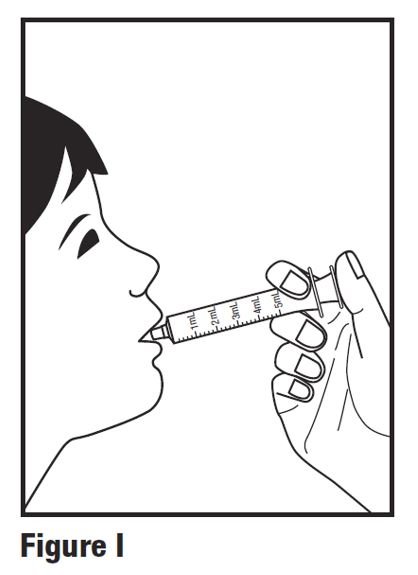
Step 8. Place the tip of the oral dosing syringe in the child’s mouth and toward the inside of their cheek. Slowly push the plunger all the way in to give the entire dose. Make sure the child has time to swallow the medicine (see Figure I).
|
|
|
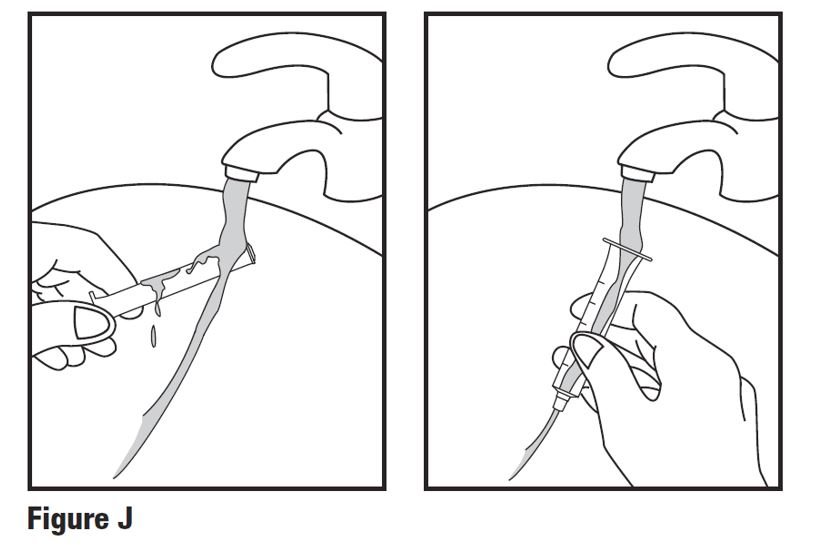
Step 9. Remove the plunger from the barrel of the oral dosing syringe. Rinse the oral dosing syringe barrel and plunger with water (see Figure J). When the barrel and plunger are dry, put the plunger back into the oral dosing syringe. Do not wash the oral dosing syringe in the dishwasher Do not throw away the oral dosing syringe. Keep this oral dosing syringe for use with this bottle of HETLIOZ LQ.
|
|
|
Storing HETLIOZ LQ:
Disposal of HETLIOZ LQ Suspension
What are the ingredients in HETLIOZ LQ oral suspension?
Inactive ingredients: ascorbic acid, cherry flavor, mannitol, microcrystalline cellulose/carboxymethylcellulose sodium, polysorbate 80, sodium benzoate, sodium chloride, sucrose, sucralose, and water. Distributed by: Vanda and HETLIOZ LQ are trademarks or registered trademarks of Vanda Pharmaceuticals Inc. in the United States and other countries. |
|
| This Instructions for Use has been approved by the U.S. Food and Drug Administration. | Issued: December 2020 |
| HETLIOZ
tasimelteon capsule |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| HETLIOZ
LQ
tasimelteon suspension |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Vanda Pharmaceuticals Inc. (133501556) |
More about Hetlioz (tasimelteon)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (2)
- Imprints, shape & color data
- Side effects
- Dosage information
- During pregnancy
- Generic availability
- FDA approval history
- Drug class: miscellaneous anxiolytics, sedatives and hypnotics
- Breastfeeding
- En español