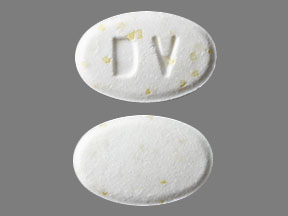
DV Pill: white, oval, 11mm
The pill with imprint DV (White, Oval, 11mm) has been identified as Doxycycline Hyclate Delayed-Release 50 mg and is used for Actinomycosis, Acne, Amebiasis, Anthrax, and Gonococcal Infection, Uncomplicated. It belongs to the drug classes miscellaneous antimalarials, tetracyclines and is not a controlled substance.
Images for DV
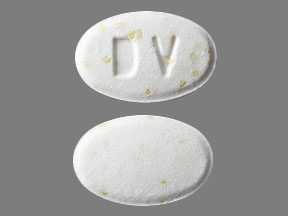
Doxycycline Hyclate Delayed-Release
- Imprint
- DV
- Strength
- 50 mg
- Color
- White
- Size
- 11.00 mm
- Shape
- Oval
- Availability
- Prescription only
- Drug Class
- Miscellaneous antimalarials, Tetracyclines
- Pregnancy Category
- D - Positive evidence of risk
- CSA Schedule
- Not a controlled drug
- Labeler / Supplier
- Mayne Pharma Inc.
- Inactive Ingredients
-
lactose monohydrate,
sodium lauryl sulfate,
sodium chloride,
magnesium silicate,
lactose anhydrous,
corn starch,
magnesium stearate,
microcrystalline cellulose,
hypromellose 2910 (6 mPa.s),
hypromellose phthalate,
triethyl citrate
Note: Inactive ingredients may vary.
Labelers / Repackagers
| NDC Code | Labeler / Repackager |
|---|---|
| 51862-0557 | Libertas Pharma |
| 51862-0709 | Libertas Pharma |
Related images for "DV"
More about doxycycline
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (1,646)
- Drug images
- Side effects
- Dosage information
- Patient tips
- During pregnancy
- Support group
- Drug class: miscellaneous antimalarials
- Breastfeeding
- En español
Patient resources
Other brands
Vibramycin, Monodox, Doryx, Doryx MPC, ... +8 more
Professional resources
- Doxycycline monograph
- Doxycycline (Topical) (AHFS Monograph)
- Doxycycline Hyclate (EENT) (AHFS Monograph)
- Doxycycline Delayed Release Capsule (FDA)
- Doxycycline Hyclate (FDA)
Other brands
Vibramycin, Monodox, Doryx, Oracea, ... +7 more
Related treatment guides
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.