Idecabtagene Vicleucel Dosage
Medically reviewed by Drugs.com. Last updated on Jun 5, 2025.
Usual Adult Dose for:
Additional dosage information:
Usual Adult Dose for Multiple Myeloma
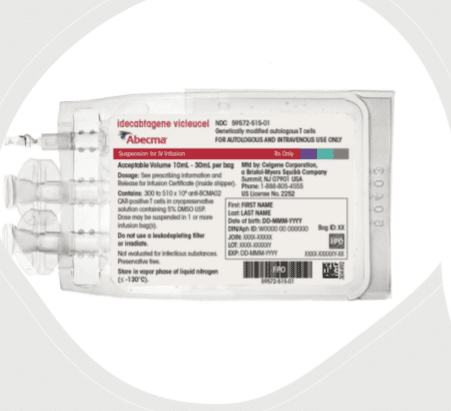
This drug is provided as a single dose for infusion containing a suspension of chimeric antigen receptor (CAR)-positive T cells in one or more infusion bags; the recommended dose is 300 to 460 x 106 CAR-positive T cells.
PRETREATMENT: Administer the lymphodepleting chemotherapy regimen: cyclophosphamide 300 mg/m2 IV and fludarabine 30 mg/m2 IV for 3 days
Administer this drug 2 days after completion of lymphodepleting chemotherapy.
Delay the infusion of this drug up to 7 days if a patient has any of the following conditions:
- Unresolved adverse events (e.g., pulmonary events, cardiac events, hypotension), including those after preceding chemotherapies
- Active infections or inflammatory disorders
Premedication:
- Administer acetaminophen (650 mg orally) and diphenhydramine (12.5 mg IV or 25 to 50 mg orally, or another H1-antihistamine) approximately 30 to 60 minutes before infusion of this drug NOTE: avoid prophylactic use of dexamethasone or other systemic corticosteroids, as the use may interfere with the activity of this drug.
Receipt of drug:
- This drug is shipped directly to the cell laboratory or clinical pharmacy associated with the infusion center in the vapor phase of a liquid nitrogen shipper.
- Confirm the patient's identity with the patient identifiers on the shipper.
- If the patient is not expected to be ready for same-day administration before the shipper expires and the infusion site is qualified for onsite storage, transfer this drug to onsite vapor phase of liquid nitrogen storage.
- If the patient is not expected to be ready for same day administration before the shipper expires and the infusion site is not qualified for onsite storage, contact Bristol-Myers Squibb to arrange for return shipment.
Preparation for Infusion:
1) Confirm the infusion time in advance and adjust the start time of the thaw of this drug so it will be available for infusion when the patient is ready.
2) Prior to thawing, confirm that tocilizumab and emergency equipment are available.
3) Doses may be contained in one or more patient-specific infusion bag(s). Verify the number of bags received for the indicated dose prior to preparation.
4) Match patient identity with identifiers on the cassette(s), infusion bag(s), and the RFI Certificate (the patient identifier number may be preceded by the letters DIN or Aph ID).
5) Do not remove the infusion bag(s) from the cassette(s) if the information on the cassette label(s) does not match the intended patient. Contact Bristol-Myers Squibb at if there are any discrepancies between the labels and the patient identifiers.
6) When patient identity is confirmed, remove the infusion bag(s) from the cassette(s) and check that the patient information on the cassette label(s) matches the patient information on the bag label(s).
7) Inspect the infusion bag(s) for breaches of container integrity such as breaks or cracks.
8) Thaw each infusion bag one at a time. Do not initiate thaw of the next bag until infusion of the previous bag is complete.
9) Place infusion bag(s) inside a second sterile bag per local guidelines.
10) Thaw infusion bag(s) at 37C using a thaw device or water bath until there is no ice in the infusion bag. Gently mix the contents to disperse clumps of cellular material. If visible clumps remain, continue to gently mix the contents. Small clumps of cellular material should disperse with gentle manual mixing. Do not wash, spin down, and/or resuspend in new media prior to infusion.
11) Administer within 1 hour of the start of thaw (drug is stable for 2 hours at room temperature when thawed).
Administration:
- For autologous use only.
- Do not use a leukodepleting filter.
- Ensure that a minimum of 2 doses of tocilizumab and emergency equipment are available.
- Central venous access is recommended in patients with poor peripheral access.
- Confirm patient's identity.
- Prime tubing with normal saline.
- Infuse contents of the bag within 1 hour after start of thaw by gravity flow.
- After infusion, rinse the tubing with 30 to 60 mL of normal saline at the same infusion rate to ensure all drug is delivered.
- Follow the same steps for any subsequent infusion bags. Do not thaw the next bag until infusion of the previous bag is complete.
Comments:
Confirm the availability of this drug prior to starting the lymphodepleting chemotherapy regimen.
Use: For the treatment of adult patients with relapsed or refractory multiple myeloma after 4 or more prior lines of therapy, including an immunomodulatory agent, a proteasome inhibitor, and an anti-CD38 monoclonal antibody
Renal Dose Adjustments
Data not available for this drug; see the prescribing information of cyclophosphamide and fludarabine for information on dose adjustment in renal impairment.
Liver Dose Adjustments
Data not available for this drug; see the prescribing information of cyclophosphamide and fludarabine for information on dose adjustment in liver impairment.
Dose Adjustments
MANAGEMENT OF SEVERE ADVERSE REACTIONS:
- Cytokine Release Syndrome (CRS): Evaluate for and treat other causes of fever, hypoxia, and hypotension. If CRS is suspected, manage according to Table 1.
- Monitor patients who experience CRS for cardiac and organ function until resolution of symptoms.
- Consider antiseizure prophylaxis with levetiracetam in patients who experience CRS.
- Patients who experience Grade 2 or higher CRS (e.g., hypotension not responsive to fluids, or hypoxia requiring supplemental oxygenation) should be monitored with continuous cardiac telemetry and pulse oximetry.
- For severe or life-threatening CRS, consider intensive care unit level monitoring and supportive therapy.
- For CRS refractory to first line interventions such as tocilizumab or tocilizumab and corticosteroids, consider alternate treatment options (i.e., higher corticosteroid dose, alternative anti-cytokine agents, anti-T cell therapies). NOTE: Refractory CRS is characterized by fevers, end-organ toxicity (e.g., hypoxia, hypotension) not improving within 12 hours of first line interventions or development of hemophagocytic lymphohistiocytosis/macrophage activation syndrome (HLH/MAS).
TABLE 1:
CRS GRADING AND MANAGEMENT GUIDANCE:
GRADE 1 (requires symptomatic treatment only [e.g., fever, fatigue, headache, myalgia, malaise]):
- Tocilizumab: If onset 72 hours or more after infusion, treat symptomatically; if onset less than 72 hours after infusion, consider tocilizumab 8 mg/kg IV over 1 hour (not to exceed 800 mg).
- Corticosteroids (if corticosteroids are initiated, continue for at least 3 doses, and taper over a maximum of 7 days): Consider dexamethasone 10 mg IV every 24 hours.
- Tocilizumab: Administer tocilizumab 8 mg/kg IV over 1 hour (not to exceed 800 mg); repeat tocilizumab every 8 hours as needed if not responsive to IV fluids or supplemental oxygen; limit to a maximum of 3 doses in a 24-hour period; maximum total of 4 doses.
- Corticosteroids (if corticosteroids are initiated, continue for at least 3 doses, and taper over a maximum of 7 days): Consider dexamethasone 10 mg IV every 12 to 24 hours.
- If no improvement of Grade 2 reaction within 24 hours or rapid progression, repeat tocilizumab and escalate dose and frequency of dexamethasone (20 mg IV every 6 to 12 hours); if no improvement within 24 hours or continued rapid progression, switch to methylprednisolone 2 mg/kg followed by 2 mg/kg divided 4 times per day; after 2 doses of tocilizumab, consider alternative anti-cytokine agents; do not exceed 3 doses of tocilizumab in 24 hours, or 4 doses total.
- Tocilizumab: Administer tocilizumab 8 mg/kg IV over 1 hour (not to exceed 800
- Corticosteroids (if corticosteroids are initiated, continue for at least 3 doses, and taper over a maximum of 7 days): Administer dexamethasone 10 mg IV every 12 hours.
- If no improvement within 24 hours or rapid progression, repeat tocilizumab and escalate dose and frequency of dexamethasone (20 mg IV every 6 to 12 hours); if no improvement within 24 hours or continued rapid progression, switch to methylprednisolone 2 mg/kg followed by 2 mg/kg divided 4 times per day; after 2 doses of tocilizumab, consider alternative anti-cytokine agents do not exceed 3 doses of tocilizumab in 24 hours, or 4 doses total.
- Tocilizumab: Administer tocilizumab 8 mg/kg IV over 1 hour (not to exceed 800
- Corticosteroids (if corticosteroids are initiated, continue for at least 3 doses, and taper over a maximum of 7 days): Administer dexamethasone 20 mg IV every 6 hours.
- After 2 doses of tocilizumab, consider alternative anti-cytokine agents; do not exceed 3 doses of tocilizumab in 24 hours, or 4 doses total if no improvement within 24 hours, consider methylprednisolone 1 to 2 g IV; repeat every 24 hours if needed with taper or other anti-T cell therapies.
NEUROLOGIC TOXICITY:
- Monitor for neurologic toxicities.
- Rule out other causes of neurologic signs or symptoms; provide supportive therapy for severe or life-threatening toxicities; if neurologic toxicity is suspected, manage according to the recommendations in Table 2.
If concurrent CRS is suspected during the neurologic toxicity, administer:
- Corticosteroids according to the more aggressive intervention based on the CRS and neurologic toxicity grades in Tables 1 and 2.
- Tocilizumab according to CRS grade in Table 1.
- Antiseizure medication according to neurologic toxicity in Table 2.
TABLE 2:
NEUROLOGIC TOXICITY GRADING AND MANAGEMENT GUIDANCE:
- Grade 1: Start non-sedating, antiseizure medicines (e.g., levetiracetam) for seizure prophylaxis; if 72 hours or more after infusion, observe patient if less than 72 hours after infusion, consider dexamethasone 10 mg IV every 12 to 24 hours for 2 to 3 days.
- Grade 2: Start non-sedating, antiseizure medicines (e.g., levetiracetam) for seizure prophylaxis; start dexamethasone 10 mg IV every 12 hours for 2 to 3 days, or longer for persistent symptoms; consider taper for a total corticosteroid exposure of greater than 3 days; corticosteroids are not recommended for isolated Grade 2 headaches if no improvement after 24 hours or worsening of neurologic toxicity, increase the dose and/or frequency of dexamethasone up to a maximum of 20 mg IV every 6 hours.
- Grade 3: Start non-sedating, antiseizure medicines (e.g., levetiracetam) for seizure prophylaxis; start dexamethasone 10 to 20 mg IV every 6 to 12 hours; corticosteroids are not recommended for isolated Grade 3 headaches; if no improvement after 24 hours or worsening of neurologic toxicity, escalate to methylprednisolone (2 mg/kg loading dose, followed by 2 mg/kg divided into 4 times a day; taper within 7 days); if cerebral edema is suspected, consider hyperventilation and hyperosmolar therapy; give high-dose methylprednisolone (1 to 2 g IV, repeat every 24 hours if needed; taper as clinically indicated) and cyclophosphamide 1.5 g/m2.
- Grade 4: Start non-sedating, antiseizure medicines (e.g., levetiracetam) for seizure prophylaxis; start dexamethasone 20 mg IV every 6 hours; if no improvement after 24 hours or worsening of neurologic toxicity, escalate to high-dose methylprednisolone (1 to 2 g IV, repeated every 24 hours if needed; taper as clinically indicated); if cerebral edema is suspected, consider hyperventilation and hyperosmolar therapy; give high-dose methylprednisolone (1 to 2 g IV, repeat every 24 hours if needed; taper as clinically indicated), and cyclophosphamide 1.5 g/m2.
Precautions
The US FDA requires a Risk Evaluation and Mitigation Strategy (REMS) for idecabtagene viceucel. It includes a medication guide, elements to assure safe use, and implementation system. For additional information: http://www.accessdata.fda.gov/scripts/cder/rems/index.cfm
Risk Evaluation and Mitigation Strategies (REMS):
US BOXED WARNINGS:
CYTOKINE RELEASE SYNDROME (CRS):
- CCRs, including fatal or life-threatening reactions, occurred in patients following treatment with this drug.
- Do not administer to patients with active infection or inflammatory disorders.
- Treat severe or life-threatening CRS with tocilizumab or tocilizumab and corticosteroids.
- Neurologic toxicities, which may be severe or life-threatening, has been reported, including concurrently with CRS, after CRS resolution, or in the absence of CRS.
- Monitor for neurologic events.
- Provide supportive care and/or corticosteroids as needed.
- HLH/MAS, including fatal and life-threatening reactions, has been reported.
- HLH/MAS can occur with CRS or neurologic toxicities.
- Prolonged cytopenia with bleeding and infection, including fatal outcomes following stem cell transplantation for hematopoietic recovery, has been reported.
This drug is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the ABECMA REMS:
The required components of the ABECMA REMS are:
- Healthcare facilities that dispense and administer ABECMA must be enrolled and comply with the REMS requirements.
- Certified healthcare facilities must have on-site, immediate access to tocilizumab.
- Ensure that a minimum of 2 doses of tocilizumab are available for each patient for infusion within 2 hours after ABECMA infusion, if needed for treatment of CRS.
- Certified healthcare facilities must ensure that healthcare providers who prescribe, dispense, or administer ABECMA are trained in the management of CRS and neurologic toxicities.
- Further information is available at www.AbecmaREMS.com or contact Bristol-Myers Squibb at 1-888-423-5436.
CONTRAINDICATIONS:
- None
Safety and efficacy have not been established in patients younger than 18 years.
Consult WARNINGS section for additional precautions.
Dialysis
Data not available
Other Comments
Administration advice:
- For autologous use only.
- For IV use only.
- Verify that the patient's identity matches the identifiers on the cassette and infusion bag; do not infuse this drug if the information on the patient-specific label does not match the intended patient.
Storage requirements:
- This drug is supplied in one or more infusion bag(s) containing a frozen suspension of genetically modified autologous T cells in 5% DMSO.
- Each infusion bag is individually packed in a metal cassette and stored in the vapor phase of liquid nitrogen and supplied in a liquid nitrogen dry vapor shipper. An RFI Certificate is affixed inside the shipper.
- Store frozen in the vapor phase of liquid nitrogen (less than or equal to minus 130C).
Reconstitution/preparation techniques:
- Thaw prior to infusion.
- The manufacturer product information should be consulted.
General:
- This drug contains human blood cells that are genetically modified with replication-incompetent, self-inactivating lentiviral vector. Follow biosafety guidelines for handling and disposal to avoid transmission of infectious diseases.
Monitoring:
- Administer this drug at a REMS-certified healthcare facility.
- Monitor patients at least daily for 7 days following infusion for CRS and neurologic toxicities.
- Instruct patients to remain within proximity of the healthcare facility for at least 4 weeks following infusion.
- Instruct patients to refrain from driving or hazardous activities for at least 8 weeks following infusion.
Frequently asked questions
More about idecabtagene vicleucel
- Check interactions
- Compare alternatives
- Latest FDA alerts (3)
- Side effects
- During pregnancy
- Drug class: miscellaneous antineoplastics
- En español
Patient resources
Other brands
Professional resources
Other brands
Related treatment guides
See also:
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.