Antacid Childrens
Dosage form: tablet, chewable
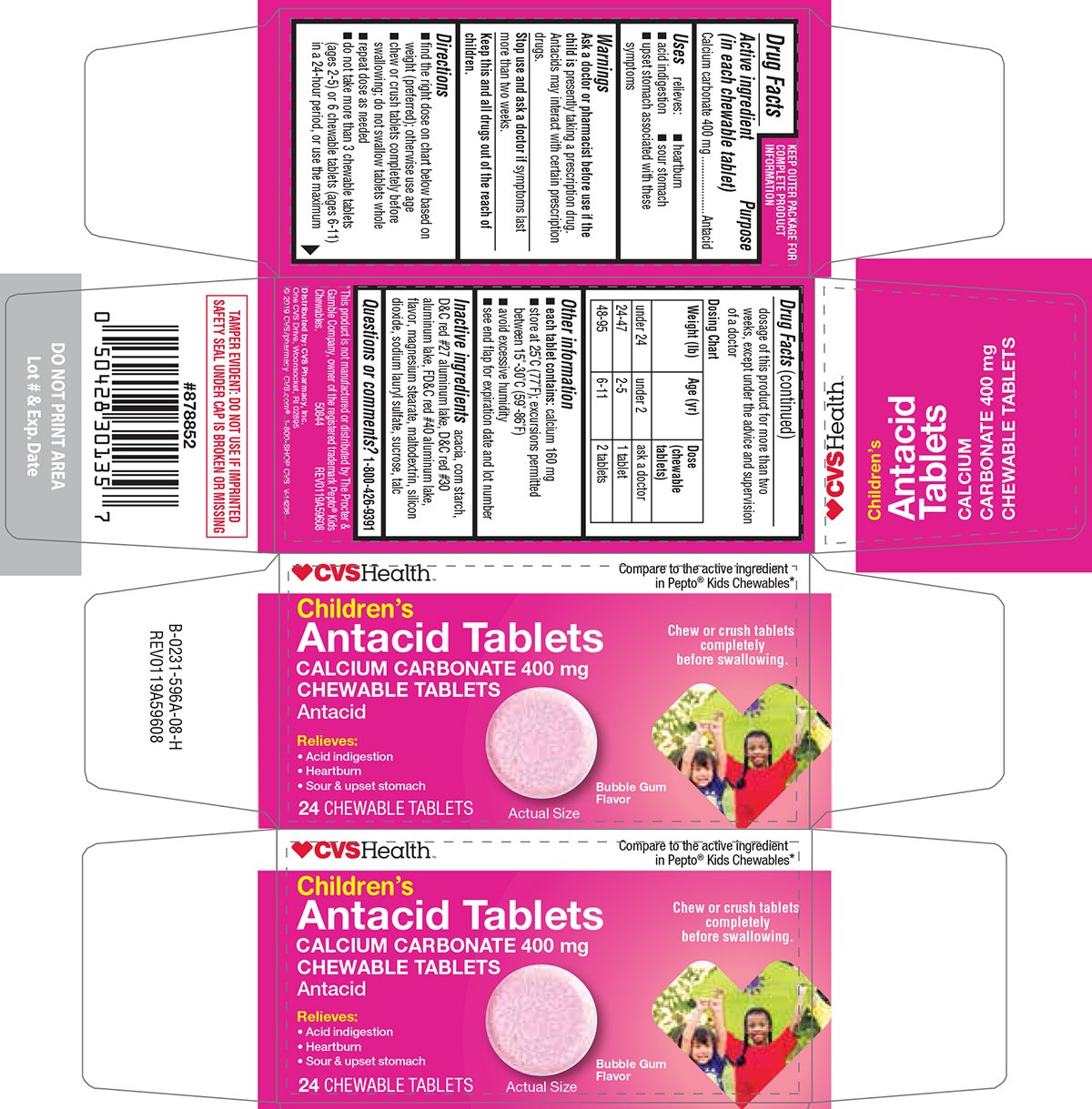
Ingredients: CALCIUM CARBONATE 400mg
Labeler: CVS Pharmacy
NDC code: 59779-596
Medically reviewed by Drugs.com. Last updated on Nov 4, 2024.
Calcium carbonate 400 mg
Antacid
relieves:
- heartburn
- acid indigestion
- sour stomach
- upset stomach associated with these symptoms
presently taking a prescription drug. Antacids may interact with certain prescription drugs.
symptoms last more than two weeks.
- find the right dose on chart below based on weight (preferred); otherwise use age
- chew or crush tablets completely before swallowing; do not swallow tablets whole
- repeat dose as needed
- do not take more than 3 chewable tablets (ages 2-5) or 6 chewable tablets (ages 6-11) in a 24-hour period, or use the maximum dosage of this product for more than two weeks, except under the advice and supervision of a doctor
| Dosing Chart
|
||
| Weight (lbs.) | Age (yr) | Dose (chewable tablets) |
| under 24 | under 2 | ask a doctor |
| 24-47 | 2-5 | 1 tablet |
| 48-95 | 6-11 | 2 tablets |
- each tablet contains: calcium 160 mg
- store at 25ºC (77ºF); excursions permitted between 15º-30ºC (59º-86ºF)
- avoid excessive humidity
- see end flap for expiration date and lot number
acacia, corn starch, D&C red #27 aluminum lake, D&C red #30 aluminum lake, FD&C red #40 aluminum lake, flavor, magnesium stearate, maltodextrin, silicon dioxide, sodium lauryl sulfate, sucrose, talc
♥CVS Health™
Compare to the active
ingredient in Pepto® Kids Chewables*
Children's
Antacid Tablets
CALCIUM CARBONATE 400 mg
CHEWABLE TABLETS
Antacid
Relieves:
• Acid indigestion
• Heartburn
• Sour & upset stomach
24 CHEWABLE TABLETS
Bubble Gum
Flavor
Actual Size
TAMPER EVIDENT: DO NOT USE IF IMPRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
*This product is not manufactured or distributed by The Procter & Gamble Company, owner of the registered trademark Pepto® Kids Chewables.
50844 REV0119A59608
Distributed by: CVS Pharmacy, Inc.
One CVS Drive, Woonsocket, RI 02895
© 2019 CVS/pharmacy
CVS.com® 1-800-SHOP CVS
V-14236
CVS® Quality
Money Back Guarantee
CVS 44-596A
| ANTACID
CHILDRENS
calcium carbonate tablet, chewable |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| Labeler - CVS Pharmacy (062312574) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| LNK International, Inc. | 038154464 | PACK(59779-596) | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| LNK International, Inc. | 832867894 | MANUFACTURE(59779-596) | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| LNK International, Inc. | 832867837 | PACK(59779-596) | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| LNK International, Inc. | 868734088 | PACK(59779-596) | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| LNK International, Inc. | 967626305 | PACK(59779-596) | |
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
