Draxxin 25 Injectable Solution (Canada)
This treatment applies to the following species: Company: Zoetis
Company: Zoetis
tulathromycin sterile injectable solution
Veterinary Use Only
Antibacterial
for swine
DIN 02430711
DESCRIPTION: Draxxin® 25 Injectable Solution is a ready-to-use sterile parenteral preparation containing tulathromycin, a semi-synthetic macrolide antibiotic of the subclass triamilide. Each mL of Draxxin 25 Injectable Solution contains 25 mg of tulathromycin as the free base in a propylene glycol vehicle.
Draxxin 25 Injectable Solution consists of an equilibrated mixture of two isomeric forms of tulathromycin in a 9:1 ratio. Structures of the isomers are shown below:
Figure 1.
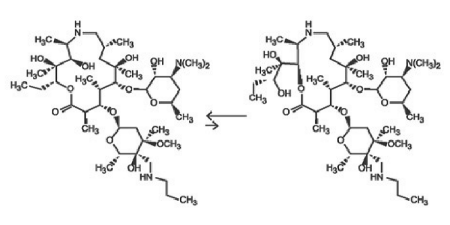
The chemical names of the isomers are (2R,3S,4R,5R,8R,10R,11R,12S,13S,14R)-13-[[2,6-dideoxy-3-C-methyl-3-O-methyl-4-C-[(propylamino) methyl] - α - L - ribo - hexopyranosyl]oxy] - 2 - ethyl - 3,4,10 - trihydroxy - 3,5,8,10,12,14 - hexamethyl - 11 - [[3,4,6 - trideoxy - 3 - (dimethylamino) - β - D - xylo - hexopyranosyl] - oxy] - 1 - oxa - 6 - azacyclopentadecan - 15 - one and (2R,3R,6R,8R,9R,10S,11S,12R) - 11 - [[2,6 - dideoxy - 3 - C - methyl - 3 - O - methyl - 4 - C - [(propylamino)methyl] - α - L - ribo - hexopyranosyl]oxy] - 2 - [(1S,2R) - 1,2 - dihydroxy - 1 - methylbutyl] - 8 - hydroxy - 3,6,8,10,12 - pentamethyl - 9 - [[3,4,6 - trideoxy - 3 - (dimethylamino) - β - D - xylohexopyranosyl]oxy] - 1 - oxa - 4 - azacyclotridecan - 13 - one, respectively.
Draxxin 25 Injectable Solution Indications
Draxxin 25 Injectable Solution is indicated for the treatment of swine respiratory disease (SRD) associated with Actinobacillus pleuropneumoniae, Pasteurella multocida and Mycoplasma hyopneumoniae and for the control of SRD caused by Actinobacillus pleuropneumoniae, Pasteurella multocida and Mycoplasma hyopneumoniae in groups of pigs where SRD has been diagnosed.
Dosage and Administration
Inject intramuscularly in the neck, a single dose of 2.5 mg/kg body weight (1.0 mL/10 kg). Do not inject more than 4 mL per injection site. Most animals will respond to treatment within 3 to 5 days. If no improvement is observed, the diagnosis should be re-evaluated.
Note: To limit the development of antimicrobial resistance, Draxxin 25 Injectable Solution should only be used for control of SRD when segregation and treatment of individual sick animals is unlikely to control the disease outbreak. Veterinarians should make treatment decision by considering among others overall farm management and outbreak associated factors.
Table 1. Draxxin 25 Injectable Solution Swine Dosing Guide
|
Animal Weight (kg BW) |
Dose Volume (mL) |
|
6 |
0.6 |
|
8 |
0.8 |
|
12 |
1.2 |
|
16 |
1.6 |
|
24 |
2.4 |
|
32 |
3.2 |
|
40 |
4.0 |
CONTRAINDICATION: Draxxin 25 Injectable Solution is contraindicated in animals previously found to be hypersensitive to macrolide antibiotics.
CAUTIONS: The effects of Draxxin 25 Injectable Solution on porcine reproductive performance, pregnancy and lactation have not been determined. Intramuscular injection in swine can cause a local tissue reaction that may result in trim loss of edible tissue at slaughter. The safety of Draxxin 25 Injectable Solution has not been demonstrated in pigs less than 4 weeks of age.
WARNINGS: Treated pigs must not be slaughtered for use in food for at least 9 days after the latest treatment with this drug. To limit the potential development of antimicrobial resistance, Draxxin 25 Injectable Solution should only be used for control of SRD outbreak when groups of pigs are at high risk of developing SRD.
Keep out of reach of children.
Note: To reduce the possibility of excess trim at the injection site, do not slaughter swine for at least 14 days after the latest treatment with this drug.
Adverse Reactions
Occasionally, transient local erythema or swelling may occur following intramuscular injection.Clinical Pharmacology
Mechanism of Action: At physiological pH, tulathromycin (a weak base) is approximately 50 times more soluble in hydrophilic than hydrophobic media. This solubility profile is consistent with the extracellular pathogen activity typically associated with the macrolides.1 Markedly higher tulathromycin concentrations are observed in the lungs as compared to the plasma. The extent to which lung concentrations represent free (active) drug was not examined. Therefore, the clinical relevance of these elevated lung concentrations is undetermined.
Although the relationship between tulathromycin and the characteristics of its antimicrobial effects has not been characterized, as a class, macrolides tend to be primarily bacteriostatic, but may be bactericidal against some pathogens.2 They also tend to exhibit concentration independent killing; the rate of bacterial eradication does not change once serum drug concentrations reach 2 to 3 times the minimum inhibitory concentration (MIC) of the targeted pathogen. Under these conditions, the time that serum concentrations remain above the MIC becomes the major determinant of antimicrobial activity. Macrolides also exhibit a post-antibiotic effect (PAE), the duration of which tends to be both drug and pathogen dependent.
In general, by increasing the macrolide concentration and the exposure time, the PAE will increase to some maximal duration. Of the two variables, concentration and exposure time, drug concentration tends to be the most powerful determinant of the duration of PAE.
Tulathromycin is eliminated from the body primarily unchanged via biliary excretion.
1Carbon, C. 1998. Pharmacodynamics of Macrolides, Azalides, and Streptogramins: Effect on Extracellular Pathogens. Cli. Infect. Dis. 27:28-32.
2Nightingale, C.J. 1997. Pharmacokinetics and Pharmacodynamics of Newer Macrolides. Pediatr. Infect. Dis. J., 16:438-443.
Pharmacokinetic: Following intramuscular administration to feeder pigs at a dosage of 2.5 mg/kg body weight, tulathromycin is completely and rapidly absorbed (Tmax~0.25 hour). Subsequently the drug rapidly distributes into body tissues, achieving a volume of distribution exceeding 15 L/kg. The free drug is rapidly cleared from the systemic circulation (CLsystemic = 187 mL/hr/kg). However, it has a long terminal elimination half-life (60 to 90 hours) owing to its extensive volume of distribution. Although pulmonary tulathromycin concentrations are substantially higher than concentrations observed in the plasma, the clinical significance of these findings is undetermined. There are no gender differences in swine tulathromycin pharmacokinetics.
Comparative Bioavailability Summary: A single intramuscular dose of 2.5 mg tulathromycin/kg body weight (BW) of either Draxxin Injectable solution (100 mg/mL) or Draxxin 25 Injectable Solution (25 mg/mL) resulted in comparable tulathromycin concentrations in swine plasma. Summary statistics for pharmacokinetic (PK) parameters per treatment group are shown in Table 2.
Table 2. A summary of PK results (average ± SD) per treatment (n = 32 for T01; n = 30 for T02) following the administration of Draxxin Injectable Solution (T01; reference) and Draxxin 25 Injectable Solution (T02; test) in swine as a single IM injection of 2.5 mg tulathromycin/kg BW
|
PK Parameter |
Draxxin Injectable Solution (reference) |
Draxxin 25 Injectable Solution |
|
Cmax (ng/mL) |
551 ± 148 |
450 ± 121 |
|
AUC0-LOQ (h•ng/mL) |
8134 ± 1318 |
8227 ± 1166 |
|
AUC0-inf (h•ng/mL) |
8664 ± 1357 |
8610 ± 1203 |
|
Tmax (h) |
0.382 ± 0.121 |
0.669 ± 0.639 |
|
T1/2 (h) |
70.1 ± 25.2 |
67.7 ± 16.0 |
Cmax - maximum plasma concentration
AUC0-LOQ - the area under the plasma concentration vs. time curve from time of injection to the limit of quantification of the assay
AUC0-inf - the area under the plasma concentration vs. time curve from time of injection extrapolated to infinity
Tmax - the time after initial injection to when Cmax occurs
T1/2 - the plasma elimination half-life of tulathromycin
The ratio of the means between Draxxin Injectable Solution (100 mg/mL), and Draxxin 25 Injectable Solution (25 mg/mL) was contained within the 90% confidence limits of 0.80 - 1.25 for AUC0-LOQ and 0.70 - 1.43 for Cmax. As a result, Draxxin 25 Injectable Solution is considered pharmacologically equivalent to Draxxin Injectable Solution when administered to swine by IM injection at a dose rate of 2.5 mg tulathromycin/kg BW.
MICROBIOLOGY:
In vitro activity of tulathromycin has been demonstrated against commonly isolated bacterial and mycoplasma pathogens involved in SRD including Actinobacillus pleuropneumoniae, Pasteurella multocida, Mycoplasma hyopneumoniae, Bordetella bronchiseptica, Haemophilus parasuis, Streptococcus suis and Arcanobacterium (Actinomyces) pyogenes.
Table 3. The MICs of tulathromycin were determined for isolates obtained from swine enrolled in SRD field studies in the U.S. and Canada during 2000 through 2002 and during 2007-2008.
|
Organism |
Date isolated |
No. Isolates |
MIC50* µg/mL |
MIC90* µg/mL |
MIC Ranges µg/mL |
|
Actinobacillus pleuropneumoniae |
2000-2002 |
135 |
16 |
32 |
16 to 32 |
|
2007-2008 |
89 |
16 |
16 |
4 to 32 |
|
|
Pasteurella multocida |
2000-2002 |
55 |
1 |
2 |
0.5 to > 64 |
|
2007-2008 |
40 |
1 |
2 |
≤ 0.03 to 2 |
|
|
Mycoplasma hyopneumoniae |
2000-2002 |
30 |
8 |
> 32 |
≤ 0.063 to > 32 |
|
2007-2008 |
46 |
>64 |
> 64 |
< 0.125 to > 64 |
* The minimum inhibitory concentration for 50% and 90% of the isolates
EFFICACY:
Plasma concentrations of tulathromycin administered as Draxxin Injectable Solution or as Draxxin 25 Injectable Solution were demonstrated to be pharmacologically equivalent (see CLINICAL PHARMACOLOGY, Comparative Bioavailability Summary). Therefore effectiveness studies conducted with Draxxin Injectable Solution support the effectiveness for Draxxin 25 Injectable Solution.
A total of 266 pigs with naturally occurring SRD were treated with Draxxin Injectable Solution in a multi-location field study (5 United States, 1 Canada). Responses to treatment were compared to 267 saline-treated controls. Success was defined as a pig with normal attitude, normal respiration, and a rectal temperature of ≤ 40°C on day 7. The treatment success rate was significantly greater (P≤0.05) in Draxxin Injectable Solution-treated pigs (71.1%) compared to saline-treated pigs (46.4%). Mortality rates were 2.6% (7 of 266) in the Draxxin Injectable Solution-treated pigs compared to 9.0% (24 of 267) in the saline-treated controls.
The efficacy of tulathromycin in the treatment of SRD associated with Mycoplasma hyopneumoniae was confirmed in two experimentally-induced infection model studies using M. hyopneumoniae strains with MIC of tulathromycin > 64 µg/mL. In each study, 36 pigs were
administered saline intramuscularly (IM) at a dosage of 0.025mL/kg body weight and 36 pigs were administered tulathromycin IM at a dosage of 2.5 mg/kg body weight. Treatments were administered ten days after the first M. hyopneumoniae inoculation. All pigs were weighed, euthanized and necropsied on Study Day 10. For each pig, the percent of gross pneumonic lesions by lobe was determined. The primary clinical endpoint to determine the efficacy of tulathromycin was the difference in lung lesions scores between treatment groups. The percentage of gross pneumonic lesions was significantly less (P<0.0001) for tulathromycin-treated pigs than for saline-treated pigs in both studies (8.52% vs. 23.62% and 11.31% vs. 26.42%). Treatment with Draxxin Injectable Solution did not eliminate Mycoplasma hyopneumoniae from infected lungs. The clinical significance of this finding, as it relates to potential relapses and/or persistent subclinical infections, is unknown.
In another multi-location field study to evaluate the control of SRD (5 United States, 1 Canada), 226 pigs exposed to naturally occurring SRD were administered Draxxin Injectable Solution. Treatment was initiated when at least 15% of the pigs in the pen expressed clinical signs associated with SRD (rectal temperature ≥ 40°C and at least moderate distress in breathing and at least moderate depression). Draxxin Injectable Solution-treated pigs had a significant (P< 0.05) higher treatment success rate (59%) compared to saline-treated pigs (41%). An animal was classified as a Treatment Success on Study Day 7, if it was alive, and had a respiration score of ≤ 1 (scale of 0 to 3 where 0 is normal), and had a rectal temperature of < 40°C. Failure to meet any one of the criteria classified the animal as a Treatment Failure.
ANIMAL SAFETY:
Plasma concentrations of tulathromycin administered as Draxxin Injectable Solution or as Draxxin 25 Injectable Solution were demonstrated to be pharmacologically equivalent (see CLINICAL PHARMACOLOGY, Comparative Bioavailability Summary). Therefore systemic target animal safety studies conducted with Draxxin Injectable Solution support the systemic safety for Draxxin 25 Injectable Solution.
Safety studies were conducted in pigs receiving a single intramuscular dose of 25 mg tulathromycin per kg body weight, or 3 weekly intramuscular doses of 2.5, 7.5 or 12.5 mg/kg body weight. In all groups, transient indications of pain after injection were seen, including restlessness and excessive vocalization. Tremors occurred briefly in one animal receiving 7.5 mg/kg body weight. Discolouration and edema of injection site tissues and corresponding histopathologic changes were seen in animals at all dosages and resolved over time. No other drug-related lesions were observed macroscopically or microscopically.
Sixteen growing pigs were injected with either saline or Draxxin 25 Injectable Solution as a single injection of 4 mL. Injection site observations included two instances of erythema in the Draxxin 25-treated group on Day 1 post-injection. No heat, sensitivity, firmness, necrosis, drainage, or swelling was observed at any injection sites in either treatment group. The gross and microscopic findings in the Draxxin 25-treated group were consistent with inflammatory changes induced by injections and were considered to be mild or moderate with progression to macroscopic resolution by Day 28 post-injection and microscopic resolution by Day 42 post-injection.
Storage
Store between 15 and 30°C. Contents should be used within 90 days after the first dose is removed.PRESENTATION: Draxxin 25 Injectable Solution is available in 100 mL vials.
Zoetis® and Draxxin are registered trademarks of Zoetis or its Licensors.
Zoetis Canada Inc., Kirkland QC H9H 4M7
40042579
10023092-11-0
CPN: 1198500.4
16,740 TRANS-CANADA HIGHWAY, KIRKLAND, QC, H9H 4M7
| Order Desk: | 800-663-8888 | |
| Technical Services Canada: | 800-461-0917 | |
| Technical Services USA: | 800-366-5288 | |
| Website: | www.zoetis.ca |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". Animalytix assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the Animalytix service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-08-27
