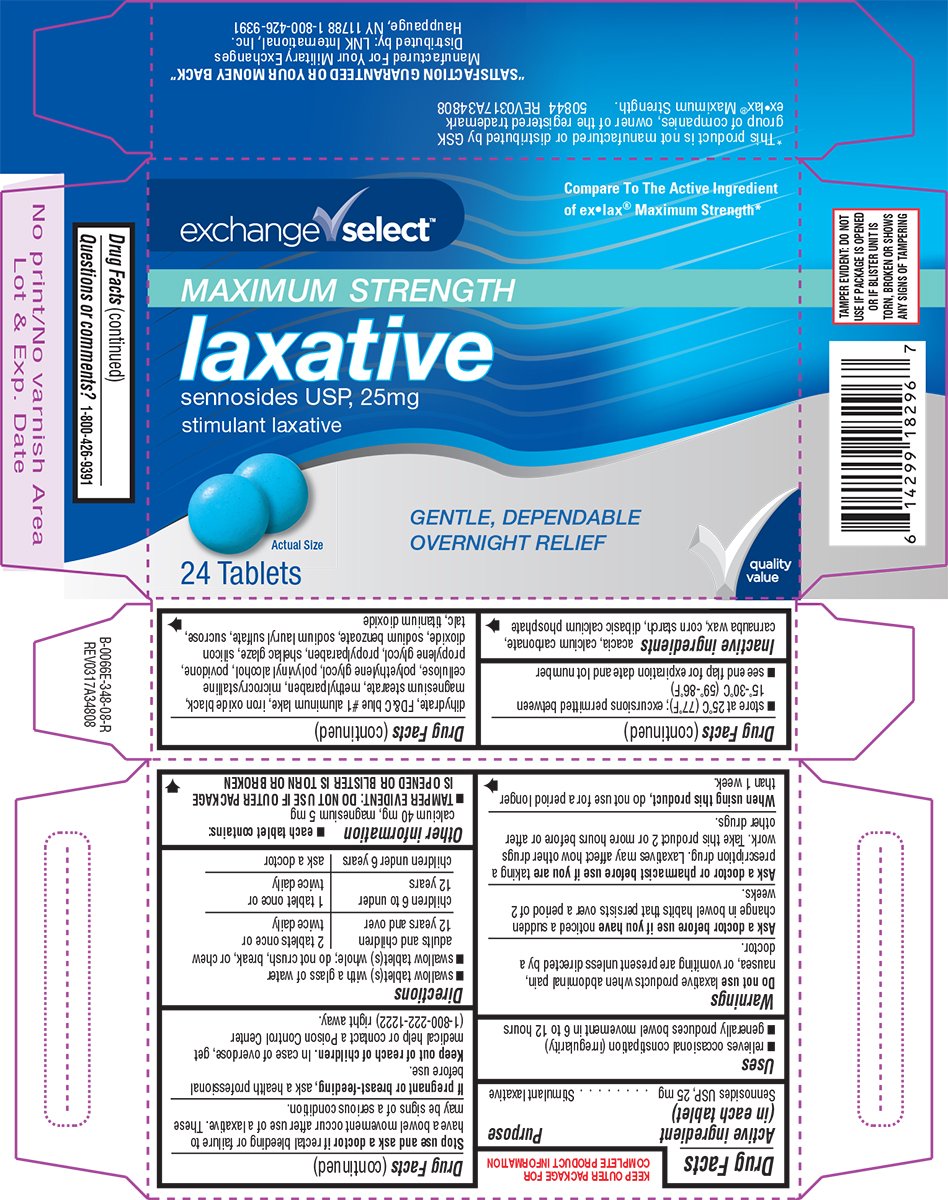
Laxative Maximum Strength
Dosage form: tablet, sugar coated
Ingredients: SENNOSIDES 25mg
Labeler: ARMY AND AIR FORCE EXCHANGE SERVICE
NDC code: 55301-348
Medically reviewed by Drugs.com. Last updated on Apr 24, 2025.
Sennosides USP, 25 mg
Stimulant laxative
- relieves occasional constipation (irregularity)
- generally produces bowel movement in 6 to 12 hours
laxative products when abdominal pain, nausea, or vomiting are present unless directed by a doctor.
noticed a sudden change in bowel habits that persists over a period of 2 weeks.
taking a prescription drug. Laxatives may affect how other drugs work. Take this product 2 or more hours before or after other drugs.
do not use for a period longer than 1 week.
rectal bleeding or failure to have a bowel movement occur after use of a laxative. These may be signs of a serious condition.
ask a health professional before use.
In case of overdose, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
- swallow tablet(s) with a glass of water
- swallow tablet(s) whole; do not crush, break, or chew
| adults and children 12 years and over | 2 tablets once or twice daily |
| children 6 to under 12 years | 1 tablet once or twice daily |
| children under 6 years | ask a doctor |
- each tablet contains: calcium 40 mg, magnesium 5 mg
- TAMPER EVIDENT: DO NOT USE IF OUTER PACKAGE IS OPENED OR BLISTER IS TORN OR BROKEN
- store at 25°C (77°F); excursions permitted between 15°-30°C (59°-86°F)
- see end flap for expiration date and lot number
acacia, calcium carbonate, carnauba wax, corn starch, dibasic calcium phosphate dihydrate, FD&C blue #1 aluminum lake, iron oxide black, magnesium stearate, methylparaben, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, propylene glycol, propylparaben, shellac glaze, silicon dioxide, sodium benzoate, sodium lauryl sulfate, sucrose, talc, titanium dioxide
1-800-426-9391
Compare To The Active Ingredient of ex•lax® Maximum Strength*
exchange ✔ select™
MAXIMUM STRENGTH
laxative
sennosides USP, 25mg
stimulant laxative
Actual Size
24 Tablets
GENTLE, DEPENDABLE
OVERNIGHT RELIEF
✔quality value
TAMPER EVIDENT: DO NOT USE IF PACKAGE IS OPENED OR IF BLISTER UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING
"SATISFACTION GUARANTEED OR YOUR MONEY BACK"
Manufactured For Your Military Exchanges
Distributed by: LNK International, Inc.
Hauppauge, NY 11788 1-800-426-9391
*This product is not manufactured or distributed by GSK group of companies, owner of the registered trademark ex•lax® Maximum Strength.
50844 REV0317A34808
Exchange Select 44-348
| LAXATIVE
MAXIMUM STRENGTH
sennosides tablet, sugar coated |
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - ARMY AND AIR FORCE EXCHANGE SERVICE (001695568) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| LNK International, Inc. | 038154464 | PACK(55301-348) | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| LNK International, Inc. | 832867894 | MANUFACTURE(55301-348) | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| LNK International, Inc. | 967626305 | PACK(55301-348) | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| LNK International, Inc. | 868734088 | PACK(55301-348) | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| LNK International, Inc. | 832867837 | PACK(55301-348) | |
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
