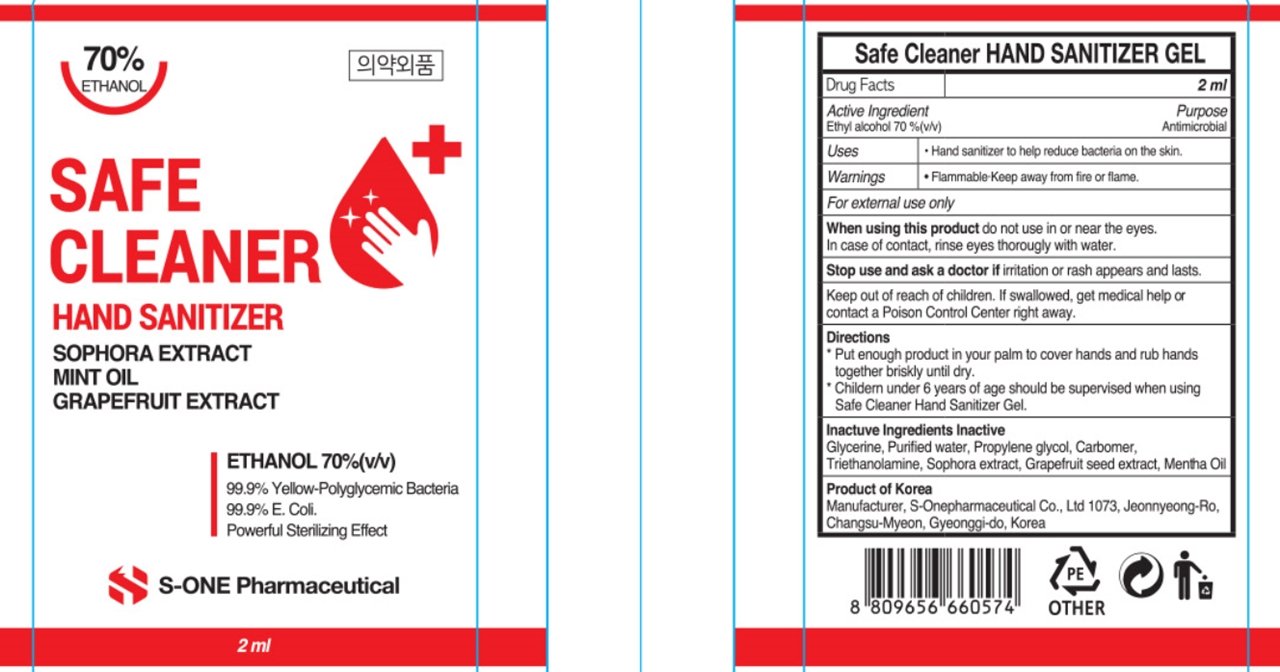
Safe Cleaner Hand Sanitizer Gel
Dosage form: gel
Ingredients: ALCOHOL 350mL in 500mL
Labeler: S ONE pharmaceutical inc.
NDC code: 74721-0001
Medically reviewed by Drugs.com. Last updated on Jun 2, 2025.
Alcohol
water, carbomer, etc
Hand sanitizer to help reduce bacteria that potentially can cause disease. For use when soap and water are not available.
keep out of reach of the children
Place enough product on hands to cover all surfaces. Rub hands together until dry.
Supervise children under 6 years of age when using this product to avoid swallowing.
For external use only. Flammable. Keep away from heat or flame.
Do not use
- in children less than 2 months of age
- on open skin wounds
When using this product keep out of eyes, ears, and mouth. In case of contact with eyes, rinse thoroughly with water.
Stop use and ask a doctor if irritation or rash occurs. These may be signs of a serious condition.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
for external use only









| SAFE CLEANER HAND SANITIZER GEL
alcohol gel |
|||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||
| Labeler - S ONE pharmaceutical inc. (694626932) |
| Registrant - S ONE pharmaceutical inc. (694626932) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| S ONE pharmaceutical inc. | 694626932 | manufacture(74721-0001), label(74721-0001), pack(74721-0001) | |
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
