Signifor LAR Injection: Package Insert / Prescribing Info
Package insert / product label
Generic name: pasireotide
Dosage form: injection
Drug class: Somatostatin and somatostatin analogs
J Code (medical billing code): J2502 (1 mg, injection)
Medically reviewed by Drugs.com. Last updated on Jul 22, 2024.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Drug Interactions
- Use In Specific Populations
- Overdosage
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
SIGNIFOR® LAR (pasireotide) for injectable suspension, for intramuscular use
Initial U.S. Approval: 2012
Recent Major Changes
|
Warnings and Precautions, Steatorrhea and Malabsorption of Dietary Fats (5.6) |
7/2024 |
Indications and Usage for Signifor LAR Injection
Signifor LAR Injection Dosage and Administration
- Evaluate fasting plasma glucose (FPG), hemoglobin A1c (HbA1c), liver enzyme tests, electrocardiogram (ECG), serum magnesium, and serum potassium prior to starting. (2.1)
- Optimize glucose control in patients with poorly controlled diabetes mellitus prior to starting. (2.1)
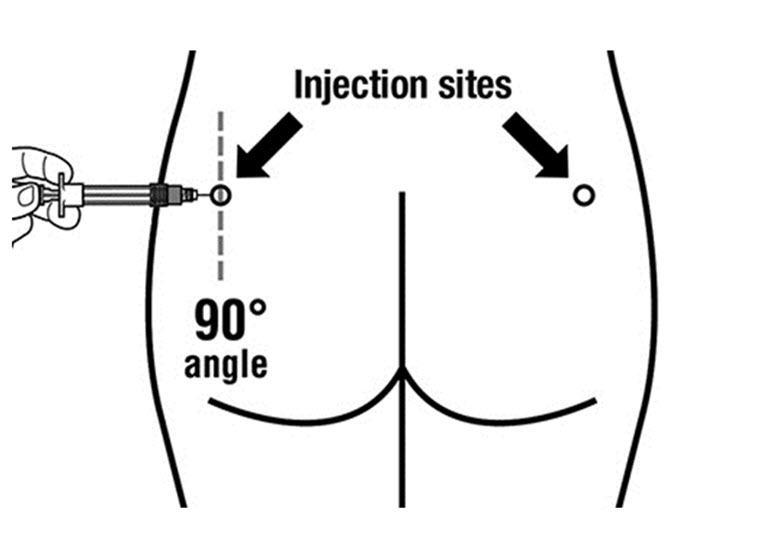
- Must be administered by a health care professional only by intramuscular injection in the right or left gluteus immediately after reconstitution. (2.2)
- Initial Dose:
- Adjust dose based on response and tolerability. (2.4)
- Patients with Hepatic Impairment:
- Follow reconstitution and administration instructions. (2.6)
Dosage Forms and Strengths
SIGNIFOR LAR for injectable suspension: 10 mg, 20 mg, 30 mg, 40 mg, and 60 mg, powder in a vial to be reconstituted with the provided 2 mL diluent. (3)
Contraindications
None. (4)
Warnings and Precautions
- Hyperglycemia, Diabetes, and Ketoacidosis: Sometimes severe. Monitor glucose levels as clinically appropriate during therapy. Monitor glucose levels more frequently in the months that follow initiation or discontinuation of SIGNIFOR LAR therapy and following SIGNIFOR LAR dose adjustment. Use anti-diabetic treatment if indicated.If ketoacidosis is suspected, discontinue SIGNIFOR LAR and promptly evaluate and treat the patient. (2.1, 5.1)
- Bradycardia and QT Prolongation: Use with caution in at-risk patients; evaluate ECG and electrolytes prior to dosing and periodically while on treatment. (2.1, 5.2, 7.1)
- Liver Test Elevations: Evaluate liver enzyme tests prior to and during treatment. (2.1, 5.3)
- Cholelithiasis and Complications of Cholelithiasis: Monitor periodically. Discontinue if complications of cholelithiasis are suspected. (5.4)
- Pituitary Hormone Deficiency(ies): Monitor for occurrence periodically and treat if clinically indicated. (5.5)
- Steatorrhea and Malabsorption of Dietary Fats: New onset steatorrhea, stool discoloration, loose stools, abdominal bloating, and weight loss may occur. If new occurrence or worsening of these symptoms are reported, evaluate for potential pancreatic exocrine insufficiency (5.6)
Adverse Reactions/Side Effects
Adverse drug reactions associated with SIGNIFOR LAR and occurring in ≥ 20% of patients were diarrhea, cholelithiasis, hyperglycemia, and diabetes mellitus. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Recordati Rare Diseases Inc. at 1-888-575-8344 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Use In Specific Populations
- Females and Males of Reproductive Potential: Advise premenopausal females of the potential for an unintended pregnancy (8.3)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 7/2024
Full Prescribing Information
1. Indications and Usage for Signifor LAR Injection
2. Signifor LAR Injection Dosage and Administration
2.1 Recommended Baseline Evaluations Prior to Initiation of SIGNIFOR LAR
Prior to the initiation of SIGNIFOR LAR, it is recommended that patients have the following baseline evaluations:
- Fasting plasma glucose (FPG) and hemoglobin A1c (HbA1c) [see Warnings and Precautions (5.1)]
- Liver tests [see Warnings and Precautions (5.3)]
- Electrocardiogram (ECG), serum potassium and serum magnesium levels [see Warnings and Precautions (5.2)]
Patients with poorly controlled diabetes mellitus, who have inadequate glucose control, should have anti-diabetic therapy optimized prior to starting SIGNIFOR LAR [see Warnings and Precautions (5.1)].
2.2 Important Administration Instructions
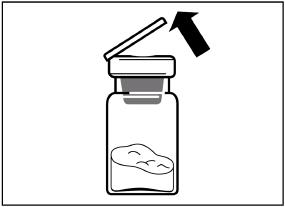
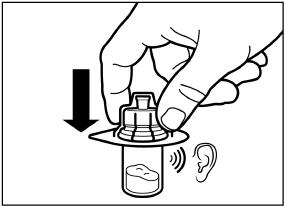
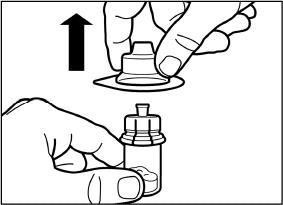
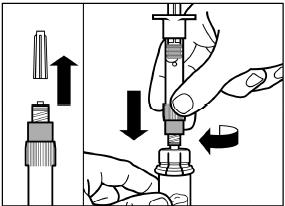
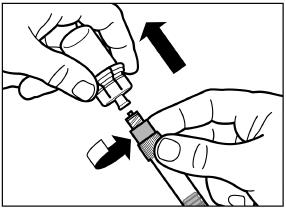
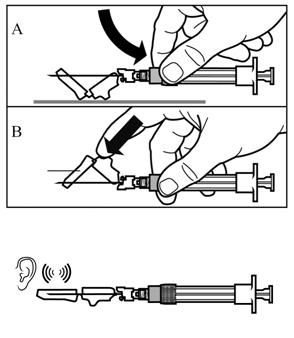
SIGNIFOR LAR must be reconstituted by a trained healthcare professional immediately before use. Illustrations on reconstitution are found in Instructions for Use [see Dosage and Administration (2.6)].
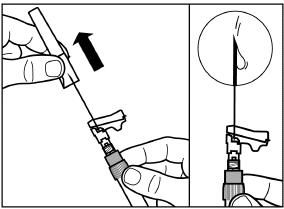
SIGNIFOR LAR must be inspected visually before use. The suspension should appear free of foreign particulates and should be homogeneous after mixing.
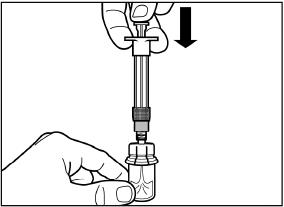
SIGNIFOR LAR must be administered by a trained healthcare professional only by intramuscular injection in the right or left gluteus immediately after reconstitution. SIGNIFOR LAR must never be administered intravenously.
2.3 Recommended Initial Dose
Acromegaly
The recommended initial dose of SIGNIFOR LAR for the treatment of acromegaly is 40 mg administered by intramuscular injection once every 4 weeks (every 28 days) [see Dosage and Administration (2.6)].
Cushing's Disease
The recommended initial dose of SIGNIFOR LAR for the treatment of Cushing's disease is 10 mg administered by intramuscular injection once every 4 weeks (every 28 days) [see Dosage and Administration (2.6)].
2.4 Dose Adjustment and Monitoring
Acromegaly
The dose may be increased to a maximum of 60 mg for patients who have not normalized growth hormone (GH) and/or age and sex adjusted insulin-like growth factor-1 (IGF-1) levels after 3 months of treatment with SIGNIFOR LAR at 40 mg and who tolerate this dose.
Management of SIGNIFOR LAR-related adverse reactions or over-response to treatment (age and sex adjusted IGF-1 less than the lower limit of normal) may require dose reduction. The dose may be decreased, either temporarily or permanently, by 20 mg decrements [see Warnings and Precautions (5)].
Cushing's Disease
Following 4 months of treatment with the initial dose of 10 mg once every 28 days, the dose may be increased for patients who have not normalized 24-hour urinary free cortisol (UFC) and who tolerate this dose, up to a maximum dose of 40 mg once every 28 days.
Management of suspected adverse reactions or over-response to treatment (e.g., cortisol levels less than the lower limit of the normal range or in the low part of the normal range in patients with symptoms suggestive of adrenal insufficiency) may require dose reduction to the previous tolerated dose, dose interruption, or drug discontinuation of SIGNIFOR LAR. For patients treated with 10 mg once every 28 days, the dose may be either interrupted or discontinued [see Warnings and Precautions (5)].
2.5 Dose in Patients With Hepatic Impairment
For patients with moderately impaired hepatic function (Child-Pugh B) [see Use in Specific Populations (8.6)]:
- Acromegaly: The recommended initial dose for acromegaly patients with moderately impaired hepatic function is 20 mg once every 4 weeks and the maximum recommended dose is 40 mg once every 4 weeks.
- Cushing's Disease: The recommended initial dose for Cushing's disease patients with moderately impaired hepatic function is 10 mg once every 4 weeks and the maximum recommended dose is 20 mg once every 4 weeks.
Avoid use in patients with severe hepatic impairment (Child-Pugh C) [see Use in Specific Populations (8.6)].
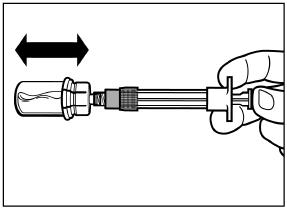
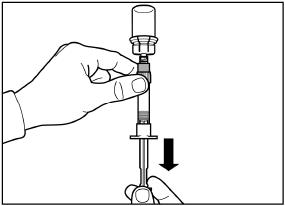
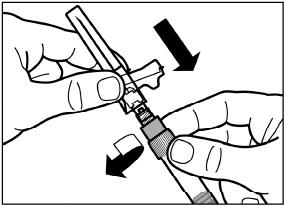
2.6 Reconstitution and Intramuscular Injection Instructions
After reconstitution of the SIGNIFOR LAR vial with the provided 2 mL diluent, the intramuscular injectable suspension will have a final concentration of:
| Strength per Vial | Final Concentration When Reconstituted (total product strength per total volume) | Final Concentration When Reconstituted (per mL) |
| 10 mg | 10 mg/2 mL | 5 mg/mL |
| 20 mg | 20 mg/2 mL | 10 mg/mL |
| 30 mg | 30 mg/2 mL | 15 mg/mL |
| 40 mg | 40 mg/2 mL | 20 mg/mL |
| 60 mg | 60 mg/2 mL | 30 mg/mL |
The entire contents of the reconstituted solution should be administered immediately.
| PAY PARTICULAR ATTENTION: |
| There are 2 critical steps in the reconstitution of SIGNIFOR LAR. Not following these 2 steps could result in failure to deliver the drug appropriately.
|
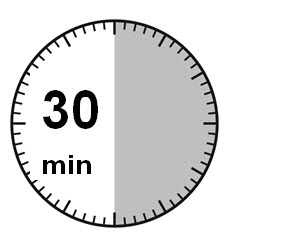
| 1) The injection kit must reach room temperature (see Step 1 in Instructions for Use). Remove the SIGNIFOR LAR injection kit from refrigerated storage and let the kit stand at room temperature for a minimum of 30 minutes before reconstitution, but do not exceed 24 hours. |
| 2) After adding the diluent solution, shake the vial moderately in a horizontal direction for a minimum of 30 seconds until uniform suspension is formed (see Step 4 in Instructions for Use). |
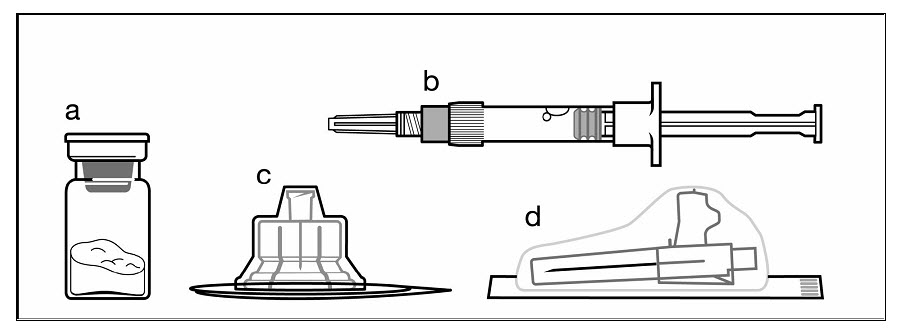
The following items are included in the injection kit:
a) One vial containing SIGNIFOR LAR powder
b) One prefilled syringe containing the diluent solution for reconstitution
c) One vial adapter for drug product reconstitution
d) One safety injection needle (20G x 1.5")
Figure 1. Items Included in Injection Kit
SIGNIFOR LAR suspension must only be reconstituted immediately before administration.
Follow the directions in the Instructions for Use to ensure proper reconstitution of SIGNIFOR LAR before intramuscular injection.
SIGNIFOR LAR should only be administered by a trained healthcare professional.
3. Dosage Forms and Strengths
SIGNIFOR LAR for injectable suspension: 10 mg, 20 mg, 30 mg, 40 mg, or 60 mg of slightly yellow to yellow powder in a vial and 2 mL diluent.
5. Warnings and Precautions
5.1 Hyperglycemia, Diabetes, and Ketoacidosis
SIGNIFOR LAR can cause increases in blood glucose levels which are sometimes severe. There have been postmarketing cases of ketoacidosis with SIGNIFOR LAR in patients with history of diabetes and in patients without history of diabetes.
In the acromegalic patient study, 5 patients naïve to drug therapy treated with SIGNIFOR LAR (2 of whom were normoglycemic at baseline) and none in the active comparator group were hospitalized for hyperglycemia (blood glucose range 359-506 mg/dL). Two additional patients who had received active comparator in the main trial and were switched to SIGNIFOR LAR in the extension trial, were hospitalized for elevated glucose levels while on SIGNIFOR LAR; one of those patients developed diabetic ketoacidosis. In the Cushing's disease study, 2 patients were hospitalized for elevated blood glucose.
In clinical studies for acromegaly and Cushing's disease, SIGNIFOR LAR caused an increase in the incidence of diabetes and prediabetes. A majority of patients, including those with normal glucose tolerance, prediabetes and diabetes, experienced increased glucose levels within the first 3 months of treatment [see Adverse Reactions (6.1)]. In the drug-naïve acromegaly study, the prevalence of diabetes increased from 30% at baseline to 60% at Month 12. In the study evaluating acromegaly patients previously treated with somatostatin analogs, the prevalence of diabetes increased from 71% at baseline to 87% at Month 6 in patients treated with SIGNIFOR LAR 40 mg, and from 60% to 84% in patients treated with SIGNIFOR LAR 60 mg. In the Cushing's disease study, the prevalence of diabetes increased from 40% at baseline to 56% at Month 12.
Patients with poor baseline glycemic control are at higher risk of developing severe hyperglycemia. Assess FPG and HbA1c prior to starting treatment with SIGNIFOR LAR [see Dosage and Administration (2.1)]. In patients with poorly controlled diabetes mellitus, optimize anti-diabetic treatment before SIGNIFOR LAR initiation. Monitor blood glucose weekly for the first 3 months after initiating SIGNIFOR LAR and the first 4- to 6 weeks after dose increases. Continue monitoring thereafter, as clinically appropriate.
Patients who develop significant hyperglycemia on SIGNIFOR LAR may require initiation of anti-diabetic treatment or adjustment in their current anti-diabetic treatment. The optimal treatment for the management of SIGNIFOR LAR-induced hyperglycemia is not known. If hyperglycemia cannot be controlled despite medical management, reduce the dose of SIGNIFOR LAR or discontinue SIGNIFOR LAR. Assess FPG and HbA1c after SIGNIFOR LAR discontinuation, if indicated. Patients receiving anti-diabetic treatment may require more frequent blood glucose monitoring and dose adjustment to their anti-diabetic drug therapy to mitigate the risk of hypoglycemia after discontinuing SIGNIFOR LAR.
Patients who present with signs and symptoms consistent with severe metabolic acidosis should be assessed for ketoacidosis regardless of diabetes history. In some reported post-marketing cases of ketoacidosis in patients taking SIGNIFOR LAR, factors predisposing to ketoacidosis such as acute illness, infection, pancreatic disorders (e.g., pancreatic malignancy or pancreatic surgery), and alcohol abuse were present. If ketoacidosis is suspected, discontinue SIGNIFOR LAR and promptly evaluate and treat the patient.
5.2 Bradycardia and QT Prolongation
Bradycardia
Bradycardia has been reported with the use of SIGNIFOR LAR [see Adverse Reactions (6.1)]. Patients with cardiac disease and/or risk factors for bradycardia, such as history of clinically significant bradycardia, high-grade heart block, or concomitant use of drugs associated with bradycardia, should be monitored. Adjustments in the dose of drugs known to slow the heart rate (e.g., beta-blockers, calcium channel blockers) and correction of electrolyte disturbances may be necessary when initiating or during the course of SIGNIFOR LAR treatment.
QT Prolongation
In cardiac electrophysiology studies (i.e., thorough QT studies) with pasireotide via subcutaneous route, QT prolongation occurred at therapeutic and supra-therapeutic doses [see Clinical Pharmacology (12.2)].
In the clinical studies, a corrected QT interval (i.e., QTcF) of greater than 480 ms was reported in 6 patients and an increase in the QTcF from baseline of greater than 60 ms was reported for seven patients on SIGNIFOR LAR. No patient on SIGNIFOR LAR had a QTcF value of greater than 500 ms [see Adverse Reactions (6.1), Clinical Pharmacology (12.2)].
SIGNIFOR LAR should be used with caution in patients who are at significant risk of developing prolongation of the QT interval, such as those listed below:
- with congenital long QT prolongation
- with uncontrolled or significant cardiac disease including recent myocardial infarction, congestive heart failure, unstable angina or clinically significant bradycardia
- on anti-arrhythmic therapy or other substances that are known to lead to QT prolongation
- with hypokalemia and/or hypomagnesemia
A baseline ECG is recommended prior to initiating therapy with SIGNIFOR LAR. Monitoring for an effect on the QT interval at the time of maximum drug concentration (21 days after injection) should be obtained in patients at risk. Hypokalemia or hypomagnesemia must be corrected prior to initiating SIGNIFOR LAR and should be monitored periodically during therapy.
5.3 Liver Test Elevations
Increases in liver enzymes have been observed with SIGNIFOR LAR. In all Phase 3 acromegaly studies and across all doses, alanine aminostransferase (ALT) or aspartate aminotransferase (AST) elevations greater than 3 times the upper limit of normal (ULN) were observed in 4% of acromegaly patients and ALT or AST elevations greater than 5 times the ULN were observed in 1% of acromegaly patients treated with SIGNIFOR LAR.
In the Phase 3 Cushing's disease study and across all doses, ALT or AST elevations greater than 3 times the ULN were observed in 14% of Cushing's disease patients and ALT or AST elevations greater than 5 times the ULN were observed in 5% of Cushing's disease patients treated with SIGNIFOR LAR.
Assessment of liver function is recommended prior to treatment with SIGNIFOR LAR , and after the first 2- to 3 weeks, then monthly for 3 months. Thereafter, liver function should be monitored as clinically indicated. Patients who develop increased transaminase levels, should be monitored until values return to pre-treatment levels. Treatment with SIGNIFOR LAR should be discontinued if signs or symptoms suggestive of clinically significant liver impairment develop. Following discontinuation of treatment with SIGNIFOR LAR, patients should be monitored until resolution. Treatment should not be restarted, if the liver function abnormalities are suspected to be related to SIGNIFOR LAR.
5.4 Cholelithiasis and Complications of Cholelithiasis
Cholelithiasis was reported in 33% of drug-naïve and 10% of inadequately controlled (40 mg dose) acromegaly patients treated with SIGNIFOR LAR in clinical trials [see Adverse Reactions (6)]. Cholelithiasis was reported in 33% of Cushing's disease patients treated with SIGNIFOR LAR. There have been postmarketing reports of cholelithiasis (gallstones) resulting in complications, including cholecystitis or cholangitis and requiring cholecystectomy in patients taking SIGNIFOR LAR. Patients should be monitored periodically. If complications of cholelithiasis are suspected, discontinue SIGNIFOR LAR and treat appropriately.
5.5 Pituitary Hormone Deficiency(ies)
Suppression of anterior pituitary hormones may occur on SIGNIFOR LAR. Monitoring pituitary function (e.g., thyroid, adrenal, gonadal) prior to initiation of therapy with SIGNIFOR LAR, as well as periodically during treatment, as clinically appropriate, is recommended. Patients should be monitored for and instructed on the signs and symptoms of adrenal insufficiency during therapy. If adrenal insufficiency is suspected, it should be confirmed and treated per standard of care with exogenous glucocorticoids at replacement doses.
5.6 Steatorrhea and Malabsorption of Dietary Fats
New onset steatorrhea, stool discoloration and loose stools have been reported in patients receiving somatostatin analogs, including pasireotide products. Somatostatin analogs reversibly inhibit secretion of pancreatic enzymes and bile acids, which may result in malabsorption of dietary fats and subsequent symptoms of steatorrhea, loose stools, abdominal bloating, and weight loss. If new occurrence or worsening of these symptoms are reported in patients receiving SIGNIFOR LAR, evaluate patients for potential pancreatic exocrine insufficiency and manage accordingly.
6. Adverse Reactions/Side Effects
Clinically significant adverse reactions that appear in other sections of the labeling include:
- Hyperglycemia, Diabetes, and Ketoacidosis [see Warnings and Precautions (5.1)]
- Bradycardia and QT Prolongation [see Warnings and Precautions (5.2)]
- Liver Test Elevations [see Warnings and Precautions (5.3)]
- Cholelithiasis and Complications of Cholelithiasis [see Warnings and Precautions (5.4)]
- Pituitary Hormone Deficiency(ies) [see Warnings and Precautions (5.5)]
- Steatorrhea and Malabsorption of Dietary Fats [see Warnings and Precautions (5.6)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of a drug cannot be directly compared to rates in clinical trials of another drug and may not reflect the rates observed in practice.
Drug-Naïve Patients With Acromegaly
The data described in Table 1 are derived from an active-controlled trial in patients with acromegaly naïve to previous drug therapy [see Clinical Studies (14.1)]. The data reflect exposure of 178 patients with acromegaly to SIGNIFOR LAR for a mean duration of 43 weeks. In the overall study population, 52% were female and the average age of patients was 45 years.
Table 1 presents common adverse reactions associated with SIGNIFOR LAR in this study. These adverse reactions were not present at baseline or, if present, worsened from baseline and occurred in at least 5% of patients treated with SIGNIFOR LAR.
| aDiabetes mellitus includes the following PTs: diabetes mellitus and type 2 diabetes mellitus. bSinus bradycardia includes the following PTs: bradycardia and sinus bradycardia. cInjection-site reaction related AEs includes the following PTs: injection-site pain, injection-site reaction, injection-site haematoma, injection-site pruritus, injection-site swelling, injection-site erythema. |
|||||
| Adverse Reaction Type | SIGNIFOR LAR (40-60 mg) % N = 178 | Active Comparator % N = 180 |
|||
| Hyperglycemia Related Adverse Reactions | |||||
| Hyperglycemia | 29 | 8 | |||
| Diabetes mellitusa | 26 | 4 | |||
| Blood glucose increased | 8 | 2 | |||
| Glycosylated hemoglobin increased | 6 | 2 | |||
| Hypoglycemia | 5 | 7 | |||
| Gastrointestinal Related Adverse Reactions | |||||
| Diarrhea | 39 | 45 | |||
| Abdominal pain | 18 | 22 | |||
| Nausea | 14 | 22 | |||
| Abdominal distension | 12 | 12 | |||
| Vomiting | 8 | 7 | |||
| Abdominal pain upper | 6 | 8 | |||
| Hepatobiliary Related Adverse Reactions | |||||
| Cholelithiasis | 26 | 36 | |||
| Cardiac Related Adverse Reactions | |||||
| Sinus bradycardiab | 10 | 7 | |||
| Hypertension | 8 | 7 | |||
| Nervous System Related Adverse Reactions | |||||
| Headache | 19 | 26 | |||
| Dizziness | 10 | 11 | |||
| Skin Related Adverse Reactions | |||||
| Alopecia | 18 | 19 | |||
| Infections Related Adverse Reactions | |||||
| Nasopharyngitis | 16 | 16 | |||
| Influenza | 8 | 4 | |||
| Upper respiratory tract infection | 7 | 3 | |||
| Cough | 5 | 8 | |||
| Laboratory Related Adverse Reactions | |||||
| Blood creatine phosphokinase increased | 13 | 12 | |||
| Alanine aminotransferase increased | 8 | 4 | |||
| Aspartate aminotransferase increased | 6 | 4 | |||
| Lipase increased | 6 | 7 | |||
| Weight decreased | 5 | 4 | |||
| General and Injection-Site Related Adverse Reactions | |||||
| Fatigue | 10 | 10 | |||
| Injection-site reactionc | 7 | 7 | |||
| Musculoskeletal and Connective Tissue Related Adverse Reactions | |||||
| Arthralgia | 10 | 12 | |||
| Back pain | 8 | 11 | |||
| Pain in extremity | 7 | 4 | |||
| Blood Related Adverse Reactions | |||||
| Anemia | 6 | 6 | |||
Other notable adverse reactions which occurred with a frequency of 5% or less for SIGNIFOR LAR were: adrenal insufficiency (3%); glucose tolerance impaired (1%); QT-prolongation (4%); blood amylase increased (2%).
Patients With Acromegaly Inadequately Controlled on Other Somatostatin Analogs at Baseline
The data described in Table 2 are derived from an active-controlled study in patients with acromegaly inadequately controlled at baseline on other somatostatin analogs [see Clinical Studies (14.2)]. These data reflect exposure of 63 and 62 patients to SIGNIFOR LAR 40 mg and 60 mg, respectively, for a mean duration of 24 weeks. In the overall study population, 56% were female and the average age of patients was 45 years.
Table 2 presents common adverse reactions associated with SIGNIFOR LAR in this study. These common adverse reactions were not present at baseline, or if present, worsened from baseline and occurred in at least 5% of patients treated with SIGNIFOR LAR.
| aDiabetes mellitus includes the following PTs: diabetes mellitus and type 2 diabetes mellitus. | |||
| Adverse Drug Reactions | SIGNIFOR LAR 40 mg % N = 63 | SIGNIFOR LAR 60 mg % N = 62 | Active Comparators % N = 66 |
| Hyperglycemia Related Adverse Reactions | |||
| Hyperglycemia | 33 | 30 | 14 |
| Diabetes mellitusa | 21 | 31 | 9 |
| Blood glucose increased | 5 | 7 | 0 |
| Hypoglycemia | 3 | 7 | 0 |
| Gastrointestinal Related Adverse Reactions | |||
| Diarrhea | 16 | 19 | 5 |
| Abdominal pain | 8 | 8 | 3 |
| Nausea | 3 | 7 | 3 |
| Hepatobiliary Related Adverse Reactions | |||
| Cholelithiasis | 10 | 13 | 14 |
| Cardiac Related Adverse Reactions | |||
| Atrioventricular block first degree | 6 | 0 | 0 |
| Nervous System Related Adverse Reactions | |||
| Headache | 14 | 3 | 5 |
| Dizziness | 8 | 2 | 3 |
| Skin and Subcutaneous Tissue Related Adverse Reactions | |||
| Alopecia | 2 | 7 | 0 |
| Infections Related Adverse Reactions | |||
| Nasopharyngitis | 6 | 11 | 3 |
| Blood Related Adverse Reactions | |||
| Anemia | 6 | 3 | 3 |
Other notable adverse reactions which occurred with a frequency of 5% or less in the SIGNIFOR LAR 40 mg, SIGNIFOR LAR 60 mg arm, respectively, were adrenal insufficiency (2% and 0%) and glucose tolerance impaired (3% and 5%).
Patients With Cushing's Disease
The data described in Table 3 are derived from a randomized clinical study in 150 patients with Cushing's disease [see Clinical Studies (14.3)]. These data reflect exposure of 74 and 76 patients to SIGNIFOR LAR at a starting dose of 10 mg and 30 mg once every 28 days, respectively, for a mean duration of 68 weeks. In the overall study population, 79% of patients were female and the average age was 39 years at study entry.
Table 3 presents common adverse reactions which were not present at baseline or, if present, worsened from baseline and occurred in at least 5% of patients treated with SIGNIFOR LAR.
| aDiabetes mellitus consists of the two terms: diabetes mellitus and type 2 diabetes mellitus. bAbdominal pain includes the term abdominal pain upper. cSinus bradycardia includes the term bradycardia. dFatigue includes the term asthenia. |
|
| Adverse Reactions | Overall % N = 150 |
| Endocrine Related Adverse Reactions | |
| Adrenal insufficiency | 7 |
| Blood cortisol decreased | 5 |
| Hyperglycemia Related Adverse Reactions | |
| Hyperglycemia | 47 |
| Diabetes mellitusa | 27 |
| Hypoglycemia | 15 |
| Blood glucose increased | 9 |
| Glycosylated hemoglobin (HbA1C) increased | 5 |
| Gastrointestinal Related Adverse Reactions | |
| Diarrhea | 39 |
| Nausea | 21 |
| Abdominal painb | 23 |
| Constipation | 7 |
| Abdominal distension | 5 |
| Flatulence | 5 |
| Vomiting | 5 |
| Hepatobiliary Related Adverse Reactions | |
| Cholelithiasis | 33 |
| Gamma-glutamyltransferase increased | 9 |
| Alanine aminotransferase increased | 7 |
| Cardiac Related Adverse Reactions | |
| Hypertension | 15 |
| Hypotension | 6 |
| Sinus bradycardiac | 5 |
| General Related Adverse Reactions | |
| Fatigued | 27 |
| Edema peripheral | 14 |
| Musculoskeletal and Connective Tissue Related Adverse Reactions | |
| Back pain | 11 |
| Arthralgia | 8 |
| Pain in extremity | 7 |
| Myalgia | 5 |
| Metabolism and Nutrition Related Adverse Reactions | |
| Decreased appetite | 10 |
| Hyperuricemia | 7 |
| Hypercholesterolemia | 6 |
| Blood Related Adverse Reactions | |
| Anemia | 5 |
Other adverse reactions which occurred at a frequency less than 5% were cholestasis (4%), glucose tolerance impaired (3%), aspartate aminotransferase increased (3%), vomiting (3%), lipase increased (3%), injection-site reactions (2%), ECG QT prolonged (1%), cholecystitis (1%), amylase increased (1%), and prothrombin time prolonged (1%).
Hyperglycemia
The average FPG levels in patients with acromegaly naïve to drug therapy study [see Clinical Studies (14.1)] across visits is shown in Figure 2 below.
|
| Figure 2. Mean Fasting Plasma Glucose (mg/dL) By Visit in the Study of Patients With Acromegaly Naïve to Drug Therapy* |
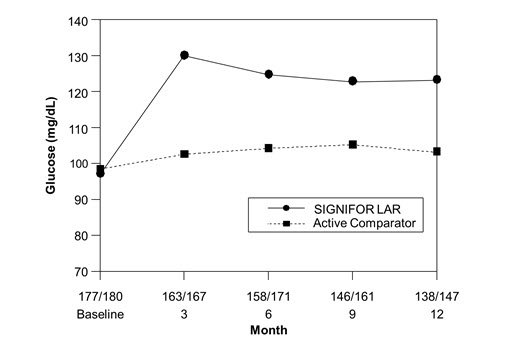 |
Pancreatic Enzyme Elevation and Pancreatitis
Asymptomatic elevations in lipase and alpha amylase were observed in 30% and 20% of patients receiving SIGNIFOR LAR in the drug naïve study in acromegaly, and in 1% and 3% of patients receiving SIGNIFOR LAR in the study of acromegaly patients previously treated. In the drug-naïve study, 2 acromegaly patients receiving SIGNIFOR LAR developed pancreatitis. In the Cushing's disease study, increased lipase was observed in 4% of patients, and 1 patient developed pancreatitis. Pancreatitis is a potential adverse reaction associated with the use of SIGNIFOR LAR due to the association between cholelithiasis and acute pancreatitis.
6.2 Postmarketing Experience
Additional adverse reactions have been identified during post-approval use of SIGNIFOR LAR. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Cholelithiasis resulting in complications, including cholecystitis and cholangitis, which have sometimes required cholecystectomy
Related/similar drugs
7. Drug Interactions
7.1 Effect of Other Drugs on SIGNIFOR LAR
Drugs That Prolong QT
Co-administration of drugs that prolong the QT interval with SIGNIFOR LAR may have additive effects on the prolongation of the QT interval. Monitoring effects on the QT interval at 21 days is recommended [see Warnings and Precautions (5.2)].
7.2 Effect of SIGNIFOR LAR on Other Drugs
Cyclosporine
Concomitant administration of cyclosporine with SIGNIFOR LAR may decrease the relative bioavailability of cyclosporine and, therefore, dose adjustment of cyclosporine to maintain therapeutic levels may be necessary.
Bromocriptine
Co-administration of SIGNIFOR LAR with bromocriptine may increase the blood levels of bromocriptine. Dose reduction of bromocriptine may be necessary.
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
The limited data with SIGNIFOR LAR in pregnant women are insufficient to inform a drug-associated risk for major birth defects and miscarriage. In embryo-fetal development studies in rabbits, findings indicating a developmental delay were observed with subcutaneous administration of pasireotide during organogenesis at doses less than the exposure in humans at the highest recommended dose; maternal toxicity was not observed at this dose (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2%-4% and 15%-20%, respectively.
Animal Data
In embryo-fetal development studies in rats given 1 mg/kg/day, 5 mg/kg/day, and 10 mg/kg/day subcutaneously throughout organogenesis, maternal toxicity was observed at all doses, including the lowest dose tested which had exposures 12 times higher than that at the maximum therapeutic dose based on area under the curve (AUC) comparisons across species. An increased incidence of early/total resorptions and malrotated limbs was observed in rats at 10 mg/kg/day. At 10 mg/kg/day in rats, the maternal systemic exposure (AUC) was 42179 ng*hr/mL, approximately 106 times the exposure in humans at the highest recommended dose of 60 mg SIGNIFOR LAR administered as an intramuscular injection once every 4 weeks.
In embryo-fetal development studies in rabbits given 0.05 mg/kg/day, 1 mg/kg/day, and 5 mg/kg/day subcutaneously through organogenesis, maternal toxicity was observed at 1 mg/kg/day, at a maternal systemic exposure (AUC) of 1906 ng*hr/mL, approximately 5 times higher than the maximum human therapeutic exposure. An increased incidence of unossified forepaw phalanx, indicative of a developmental retardation, was observed in rabbits at 0.05 mg/kg/day, with maternal systemic exposures less than the systemic exposure in humans at the highest recommended dose.
In pre- and post-natal developmental studies in rats given subcutaneous doses of 2 mg/kg/day, 5 mg/kg/day, and 10 mg/kg/day during gestation through lactation and weaning, maternal toxicity was observed at all doses including the lowest dose (9 times higher than the maximum therapeutic dose based on surface area comparisons across species). Retardation of physiological growth, attributed to GH inhibition was observed at 2 mg/kg/day during a pre- and post-natal study in rats. After weaning, body weight gains in the rat pups (F1 generation) exposed to pasireotide were comparable to controls, showing reversibility of this developmental delay.
8.2 Lactation
Risk Summary
There is no information available on the presence of SIGNIFOR LAR in human milk, the effects of the drug on the breastfed infant, or the effects of the drug on milk production. Studies show that pasireotide administered subcutaneously passes into the milk of lactating rats; however, due to species-specific differences in lactation physiology, animal data may not reliably predict drug levels in human milk (see Data). The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for SIGNIFOR LAR, and any potential adverse effects on the breastfed child from SIGNIFOR LAR or from the underlying maternal condition.
Data
Available data in animals have shown excretion of pasireotide in milk. After a single 1 mg/kg [14C]-pasireotide subcutaneous dose to lactating rats, the transfer of radioactivity into milk was observed. The overall milk:plasma (M/P) exposure ratio of total radioactivity was 0.28, based on AUC0-∞ values.
8.3 Females and Males of Reproductive Potential
Discuss the potential for unintended pregnancy with premenopausal women as the therapeutic benefits of a reduction in GH levels and normalization of IGF-1 in acromegalic females treated with pasireotide may lead to improved fertility.
Similarly, the therapeutic benefits of a reduction or normalization of serum cortisol levels in female patients with Cushing's disease treated with pasireotide may also lead to improved fertility.
8.4 Pediatric Use
Safety and effectiveness of SIGNIFOR LAR have not been established in pediatric patients.
8.5 Geriatric Use
Clinical studies of SIGNIFOR LAR did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
8.6 Hepatic Impairment
Dose adjustment is not required in patients with mild impaired hepatic function (Child-Pugh A), but is required for patients with moderately impaired hepatic function (Child-Pugh B). The safety and efficacy of SIGNIFOR LAR have not been established in patients with severe hepatic impairment (Child-Pugh C). No dosage recommendation can be given for patients with severe hepatic impairment (Child-Pugh C) [see Dosage and Administration (2.5), Clinical Pharmacology (12.3)].
10. Overdosage
In the event of overdosage, it is recommended that appropriate supportive treatment be initiated, as dictated by the patient's clinical status, until resolution of the symptoms.
Up-to-date information about the treatment of overdose can be obtained from a certified Regional Poison Center. In the event of an overdose, contact the National Capital Poison Center at 1-800-222-1222 or www.poison.org.
11. Signifor LAR Injection Description
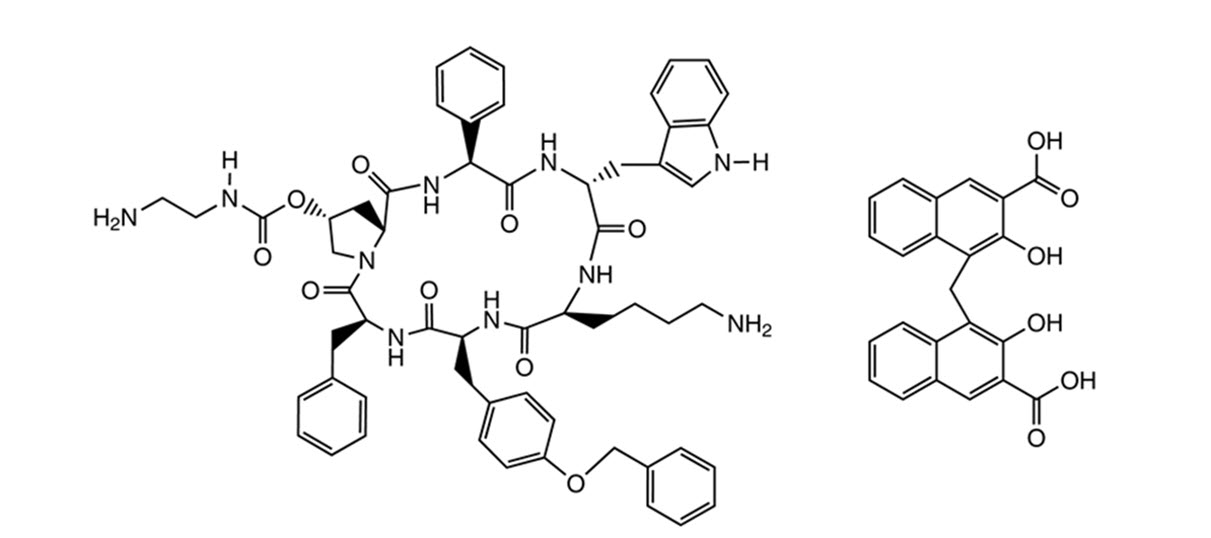
SIGNIFOR LAR (pasireotide) for injectable suspension is a long-acting release form of pasireotide pamoate, as powder to be suspended in the provided diluent immediately prior to intramuscular injection. SIGNIFOR LAR contains pasireotide, a somatostatin analog in the form of pasireotide pamoate (pamoic acid salt). Pasireotide is a cyclohexapeptide with pharmacologic properties mimicking those of the natural hormone somatostatin. Pasireotide pamoate has a chemical name of (2-Aminoethyl) carbamic acid (2R,5S,8S,11S,14R,17S,19aS)-11-(4-aminobutyl)-5-benzyl-8-(4-benzyloxybenzyl)-14-(1H-indol-3-ylmethyl)-4,7,10,13,16,19-hexaoxo-17-phenyloctadecahydro-3a,6,9,12,15,18-hexaazacyclopentacyclooctadecen-2-yl ester pamoic acid salt.
The molecular formula of pasireotide pamoate is C58H66N10O9 • C23H16O6 and the molecular weight is 1435.58 g/mol.
The structural formula is:
The drug product consists of pasireotide pamoate uniformly distributed within microspheres which are made of biodegradable copolymers of poly (D,L-lactide-co-glycolide) acids (PLGA).
SIGNIFOR LAR is available in a vial containing the sterile pasireotide pamoate, PLGA microspheres powder, 10 mg, 20 mg, 30 mg, 40 mg, and 60 mg to be reconstituted with the provided 2 mL sterile diluent.
Each vial contains:
| *corresponds to 10 mg, 20 mg, 30 mg, 40 mg, and 60 mg of pasireotide base, respectively. | |||||
| 10 mg | 20 mg | 30 mg | 40 mg | 60 mg | |
| Pasireotide pamoate | 13.71 mg* | 27.42 mg* | 41.13 mg* | 54.84 mg* | 82.26 mg* |
| Poly(D,L-lactide-co-glycolide) [50-60:40-50] | 13.15 mg | 26.29 mg | 39.44 mg | 52.58 mg | 78.87 mg |
| Poly(D,L-lactide-co-glycolide) [50:50] | 13.15 mg | 26.29 mg | 39.44 mg | 52.58 mg | 78.87 mg |
Each diluent prefilled syringe contains:
| Mannitol | 90 mg |
| Carboxymethylcellulose sodium | 14 mg |
| Poloxamer 188 | 4 mg |
| Water for injections | add to 2 mL |
12. Signifor LAR Injection - Clinical Pharmacology
12.1 Mechanism of Action
SIGNIFOR LAR is an injectable cyclohexapeptide, somatostatin analog. Pasireotide exerts its pharmacological activity via binding to somatostatin receptors (SSTR). There are 5 known human somatostatin receptor subtypes: SSTR 1, 2, 3, 4, and 5. These receptor subtypes are expressed in different tissues under normal physiological conditions. Somatostatin analogs bind to SSTRs with different potencies. Pasireotide binds with high affinity to 4 of the 5 SSTRs (see Table 4).
| Results are the mean + SEM of IC50 values expressed as nmol/l (nM). | |||||
| Compound | SSTR1 | SSTR2 | SSTR3 | SSTR4 | SSTR5 |
| Somatostatin (SRIF-14) | 0.93 ± 0.12 | 0.15 ± 0.02 | 0.56 ± 0.17 | 1.5 ± 0.4 | 0.29 ± 0.04 |
| Pasireotide | 9.3 ± 0.1 | 1.0 ± 0.1 | 1.5 ± 0.3 | > 100 | 0.16 ± 0.01 |
12.2 Pharmacodynamics
Somatostatin receptors are expressed in many tissues including neuroendocrine tumors (e.g., growth hormone or adrenocorticotropic hormone secreting pituitary adenomas).
Acromegaly
Pasireotide binds to SSTR2 and SSTR5 subtype receptors which may be relevant for inhibition of GH secretion. In vivo studies show that SIGNIFOR LAR lowers GH and IGF-1 levels in patients with acromegaly.
Cushing's Disease
Corticotroph tumor cells from Cushing's disease patients frequently over-express SSTR5 whereas the other receptor subtypes are often not expressed or are expressed at lower levels. Pasireotide binds and activates the SSTRs resulting in inhibition of ACTH secretion, which leads to decreased cortisol secretion.
Cardiac Electrophysiology
Individually corrected QT (QTcI) interval was evaluated in a randomized, blinded, crossover study in healthy subjects investigating pasireotide subcutaneous doses of 0.6 mg and 1.95 mg twice daily, respectively. The maximum mean (95% upper confidence bound) placebo-subtracted QTcI change from baseline was 12.7 (14.7) ms and 16.6 (18.6) ms, respectively. Both pasireotide doses decreased heart rate, with a maximum mean (95% lower confidence bound) placebo-subtracted change from baseline of -10.9 (-11.9) beats per minute (bpm) observed at 1.5 hours for pasireotide 0.6 mg twice daily, and -15.2 (-16.5) bpm at 0.5 hours for pasireotide 1.95 mg twice daily.
The predicted pasireotide peak concentration (25.8 ng/mL) following SIGNIFOR LAR 60-mg dose in acromegaly patients is similar to the observed peak concentration (24.3 mg/mL) of the subcutaneous SIGNIFOR 0.6 mg twice daily dose and below the observed peak concentration (80.6 ng/mL) of the subcutaneous SIGNIFOR 1.95 mg twice daily dose. The predicted pasireotide peak concentration for the SIGNIFOR LAR dose of 40 mg in Cushing's disease patients is 14 ng/mL.
12.3 Pharmacokinetics
Pasireotide for intramuscular use is formulated as microspheres for long-acting release. After a single injection, the plasma pasireotide concentration shows an initial burst release on the injection day, followed by a dip from Day 2 to Day 7, then a slow increase to the maximum concentration around Day 21, and a slow declining phase over the next weeks, concomitant with the terminal degradation phase of the polymer matrix of the dosage form.
Absorption and Distribution
No studies have been conducted to evaluate the absolute bioavailability of pasireotide in humans. Food effect is unlikely to occur since SIGNIFOR LAR is administered via a parenteral route.
In healthy volunteers, pasireotide administered as SIGNIFOR LAR is widely distributed with large apparent volume of distribution (Vz/F > 100 L). Distribution between blood and plasma is concentration-independent and shows that pasireotide is primarily located in the plasma (91%). Plasma protein binding is moderate (88%) and independent of concentration.
Pasireotide has low passive permeability and is likely to be a substrate of P-glycoprotein (P-gp), but the impact of P-gp on the ADME (absorption, distribution, metabolism, excretion) of pasireotide is expected to be low. In clinical testing in healthy volunteers, P-gp inhibition did not affect the rate or extent of pasireotide availability [see Drug Interactions (7.1)]. At therapeutic dose levels, pasireotide is not expected to be a substrate of BCRP (breast cancer resistance protein), OCT1 (organic cation transporter 1), or OATP (organic anion-transporting polypeptides) 1B1, 1B3, or 2B1.
Elimination
Metabolism and Excretion
Pasireotide was shown to be highly metabolically stable in human liver and kidney microsomes. In healthy volunteers, pasireotide in its unchanged form is the predominant form found in plasma, urine and feces. Somatropin may increase CYP450 enzymes and, therefore, suppression of GH secretion by somatostatin analogs including pasireotide may decrease the metabolic clearance of compounds metabolized by CYP450 enzymes.
Pasireotide is eliminated mainly via hepatic clearance (biliary excretion) with a small contribution of the renal route. In a human ADME study with subcutaneous SIGNIFOR with a single dose 0.6 mg, 55.9 ± 6.63% of the radioactivity dose was recovered over the first 10 days post dosing, including 48.3 ± 8.16% of the radioactivity in feces and 7.63 ± 2.03% in urine.
The apparent clearance (CL/F) of SIGNIFOR LAR in healthy volunteers is on average 4.5-8.5 L/hour.
Steady-State Pharmacokinetics
PK steady-state for SIGNIFOR LAR is achieved after 3 monthly doses. Following multiple intramuscular doses every 4 weeks (every 28 days), SIGNIFOR LAR demonstrates approximately dose-proportional PK exposures (steady-state trough; Ctrough, ss) in the dose range of 10 mg to 60 mg every 4 weeks.
Special Populations
Population PK analyses of SIGNIFOR LAR suggest that race, gender, and body weight do not have clinically relevant influence on circulating levels of pasireotide. No dose adjustment is required for demographics.
Pediatric Patients
No studies have been performed in pediatric patients [see Use in Specific Populations (8.4)].
Geriatric Patients
Age is not a significant covariate in the population PK analysis. Therefore age is not expected to significantly impact circulating levels of pasireotide.
Efficacy and safety data on patients older than 65 years are limited [see Use in Specific Populations (8.5)].
Hepatic Impairment
In a clinical study with a single subcutaneous dose of 600 µg pasireotide in subjects with impaired hepatic function (Child-Pugh A, B, and C), subjects with moderate and severe hepatic impairment (Child-Pugh B and C) showed significantly higher exposures than subjects with normal hepatic function. Upon comparing with the control group, AUCinf was increased by 12%, 56%, and 42%; and Cmax was increased by 3%, 46%, and 33% respectively, in the mild, moderate, and severe hepatic impairment groups [see Use in Specific Populations (8.6), Dosage and Administration (2.5)].
Renal Impairment
Clinical studies have not been performed in patients with renal impairment. However, renal clearance has a minor contribution to the elimination of pasireotide in humans. Renal function (creatinine clearance and estimated glomerular filtration rate) is not a covariate in the population PK analysis. Therefore, renal function is not expected to significantly impact the circulating levels of pasireotide [see Use in Specific Populations (8.7)].
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
A lifetime carcinogenicity study was conducted in rats and transgenic mice. Rats were given daily subcutaneous doses of pasireotide at 0.01 mg/kg/day, 0.05 mg/kg/day, and 0.3 mg/kg/day for 104 weeks. There were no drug-related tumors in rats at exposures up to 5-times higher than the maximum recommended clinical exposure of the pasireotide LAR 60 mg dose. Mice were given subcutaneous doses of pasireotide at 0.5 mg/kg/day, 1 mg/kg/day, and 2.5 mg/kg/day for 26 weeks and did not identify any carcinogenic potential.
Mutagenesis
Pasireotide was not genotoxic in a battery of in vitro assays (Ames mutation test in Salmonella and Escherichia coli and mutation test in human peripheral lymphocytes). Pasireotide was not genotoxic in an in vivo rat bone marrow nucleus test.
Impairment of Fertility
Subcutaneous dosing at 0.1 mg/kg/day before mating and continuing into gestation in rats at exposures less than the human clinical exposure based on body surface area comparisons across species resulted in statistically significant increased implantation loss and decreased viable fetuses, corpora lutea, and implantation sites. Abnormal cycles or acyclicity were observed at 1 mg/kg/day (4-fold higher than the maximum therapeutic exposure of pasireotide LAR based on surface area, comparisons across species).
14. Clinical Studies
14.1 Drug-Naïve Patients with Acromegaly
A multicenter, randomized, double-blind study was conducted to assess the safety and efficacy of SIGNIFOR LAR in patients with active acromegaly. A total of 358 patients naïve to drugs used to treat acromegaly were randomized in a 1:1 ratio to SIGNIFOR LAR or another somatostatin analog active comparator. Randomization was stratified based on previous pituitary surgical status (e.g., at least 1 prior pituitary surgery versus no prior pituitary surgery).
In the overall study population, 52% were female and the average age of patients was 45 years. Sixty percent of patients were Caucasian, 23% Asian, 12% Other, 3% American Indian, and 2% were Black. Forty-two percent of patients had previous pituitary surgery, and 1 patient had a history of pituitary radiation therapy. Median time between diagnosis and trial participation was 6 months. Median GH was 8.8 mcg/L (range: 0.8-200 mcg/L) and 10.1 mcg/L (range: 0.6-169.6 mcg/L) for SIGNIFOR LAR and active comparator, respectively at baseline. Median standardized IGF-1, defined as IGF-1 value divided by the ULN (i.e., fold above the ULN), was 2.9 (range: 0.9-6.9) and 2.9 (range: 0.8-7.3), for SIGNIFOR LAR and active comparator, respectively, at baseline.
The starting dose of SIGNIFOR LAR was 40 mg. Dose increase was allowed in both arms, at the discretion of investigators, after 3 and 6 months of treatment if mean GH was greater than or equal to 2.5 mcg/L and/or IGF-1 was greater than the ULN for age and sex. The maximum allowed dose for SIGNIFOR LAR was 60 mg. The maximum dose of the active comparator was not used in this trial because the trial was multi-national and the maximum dose approved in the US was not approved in all participating countries.
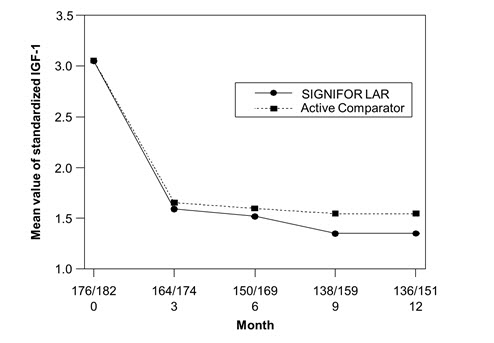
The efficacy endpoint was the proportion of patients with a mean GH level less than 2.5 mcg/L and a normal IGF-1 levels at month 12 (age- and sex-adjusted) (see Table 5, Figure 3, and Figure 4). The proportion of patients achieving this level of control was 31.3% and 19.2% for SIGNIFOR LAR and active comparator, respectively. The changes in mean GH and IGF-1 levels by study visits in subjects with a measurement at these visits (observed cases) are shown in Figures 3 and 4.
|
SIGNIFOR LAR (40-60 mg) % N = 176 |
Active Comparatorc % N = 182 |
|
| GH < 2.5 mcg/L and normalized IGF-1a | 31.3%b | 19.2% |
| GH < 2.5 mcg/L and IGF-1 ≤ ULN | 35.8% | 20.9% |
| Normalized IGF-1 | 38.6%b | 23.6% |
| GH < 2.5 mcg/L | 48.3% | 51.6% |
| aPrimary endpoint [patients with IGF-1< lower limit of normal (LLN) were not considered as "responders"]. ULN = Upper limit of normal. bp-value < 0.01 for treatment difference. cThe maximum dose approved for use in the United States was not used in this trial but the majority of patients were receiving the dose most commonly used in the United States to treat acromegaly. |
||
|
| Figure 3. Mean GH (mcg/L) Levels By Visit in Drug Naïve Patient Study* |
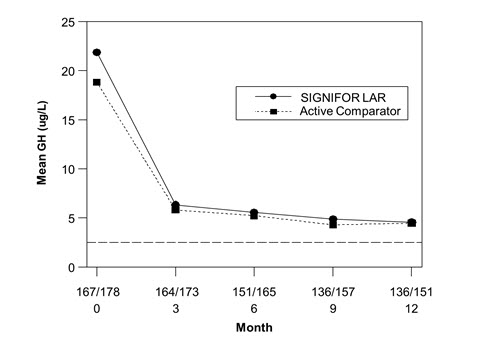 |
Biochemical control was achieved by Month 3 in 30.1% of patients in the SIGNIFOR LAR arm. Ninety-eight percent of patients treated with SIGNIFOR LAR had either a reduction or no change in tumor volume from baseline assessed by MRI at Month 12. The median (range) change in tumor volume was a reduction of 39.8% (-97.6% to 16.9%).
Additionally, ring size and acromegaly symptoms score (i.e., headache, fatigue, perspiration, paresthesia, and osteoarthralgia) were followed. At Month 12, reductions in ring size and in symptom severity scores in both treatment groups compared to baseline were noted.
14.2 Patients with Acromegaly Inadequately Controlled on Other Somatostatin Analogs
A multicenter, randomized, 3-arm study was conducted in patients with acromegaly inadequately controlled on somatostatin analogs. Patients were randomized to double-blind SIGNIFOR LAR 40 mg (n = 65) or SIGNIFOR LAR 60 mg (n = 65) or to continued open-label pretrial somatostatin analog therapies at maximal or near maximal doses (n = 68). A total of 181 patients completed the 6-month trial.
Inadequate control was defined as a GH concentration of greater than 2.5 mcg/L (i.e., mean of 5 samples over 2 hours) and a sex- and age-adjusted IGF-1 level greater than 1.3 times the upper limit of normal. Patients were required to have been treated with other somatostatin analogs for at least 6 months prior to randomization. Note that the maximum dose for one of the active comparators approved for use in the United States was not used in this multinational trial; approximately 75% of the population in the comparator group was receiving this active comparator.
In the overall study population, 56% were female and the average age of patients was 45 years. Eighty-one percent of patients were Caucasian, 7% Other, 8% Black, 2% American Indian, and 2% Asian. The percentage of patients with previous pituitary surgery in the SIGNIFOR LAR 40 mg and 60 mg arms, and in the active control arm was 77%, 63%, and 60%, respectively. Three percent of patients in the SIGNIFOR LAR groups and 7% of patients in the active control arm had prior radiation therapy. Median (range) time from diagnosis to participation in this trial was 50 (10-337) months, 55 (8-357) months, and 54 (8-357) months in the SIGNIFOR LAR 40 mg, 60 mg and the pretrial therapy arms, respectively. At baseline, median (range) GH was 7.1 (1.0-200) mcg/L, 5.3 (1.4-113.8) mcg/L and 6.1 (1.0-92.4) mcg/L in the SIGNIFOR LAR 40 mg, 60 mg and the pretrial therapy arms, respectively. Baseline median standardized IGF-1 (defined as IGF-1 value divided by the ULN) values were 2.3, 2.6 and 2.9 in the SIGNIFOR LAR 40 mg, 60 mg and the pretrial therapy arms, respectively.
The efficacy endpoint was the proportion of patients with a mean GH level less than 2.5 mcg/L and normal IGF-1 levels at week 24. The primary analysis compared SIGNIFOR LAR 60 mg and 40 mg to continued pretrial therapy (i.e., no change in treatment). The proportion of patients achieving biochemical control was 15.4% and 20.0% for SIGNIFOR LAR 40 mg and 60 mg, respectively, at 6 months.
Biochemical control was achieved by Month 3 in 15.4% and 18.5% of patients in the SIGNIFOR LAR 40 mg and 60 mg arms, respectively.
| aPrimary endpoint (patients with IGF-1 < lower limit of normal (LLN) were not considered as "responders"). bFor one of the active comparators, the maximum dose approved for use in the United States was not used in this trial but the majority of patients were receiving the dose most commonly used in the United States to treat acromegaly. |
|||
| SIGNIFOR LAR 40 mg % N = 65 | SIGNIFOR LAR 60 mg % N = 65 | Continued Pre-Trial Therapy Control Armb % N = 68 |
|
| GH < 2.5 mcg/L and normalized IGF-1a | 15.4% | 20.0% | 0% |
| Normalization of IGF-1 | 24.6% | 26.2% | 0% |
| GH < 2.5 mcg/L | 35.4% | 43.1% | 13.2% |
Eighty-one percent and 70% of patients treated with SIGNIFOR LAR 40 mg and 60 mg, respectively, had either a reduction or no change in tumor volume from baseline assessed by MRI at Month 6. The median (range) change in tumor volume was a reduction of -10.4% (-74.5% to 19.4%) and -6.3% (-66.7% to 14.5%) from baseline for SIGNIFOR LAR 40 mg and 60 mg, respectively.
14.3 Patients with Cushing's Disease
A Phase 3, randomized, double-blind, multicenter study was conducted to evaluate the safety and efficacy of two dose regimens of SIGNIFOR LAR over a 12-month treatment period in patients with persistent or recurrent Cushing's disease, or de novo patients who were not considered candidates for pituitary surgery.
The study enrolled 150 patients with a screening mean urinary free cortisol level (mUFC) ≥ 1.5 and ≤ 5 x ULN, who were randomized in a 1:1 ratio to receive a SIGNIFOR LAR starting dose of either 10 mg intramuscularly once every 28 days or 30 mg intramuscularly once every 28 days. Randomization was stratified by values of mUFC at screening (1.5 to < 2 x ULN versus 2 to 5 x ULN, respectively).
After four months of treatment, patients who had a mUFC ≤ 1.5 x ULN continued on the blinded dose to which they were randomized. Patients with a mUFC > 1.5 x ULN at Month 4 had their doses increased in a blinded manner from 10 mg to 30 mg, or from 30 mg to 40 mg, provided there were no tolerability concerns. Additional dose increases were allowed at Month 7 and Month 9 (by one dose level if the mUFC was > 1 x ULN).
Dose reduction by one dose level for tolerability was allowed in a blinded fashion for the first seven months, with a minimum dose level of 5 mg. After the first seven months, blinded down titration of more than one dose level was allowed at any month.
After twelve months of treatment (core phase), patients had the option to enter an extension to continue to receive SIGNIFOR LAR if they benefited from treatment.
The primary efficacy endpoint was the proportion of patients in each arm who were mUFC responders (mUFC ≤ ULN) after seven months of treatment, with or without up-titration at Month 4. The key secondary endpoint was the proportion of patients in each arm who were mUFC responders after seven months of treatment and who did not up-titrate the dose prior to Month 7. The pre-specified boundary of the lower limit of the 95% confidence interval (CI) for efficacy for both the primary and key secondary endpoints was 15%. Patients with missing mUFC assessment at Month 7 were classified as non-responders. Other secondary endpoints included changes from baseline in 24-hour UFC, plasma ACTH, and serum cortisol levels. All analyses were conducted based on the randomized dose groups.
Baseline demographics and disease history were well balanced between the two randomized dose groups and consistent with the epidemiology of the disease. The mean age of patients was approximately 38.5 years with a predominance of female patients (78.7%). The majority of the patients had persistent or recurrent Cushing's disease (82.0%). The median value of the baseline 24-hour mUFC for all patients was 396.9 nmol/24 hours (ULN: 166.5 nmol/24 hours). About three-quarters of all randomized patients completed seven months of treatment, and about two-thirds of all randomized patients completed twelve months of treatment.
The study met the primary efficacy objective for both dose groups. Patients were considered responders if they remained on treatment until at least Month 7 and achieved a Month 7 mUFC ≤ 1 x ULN, regardless of up-titration at Month 4. The proportion of patients with mUFC response at Month 7 was 39.2% (95% CI: 28.0, 51.2) in the 10 mg arm and 40.8% (95% CI: 29.7, 52.7) in the 30 mg arm. The responder rate at Month 12 was 35.1% (26/74) and 25.0% (19/76) in the 10 mg and 30 mg starting dose groups, respectively.
| There were 17 (23.0%) patients in 10 mg arm and 9 (11.8%) in the 30 mg arm with missing value mUFC assessments at Month 7 who were classified as non-responders. | ||
| Pasireotide LAR 10 mg Every 28 Days | Pasireotide LAR 30 mg Every 28 Days |
|
| Screening mUFC category | n/N (%) 95% CI | n/N (%) 95% CI |
| All patients | 29/74 (39.2) (28.0, 51.2) | 31/76 (40.8) (29.7, 52.7) |
| ≥ 1.5 x ULN to < 2 x ULN | 11/25 (44.0) (24.4, 65.1) | 13/25 (52.0) (31.3, 72.2) |
| ≥ 2 x ULN to ≤ 5 x ULN | 18/49 (36.7) (23.4, 51.7) | 18/51 (35.3) (22.4, 49.9) |
The study met the key secondary efficacy objective for both dose groups. At Month 4, 31/74 (41.9%) and 28/76 (36.8%) patients were up-titrated in the pasireotide LAR 10 mg and 30 mg arms, respectively. When all patients who up-titrated prior to Month 7 were counted as non-responders, Month 7 mUFC response was observed in 25.7% (95% CI: 16.2 to 37.2) and 31.6% (95% CI: 21.4 to 43.3) of patients randomized to pasireotide LAR at a starting dose of 10 mg once every 28 days and 30 mg once every 28 days, respectively.
A secondary efficacy analysis was conducted for the combined proportion of patients who attained mUFC ≤ ULN (controlled) or had at least 50% reduction in mUFC (partially controlled) in the core phase of the study. The combined rate of controlled or partially controlled responders at Month 7 constituted 44.6% and 53.9% of patients randomized to the 10 mg and 30 mg dose groups, respectively (Table 8).
| Response Category | Pasireotide LAR 10 mg Every 28 Days (N = 74) n (%) | Pasireotide LAR 30 mg Every 28 Days (N = 76) n (%) |
| Controlled (mUFC ≤ ULN) | 29 (39.2%) | 31 (40.8%) |
| Partially controlled (≥ 50% reduction in mUFC) | 4 (5.4%) | 10 (13.2%) |
| Combined | 33 (44.6%) | 41 (53.9%) |
Decreases in median mUFC levels at Month 7 compared to baseline, as measured by overall percentage of reduction, are shown in Table 9. Reductions in serum cortisol and plasma ACTH levels were also observed at Months 7 and 12 for each dose group.
| Note: Median % change from baseline in mUFC at Month 7 are calculated by imputing missing values with the worst observed % change in mUFC at Month 7 within each treatment group. | |||
| Pasireotide LAR 10 mg Every 28 Days % change | Pasireotide LAR 30 mg Every 28 Days % change |
||
| mUFC levels (nmol/24hr) Baseline | N Mean (SD) Median | 74 462.6 (256.41) 409.8 | 76 477.1 (331.75) 371.6 |
| Median change in mUFC (% from baseline) | Month 7 | -41.3% | -41.4% |
16. How is Signifor LAR Injection supplied
16.1 How Supplied
SIGNIFOR LAR for injectable suspension is supplied in a single-use kit containing the following:
- One 6-mL brownish glass vial with a grey rubber stopper of SIGNIFOR LAR containing slightly yellow to yellow powder with a flip-off cap
- One 3-mL glass barrel/grey rubber stopper prefilled syringe containing 2 mL of clear, colorless to slightly yellow/brown diluent solution for reconstitution
- One sterile 20G x 1.5" stainless steel, polypropylene safety injection needle
- One vial adapter made of polycarbonate for drug product reconstitution
SIGNIFOR LAR kits are available in the following strengths:
| SIGNIFOR LAR Kit | Final Concentration When Reconstituted (total product strength per total volume) | Final Concentration When Reconstituted (per mL) | Flip-off Cap Color | NDC |
| 10 mg | 10 mg/2 mL | 5 mg/mL | Brown | 55292-139-01 |
| 20 mg | 20 mg/2 mL | 10 mg/mL | Gray | 55292-140-01 |
| 30 mg | 30 mg/2 mL | 15 mg/mL | Lilac | 55292-141-01 |
| 40 mg | 40 mg/2 mL | 20 mg/mL | Red | 55292-142-01 |
| 60 mg | 60 mg/2 mL | 30 mg/mL | Orange | 55292-143-01 |
16.2 Storage and Handling
Store at 2°C to 8°C (36°F-46°F). Do not freeze.
SIGNIFOR LAR should be stored at refrigerated temperatures between 2°C to 8°C (36°F-46°F) until the time of use. SIGNIFOR LAR drug product kit should remain at room temperature for a minimum of 30 minutes before reconstitution, but should not exceed 24 hours at room temperature. However, after preparation of the drug suspension, it must be administered immediately.
17. Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
- Instruct patients on the importance of adhering to their return visit schedule.
- Advise patients that SIGNIFOR LAR should only be administered by a trained healthcare professional.
Hyperglycemia, Diabetes, and Ketoacidosis
- Advise patients to assess FPG and HbA1c prior to starting treatment with SIGNIFOR LAR and advise patients to consistently monitor blood glucose levels, particularly after the start of treatment or after dose changes, so that appropriate action can be taken. Advise patients to contact their healthcare provider if they experience signs or symptoms of diabetes or ketoacidosis [see Warnings and Precautions (5.1)].
Bradycardia and QT Prolongation
- Advise patients that an ECG will be taken before treatment and periodically thereafter. Advise patients with cardiac disease and with risk factors for QT prolongation and bradycardia that adjustments in cardiac medications may be made and electrolyte disturbances may require correction [see Warnings and Precautions (5.2)].
Liver Test Elevations
- Advise patients that liver function will be assessed prior to starting treatment with SIGNIFOR LAR and will be closely monitored for the first three months of treatment and thereafter as clinically indicated [see Warnings and Precautions (5.3)].
Cholelithiasis and Complications of Cholelithiasis
- Inform patients that cholelithiasis and complications of cholelithiasis have been reported with the use of SIGNIFOR LAR [see Warnings and Precautions (5.4)].
- Advise patients to contact their healthcare provider if they experience signs or symptoms of gallstones (cholelithiasis) or complications of cholelithiasis (e.g., cholecystitis or cholangitis) [see Warnings and Precautions (5.4)].
- Advise patients that the gallbladder will be monitored by ultrasound periodically [see Warnings and Precautions (5.4)].
Pituitary Hormone Deficiency(ies)
- Advise patients that monitoring of anterior pituitary function will be performed periodically [see Warnings and Precautions (5.5)].
Steatorrhea and Malabsorption of Dietary Fats
- Advise patients to contact their healthcare provider if they experience new or worsening symptoms of steatorrhea, stool discoloration, loose stools, abdominal bloating, and weight loss [see Warnings and Precautions (5.6)].
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 7/2024 | ||
| Patient Information SIGNIFOR® LAR (sig-na-for L.A.R.) (pasireotide) for injectable suspension, for intramuscular use |
|||
|
Read this Patient Information before you start receiving SIGNIFOR LAR, and each time you receive it. There may be new information. This leaflet does not take the place of talking to your healthcare provider about your medical condition or your treatment. |
|||
|
What is SIGNIFOR LAR? SIGNIFOR LAR is a prescription medicine used to treat people with:
It is not known if SIGNIFOR LAR is safe and effective for use in children. |
|||
|
What should I tell my healthcare provider before receiving SIGNIFOR LAR? Before you receive SIGNIFOR LAR, tell your healthcare provider about all of your medical conditions, including if you:
Treatment with SIGNIFOR LAR may result in improved fertility and the possibility of unplanned pregnancy in females who have acromegaly or Cushing's disease and have not gone through menopause. Talk to your healthcare provider about birth control methods that may be right for you during treatment with SIGNIFOR LAR.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Ask your healthcare provider for a list of these medicines if you are not sure. |
|||
|
How will I receive SIGNIFOR LAR?
|
|||
|
What are the possible side effects of SIGNIFOR LAR? SIGNIFOR LAR may cause serious side effects, including:
|
|||
|
o feel very thirsty o nausea |
o increased appetite with weight loss o fruity scented breath o confusion | ||
|
If you get hyperglycemia while receiving SIGNIFOR LAR, your healthcare provider may give you another medicine to lower your blood sugar. Your healthcare provider may also change your dose of SIGNIFOR LAR or advise you to stop receiving it. |
|||
|
|||
|
o weakness |
o fainting or near fainting spells | ||
|
|||
|
o weakness |
o fainting or near fainting spells | ||
|
|||
|
o sudden pain in your upper right stomach area (abdomen) |
o yellowing of your skin and whites of your eyes | ||
|
|||
|
o nausea and vomiting |
o low blood sugar levels | ||
The most common side effects of SIGNIFOR LAR include: |
|||
| • diarrhea • headache • stomach-area pain • hair loss • stuffy nose and sore throat • low blood sugar • limb swelling • loss of appetite | • nausea • increase in the level of an enzyme in your blood called creatine phosphokinase (CPK) • tiredness • stomach bloating • high blood pressure • back pain | ||
|
Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of SIGNIFOR LAR. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1–800–FDA–1088. |
|||
|
How should I store SIGNIFOR LAR?
Keep SIGNIFOR LAR and all medicines out of the reach of children. |
|||
|
General information about the safe and effective use of SIGNIFOR LAR. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use SIGNIFOR LAR for a condition for which it was not prescribed. Do not give SIGNIFOR LAR to other people, even if they have the same symptoms that you have. It may harm them. This Patient Information leaflet summarizes the most important information about SIGNIFOR LAR. If you would like more information, talk to your healthcare provider. You can ask your pharmacist or healthcare provider for information about SIGNIFOR LAR that is written for health professionals. For more information go to www.SIGNIFORLAR.com or call 1-888-575-8344. |
|||
|
What are the ingredients in SIGNIFOR LAR? Active ingredient: pasireotide pamoate Inactive ingredients: Vial: Poly(D,L-lactide-co-glycolide) Prefilled syringe: Mannitol, carboxymethylcellulose sodium, poloxamer 188, and water for injections Manufactured for: |
|||
PRINCIPAL DISPLAY PANEL - Kit Carton - NDC 55292 139 01
NDC 55292 139 01
Signifor® LAR
(pasireotide)
For Injectable Suspension
For Intramuscular Use
Should only be administered by a trained health care professional.
Remove the injection kit from the refrigerator and let the kit stand at room
temperature for a minimum of 30 minutes before reconstitution,
but do not exceed 24 hours.
RECORDATI
RARE DISEASES
GROUP
Rx only
10 mg
Each carton contains:
- One vial containing Signifor® LAR powder
- One prefilled syringe containing diluent
solution for reconstitution - One vial adapter for drug product reconstitution
- One 20G x 1.5" safety injection needle
PRINCIPAL DISPLAY PANEL - Kit Carton - NDC 55292 140 01
NDC 55292 140 01
Signifor® LAR
(pasireotide)
For Injectable Suspension
For Intramuscular Use
Should only be administered by a trained health care professional.
Remove the injection kit from the refrigerator and let the kit stand at room
temperature for a minimum of 30 minutes before reconstitution,
but do not exceed 24 hours.
RECORDATI
RARE DISEASES
GROUP
Rx only
20 mg
Each carton contains:
- One vial containing Signifor® LAR powder
- One prefilled syringe containing diluent
solution for reconstitution - One vial adapter for drug product reconstitution
- One 20G x 1.5" safety injection needle
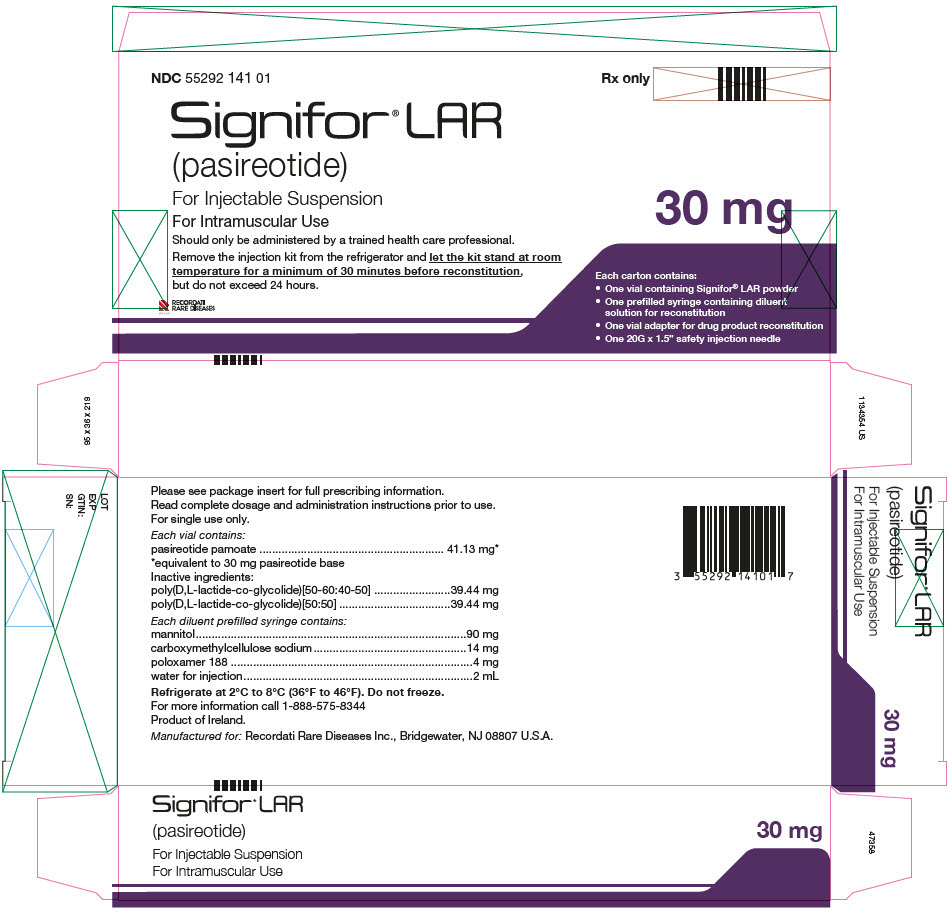
PRINCIPAL DISPLAY PANEL - Kit Carton - NDC 55292 141 01
NDC 55292 141 01
Signifor® LAR
(pasireotide)
For Injectable Suspension
For Intramuscular Use
Should only be administered by a trained health care professional.
Remove the injection kit from the refrigerator and let the kit stand at room
temperature for a minimum of 30 minutes before reconstitution,
but do not exceed 24 hours.
RECORDATI
RARE DISEASES
GROUP
Rx only
30 mg
Each carton contains:
- One vial containing Signifor® LAR powder
- One prefilled syringe containing diluent
solution for reconstitution - One vial adapter for drug product reconstitution
- One 20G x 1.5" safety injection needle
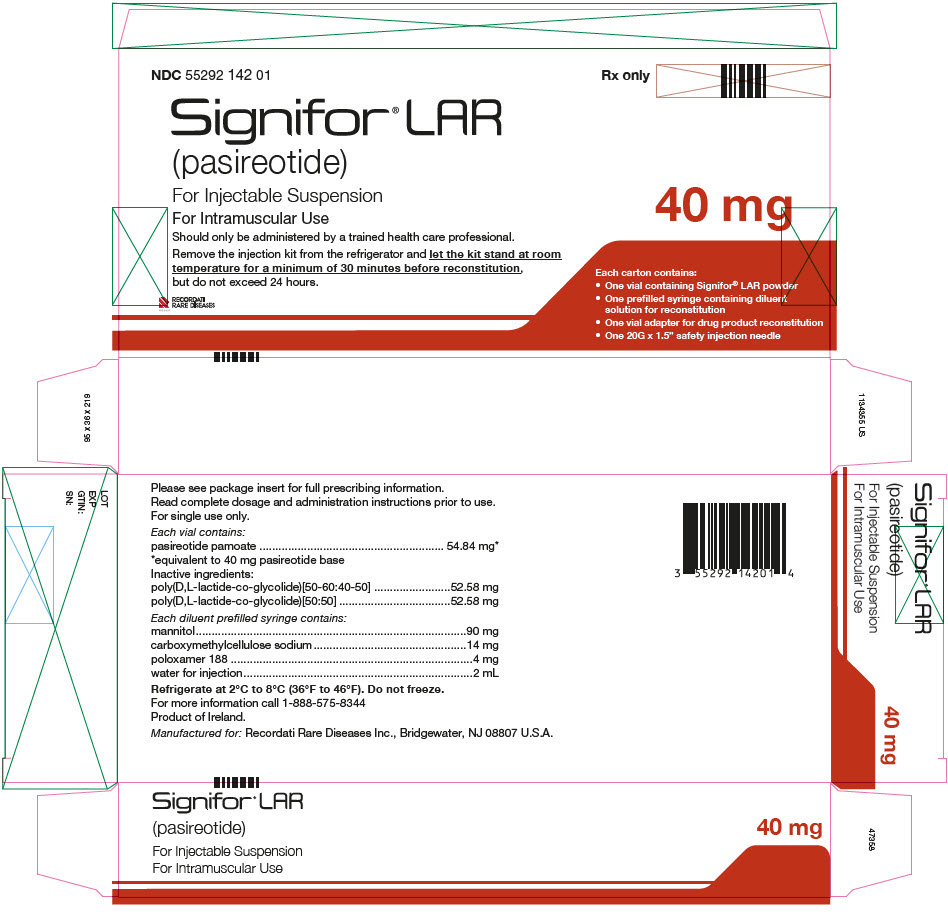
PRINCIPAL DISPLAY PANEL - Kit Carton - NDC 55292 142 01
NDC 55292 142 01
Signifor® LAR
(pasireotide)
For Injectable Suspension
For Intramuscular Use
Should only be administered by a trained health care professional.
Remove the injection kit from the refrigerator and let the kit stand at room
temperature for a minimum of 30 minutes before reconstitution,
but do not exceed 24 hours.
RECORDATI
RARE DISEASES
GROUP
Rx only
40 mg
Each carton contains:
- One vial containing Signifor® LAR powder
- One prefilled syringe containing diluent
solution for reconstitution - One vial adapter for drug product reconstitution
- One 20G x 1.5" safety injection needle
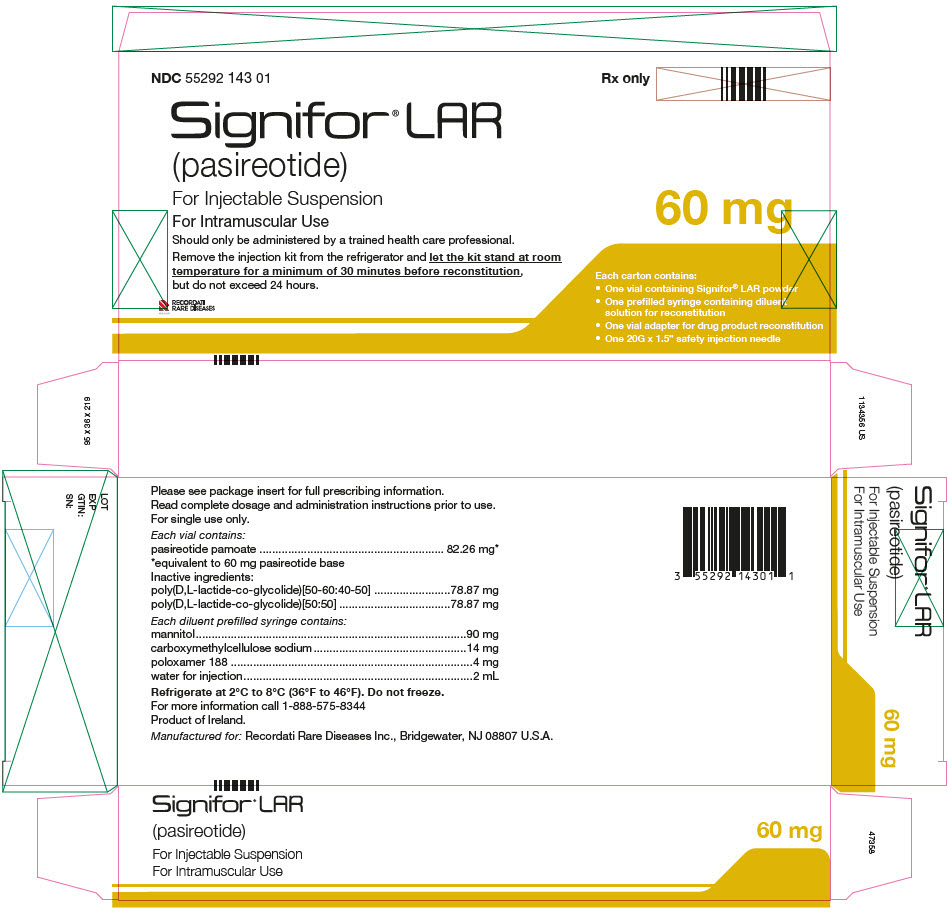
PRINCIPAL DISPLAY PANEL - Kit Carton - NDC 55292 143 01
NDC 55292 143 01
Signifor® LAR
(pasireotide)
For Injectable Suspension
For Intramuscular Use
Should only be administered by a trained health care professional.
Remove the injection kit from the refrigerator and let the kit stand at room
temperature for a minimum of 30 minutes before reconstitution,
but do not exceed 24 hours.
RECORDATI
RARE DISEASES
GROUP
Rx only
60 mg
Each carton contains:
- One vial containing Signifor® LAR powder
- One prefilled syringe containing diluent
solution for reconstitution - One vial adapter for drug product reconstitution
- One 20G x 1.5" safety injection needle
| SIGNIFOR LAR
pasireotide kit |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| SIGNIFOR LAR
pasireotide kit |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| SIGNIFOR LAR
pasireotide kit |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| SIGNIFOR LAR
pasireotide kit |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| SIGNIFOR LAR
pasireotide kit |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - Recordati Rare Diseases, Inc. (181699406) |
More about Signifor LAR (pasireotide)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Side effects
- Dosage information
- During pregnancy
- FDA approval history
- Drug class: somatostatin and somatostatin analogs
- Breastfeeding
- En español