Magnesium Sulfate: Package Insert / Prescribing Info
Package insert / product label
Dosage form: injection, solution
Drug classes: Laxatives, Minerals and electrolytes, Miscellaneous anticonvulsants
J Code (medical billing code): J3475 (500 mg, injection)
Medically reviewed by Drugs.com. Last updated on Jun 9, 2025.
On This Page
Magnesium Sulfate Description
Magnesium Sulfate Injection, USP is a sterile, nonpyrogenic, concentrated solution of magnesium sulfate heptahydrate in Water for Injection. It is administered by the intravenous (IV) or intramuscular (IM) routes as an electrolyte replenisher or anticonvulsant. Must be diluted before IV use.
Each mL contains: Magnesium sulfate heptahydrate, USP 500 mg; water for injection, q.s. Sulfuric acid and/or sodium hydroxide may have been added for pH adjustment. The pH of a 5% solution is between 5.5 and 7.0. (Osmolarity: 4,060 mOsmol/L (calc.); 2.03 mM/mL magnesium sulfate anhydrous; 4.06 mEq/mL magnesium sulfate anhydrous).
The solution contains no bacteriostat, antimicrobial agent or added buffer (except for pH adjustment) and is intended only for use as a single dose injection. When smaller doses are required the unused portion should be discarded with the entire unit.
Magnesium sulfate heptahydrate, USP is chemically designated MgSO
4•7H
2O, with a molecular weight of 246.47 and occurs as colorless crystals or white powder freely soluble in water. The chemical structure is

Magnesium Sulfate - Clinical Pharmacology
Magnesium is an important cofactor for enzymatic reactions and plays an important role in neurochemical transmission and muscular excitability.
As a nutritional adjunct in hyperalimentation, the precise mechanism of action for magnesium is uncertain. Early symptoms of hypomagnesemia (less than 1.5 mEq/L) may develop as early as three to four days or within weeks.
Predominant deficiency effects are neurological, e.g., muscle irritability, clonic twitching and tremors. Hypocalcemia and hypokalemia often follow low serum levels of magnesium. While there are large stores of magnesium present intracellularly and in the bones of adults, these stores often are not mobilized sufficiently to maintain plasma levels. Parenteral magnesium therapy repairs the plasma deficit and causes deficiency symptoms and signs to cease.
Magnesium prevents or controls convulsions by blocking neuromuscular transmission and decreasing the amount of acetylcholine liberated at the end-plate by the motor nerve impulse. Magnesium is said to have a depressant effect on the central nervous system (CNS), but it does not adversely affect the woman, fetus or neonate when used as directed in eclampsia or pre-eclampsia. Normal plasma magnesium levels range from 1.5 to 2.5 mEq/L.
As plasma magnesium rises above 4 mEq/L, the deep tendon reflexes are first decreased and then disappear as the plasma level approaches 10 mEq/L. At this level respiratory paralysis may occur. Heart block also may occur at this or lower plasma levels of magnesium. Serum magnesium concentrations in excess of 12 mEq/L may be fatal.
Magnesium acts peripherally to produce vasodilation. With low doses only flushing and sweating occur, but larger doses cause lowering of blood pressure. The central and peripheral effects of magnesium poisoning are antagonized to some extent by IV administration of calcium.
Pharmacokinetics
With IV administration the onset of anticonvulsant action is immediate and lasts about 30 minutes. Following IM administration, the onset of action occurs in about one hour and persists for three to four hours. Effective anticonvulsant serum levels range from 2.5 to 7.5 mEq/L. Magnesium is excreted solely by the kidneys at a rate proportional to the plasma concentration and glomerular filtration.
Indications and Usage for Magnesium Sulfate
Magnesium Sulfate Injection, USP is suitable for replacement therapy in magnesium deficiency, especially in acute hypomagnesemia accompanied by signs of tetany similar to those observed in hypocalcemia. In such cases, the serum magnesium level is usually below the lower limit of normal (1.5 to 2.5 mEq/L) and the serum calcium level is normal (4.3 to 5.3 mEq/L) or elevated.
In total parenteral nutrition (TPN), magnesium sulfate may be added to the nutrient admixture to correct or prevent hypomagnesemia which can arise during the course of therapy.
Magnesium sulfate injection is also indicated for the prevention and control of seizures in pre-eclampsia and eclampsia, respectively.
Contraindications
Parenteral administration of the drug is contraindicated in patients with heart block or myocardial damage.
Warnings
FETAL HARM: Continuous administration of magnesium sulfate beyond 5 to 7 days to pregnant women can lead to hypocalcemia and bone abnormalities in the developing fetus. These bone abnormalities include skeletal demineralization and osteopenia. In addition, cases of neonatal fracture have been reported. The shortest duration of treatment that can lead to fetal harm is not known. Magnesium sulfate should be used during pregnancy only if clearly needed. If magnesium sulfate is given for treatment of preterm labor, the woman should be informed that the efficacy and safety of such use have not been established and that use of magnesium sulfate beyond 5 to 7 days may cause fetal abnormalities.
ALUMINUM TOXICITY: This product contains aluminum that may be toxic. Aluminum may reach toxic levels with prolonged parenteral administration if kidney function is impaired. Premature neonates are particularly at risk because their kidneys are immature, and they require large amounts of calcium and phosphate solutions, which contain aluminum.
Research indicates that patients with impaired kidney function, including premature neonates, who receive parenteral levels of aluminum at greater than 4 to 5 mcg/kg/day accumulate aluminum at levels associated with central nervous system and bone toxicity. Tissue loading may occur at even lower rates of administration.
Parenteral use in the presence of renal insufficiency may lead to magnesium intoxication. IV use in eclampsia should be reserved for immediate control of life-threatening convulsions.
Precautions
General
Administer with caution if flushing and sweating occurs. When barbiturates, narcotics or other hypnotics (or systemic anesthetics) are to be given in conjunction with magnesium, their dosage should be adjusted with caution because of additive CNS depressant effects of magnesium.
Because magnesium is removed from the body solely by the kidneys, the drug should be used with caution in patients with renal impairment. Urine output should be maintained at a level of 100 mL or more during the four hours preceding each dose. Monitoring serum magnesium levels and the patient’s clinical status is essential to avoid the consequences of overdosage in toxemia. Clinical indications of a safe dosage regimen include the presence of the patellar reflex (knee jerk) and absence of respiratory depression (approximately 16 breaths or more/min). When repeated doses of the drug are given parenterally, knee jerk reflexes should be tested before each dose and if they are absent, no additional magnesium should be given until they return. Serum magnesium levels usually sufficient to control convulsions range from 3 to 6 mg/100 mL (2.5 to 5 mEq/L). The strength of the deep tendon reflexes begins to diminish when magnesium levels exceed 4 mEq/L. Reflexes may be absent at 10 mEq magnesium/L, where respiratory paralysis is a potential hazard. An injectable calcium salt should be immediately available to counteract the potential hazards of magnesium intoxication in eclampsia.
Magnesium sulfate injection (50%) must be diluted to a concentration of 20% or less prior to IV infusion. Rate of administration should be slow and cautious, to avoid producing hypermagnesemia. The 50% solution also should be diluted to 20% or less for IM injection in infants and children.
Laboratory Tests
Magnesium sulfate injection should not be given unless hypomagnesemia has been confirmed and the serum concentration of magnesium is monitored. The normal serum level is 1.5 to 2.5 mEq/L.
Drug Interactions
CNS Depressants–When barbiturates, narcotics or other hypnotics (or systemic anesthetics), or other CNS depressants are to be given in conjunction with magnesium, their dosage should be adjusted with caution because of additive CNS depressant effects of magnesium. CNS depression and peripheral transmission defects produced by magnesium may be antagonized by calcium.
Neuromuscular Blocking Agents–Excessive neuromuscular block has occurred in patients receiving parenteral magnesium sulfate and a neuromuscular blocking agent; these drugs should be administered concomitantly with caution.
Cardiac Glycosides–Magnesium sulfate should be administered with extreme caution in digitalized patients, because serious changes in cardiac conduction which can result in heart block may occur if administration of calcium is required to treat magnesium toxicity.
Pregnancy
Teratogenic Effects:
Pregnancy Category D(see WARNINGSand PRECAUTIONS)
See
WARNINGSand
PRECAUTIONS.
Magnesium sulfate can cause fetal abnormalities when administered beyond 5 to 7 days to pregnant women. There are retrospective epidemiological studies and case reports documenting fetal abnormalities such as hypocalcemia, skeletal demineralization, osteopenia and other skeletal abnormalities with continuous maternal administration of magnesium sulfate for more than 5 to 7 days.
1-10 Magnesium sulfate injection should be used during pregnancy only if clearly needed. If this drug is used during pregnancy, the woman should be apprised of the potential hazard to the fetus.
Labor and Delivery
Continuous administration of magnesium sulfate is an unapproved treatment for preterm labor. The safety and efficacy of such use have not been established. The administration of magnesium sulfate outside of its approved indication in pregnant women should be by trained obstetrical personnel in a hospital setting with appropriate obstetrical care facilities.
Adverse Reactions/Side Effects
The adverse effects of parenterally administered magnesium usually are the result of magnesium intoxication. These include flushing, sweating, hypotension, depressed reflexes, flaccid paralysis, hypothermia, circulatory collapse, cardiac and CNS depression proceeding to respiratory paralysis. Hypocalcemia with signs of tetany secondary to magnesium sulfate therapy for eclampsia has been reported.
Related/similar drugs
Overdosage
Magnesium intoxication is manifested by a sharp drop in blood pressure and respiratory paralysis. Disappearance of the patellar reflex is a useful clinical sign to detect the onset of magnesium intoxication. In the event of overdosage, artificial ventilation must be provided until a calcium salt can be injected IV to antagonize the effects of magnesium.
For Treatment of Overdose
Artificial respiration is often required. Intravenous calcium, 10 to 20 mL of a 5% solution (diluted if desirable with isotonic sodium chloride for injection) is used to counteract effects of hypermagnesemia. Subcutaneous physostigmine, 0.5 to 1 mg may be helpful.
Hypermagnesemia in the newborn may require resuscitation and assisted ventilation via endotracheal intubation or intermittent positive pressure ventilation as well as IV calcium.
Magnesium Sulfate Dosage and Administration
Dosage of magnesium sulfate must be carefully adjusted according to individual requirements and response, and administration of the drug should be discontinued as soon as the desired effect is obtained.
Both IV and IM administration are appropriate. IM administration of the undiluted 50% solution results in therapeutic plasma levels in 60 minutes, whereas IV doses will provide a therapeutic level almost immediately. The rate of IV injection should generally not exceed 150 mg/minute (1.5 mL of a 10% concentration or its equivalent), except in severe eclampsia with seizures (see below). Continuous maternal administration of magnesium sulfate in pregnancy beyond 5 to 7 days can cause fetal abnormalities.
Solutions for IV infusion must be diluted to a concentration of 20% or less prior to administration. The diluents commonly used are 5% Dextrose Injection, USP and 0.9% Sodium Chloride Injection, USP. Deep IM injection of the undiluted (50%) solution is appropriate for adults, but the solution should be diluted to a 20% or less concentration prior to such injection in children.
In Magnesium Deficiency
In the treatment of mild magnesium deficiency, the usual adult dose is 1 g, equivalent to 8.12 mEq of magnesium (2 mL of the 50% solution) injected IM every six hours for four doses (equivalent to a total of 32.5 mEq of magnesium per 24 hours). For severe hypomagnesemia, as much as 250 mg (approximately 2 mEq) per kg of body weight (0.5 mL of the 50% solution) may be given IM within a period of four hours if necessary. Alternatively, 5 g (approximately 40 mEq) can be added to one liter of 5% Dextrose Injection, USP or 0.9% Sodium Chloride Injection, USP for slow IV infusion over a three-hour period. In the treatment of deficiency states, caution must be observed to prevent exceeding the renal excretory capacity.
In Hyperalimentation
In TPN, maintenance requirements for magnesium are not precisely known. The maintenance dose used in adults ranges from 8 to 24 mEq (1 to 3 g) daily; for infants, the range is 2 to 10 mEq (0.25 to 1.25 g) daily.
In Pre-eclampsia or Eclampsia
In severe pre-eclampsia or eclampsia, the total initial dose is 10 to 14 g of magnesium sulfate. Intravenously, a dose of 4 to 5 g in 250 mL of 5% Dextrose Injection, USP or 0.9% Sodium Chloride Injection, USP may be infused. Simultaneously, IM doses of up to 10 g (5 g or 10 mL of the undiluted 50% solution in each buttock) are given. Alternatively, the initial IV dose of 4 g may be given by diluting the 50% solution to a 10 or 20% concentration; the diluted fluid (40 mL of a 10% solution or 20 mL of a 20% solution) may then be injected IV over a period of three to four minutes. Subsequently, 4 to 5 g (8 to 10 mL of the 50% solution) are injected IM into alternate buttocks every four hours as needed, depending on the continuing presence of the patellar reflex and adequate respiratory function. Alternatively, after the initial IV dose, some clinicians administer 1 to 2 g/hour by constant IV infusion. Therapy should continue until paroxysms cease. A serum magnesium level of 6 mg/100 mL is considered optimal for control of seizures. A total daily (24 hr) dose of 30 to 40 g should not be exceeded. In the presence of severe renal insufficiency, the maximum dosage of magnesium sulfate is 20 grams/48 hours and frequent serum magnesium concentrations must be obtained. Continuous use of magnesium sulfate in pregnancy beyond 5 to 7 days can cause fetal abnormalities.
Other uses
In counteracting the muscle-stimulating effects of barium poisoning, the usual dose of magnesium sulfate is 1 to 2 g given IV.
For controlling seizures associated with epilepsy, glomerulonephritis or hypothyroidism, the usual adult dose is 1 g administered IM or IV.
In paroxysmal atrial tachycardia, magnesium should be used only if simpler measures have failed and there is no evidence of myocardial damage. The usual dose is 3 to 4 g (30 to 40 mL of a 10% solution) administered IV over 30 seconds
with extreme caution.
For reduction of cerebral edema, 2.5 g (25 mL of a 10% solution) is given IV.
Incompatibilities
Magnesium sulfate in solution may result in a precipitate formation when mixed with solutions containing:
Alcohol (in high concentrations) Heavy metals
Alkali carbonates and bicarbonates Hydrocortisone sodium succinate
Alkali hydroxides Phosphates
Arsenates Polymyxin B sulfate
Barium Procaine hydrochloride
Calcium Salicylates
Clindamycin phosphate Strontium
Tartrates
The potential incompatibility will often be influenced by the changes in the concentration of reactants and the pH of the solutions.
It has been reported that magnesium may reduce the antibiotic activity of streptomycin, tetracycline and tobramycin when given together.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
How is Magnesium Sulfate supplied
| NDC No.
| Magnesium Sulfate Heptahydrate
| Fill
Volume in each Vial | Magnesium
mg/mL | Sulfate
mg/mL |
| 31722-394-32 (Unit of 25 vials)
| 500 mg per mL
| 2 mL
| 49.3
| 194.7
|
| 31722-394-33 (Unit of 25 vials)
| 500 mg per mL
| 10 mL
| 49.3
| 194.7
|
| 31722-394-34 (Unit of 25 vials)
| 500 mg per mL
| 20 mL
| 49.3
| 194.7
|
| 31722-394-35 (Unit of 25 vials)
| 500 mg per mL
| 50 mL
| 49.3
| 194.7
|
Do not administer unless solution is clear and seal is intact.
Preservative free.Discard unused portion.
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
The vial stoppers are not made with natural rubber latex.
References
1. Yokoyama K, Takahashi N, Yada Y. Prolonged maternal magnesium administration and bone metabolism in neonates.
Early Hum Dev.2010;86(3):187-91. Epub 2010 Mar 12.
2. Wedig KE, Kogan J, Schorry EK et al. Skeletal demineralization and fractures caused by fetal magnesium toxicity.
J Perinatol.2006; 26(6):371-4.
3. Nassar AH, Sakhel K, Maarouf H, et al. Adverse maternal and neonatal outcome of prolonged course of magnesium sulfate tocolysis.
Acta Obstet Gynecol Scan.2006;85(9):1099-103.
4. Malaeb SN, Rassi A, Haddad MC. Bone mineralization in newborns whose mothers received magnesium sulphate for tocolysis of premature labor.
Pediatr Radiol.2004;34(5):384-6. Epub 2004 Feb 18.
5. Matsuda Y, Maeda Y, Ito M, et al. Effect of magnesium sulfate treatment on neonatal bone abnormalities.
Gynecol Obstet Invest.1997;44(2):82-8.
6. Schanler RJ, Smith LG, Burns PA. Effects of long-term maternal intravenous magnesium sulfate therapy on neonatal calcium metabolism and bone mineral content.
Gynecol Obstet Invest.1997;43(4):236-41.
7. Santi MD, Henry GW, Douglas GL. Magnesium sulfate treatment of preterm labor as a cause of abnormal neonatal bone mineralization.
J Pediatr Orthop.1994;14(2):249-53.
8. Holcomb WL, Shackelford GD, Petrie RH. Magnesium tocolysis and neonatal bone abnormalities: a controlled study.
Obstet Gynecol.1991; 78(4):611-4.
9. Cumming WA, Thomas VJ. Hypermagnesemia: a cause of abnormal metaphyses in the neonate.
Am J Roentgenol.1989; 152(5):1071-2.
10. Lamm CL, Norton KL, Murphy RJ. Congenital rickets associated with magnesium sulfate infusion for tocolysis.
J Pediatr.1988; 113(6):1078-82.
11. McGuinness GA, Weinstein MM, Cruikshank DP, et al. Effects of magnesium sulfate treatment on perinatal calcium metabolism. II. Neonatal responses.
Obstet Gynecol.1980; 56(5):595-600.
12. Riaz M, Porat R, Brodsky NL, et al. The effects of maternal magnesium sulfate treatment on newborns: a prospective controlled study.
J Perinatol.1998;18(6 pt 1):449-54.

Manufactured for:
Camber Pharmaceuticals, Inc.,
Piscataway, NJ 08854.
Manufactured by:

Aspiro Pharma Limited
Survey No. 321, Biotech Park, Phase – III,
Karkapatla Village, Markook (Mandal),
Siddipet, Telangana-502281, India.
Revised: 04/2025
Revised:04/2025
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Magnesium Sulfate Injection 1 gram per 2 mL Carton Label

Magnesium Sulfate Injection 1 gram per 2 mL Container Label

Magnesium Sulfate Injection 5 gram per 10 mL Carton Label

Magnesium Sulfate Injection 5 gram per 10 mL Container Label
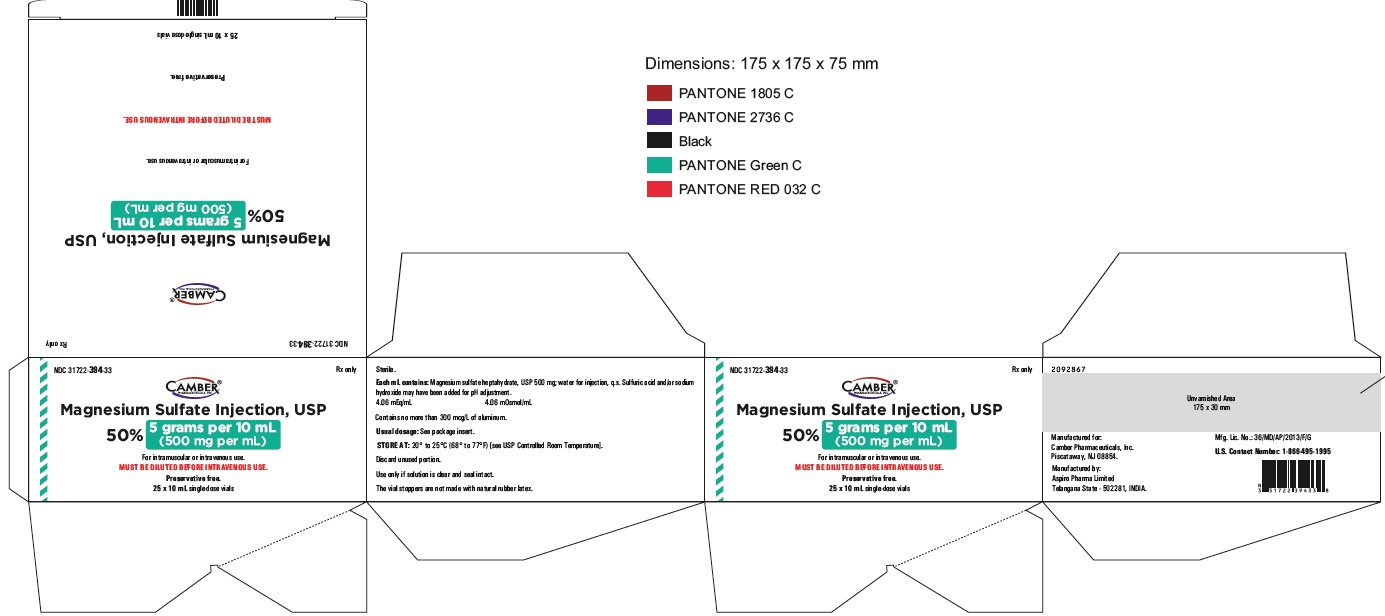
Magnesium Sulfate Injection 10 gram per 20 mL Carton Label

Magnesium Sulfate Injection 10 gram per 20 mL Container Label
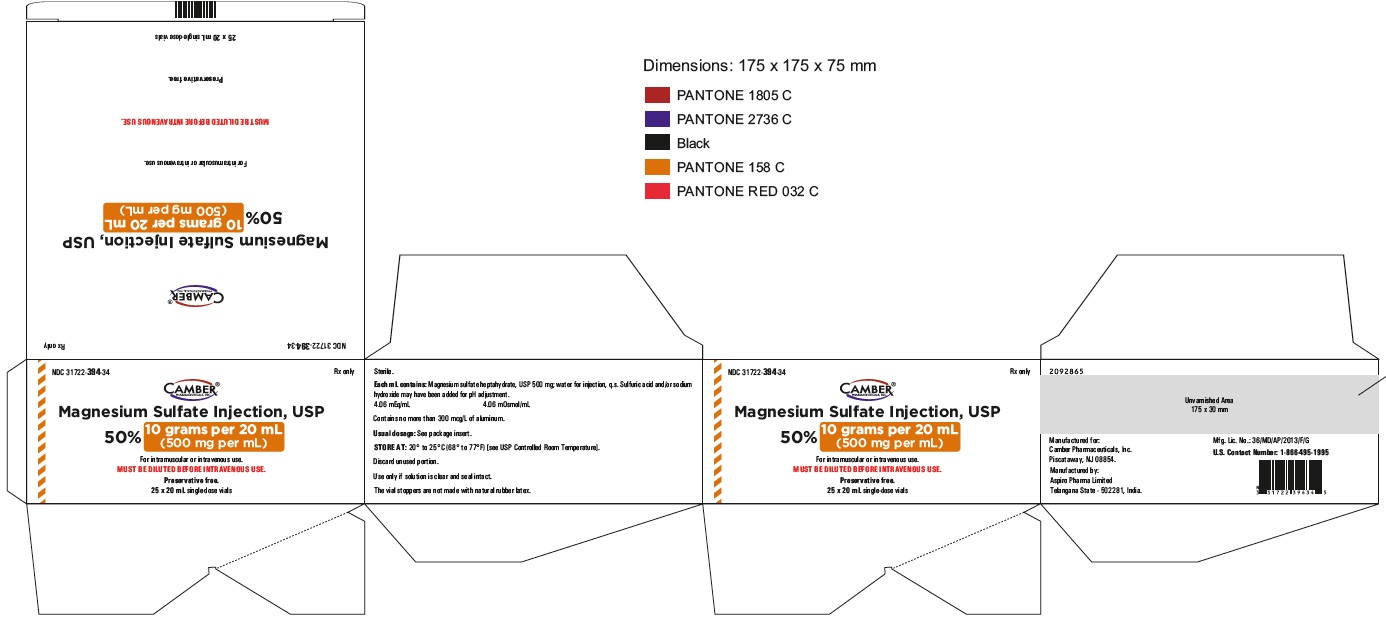
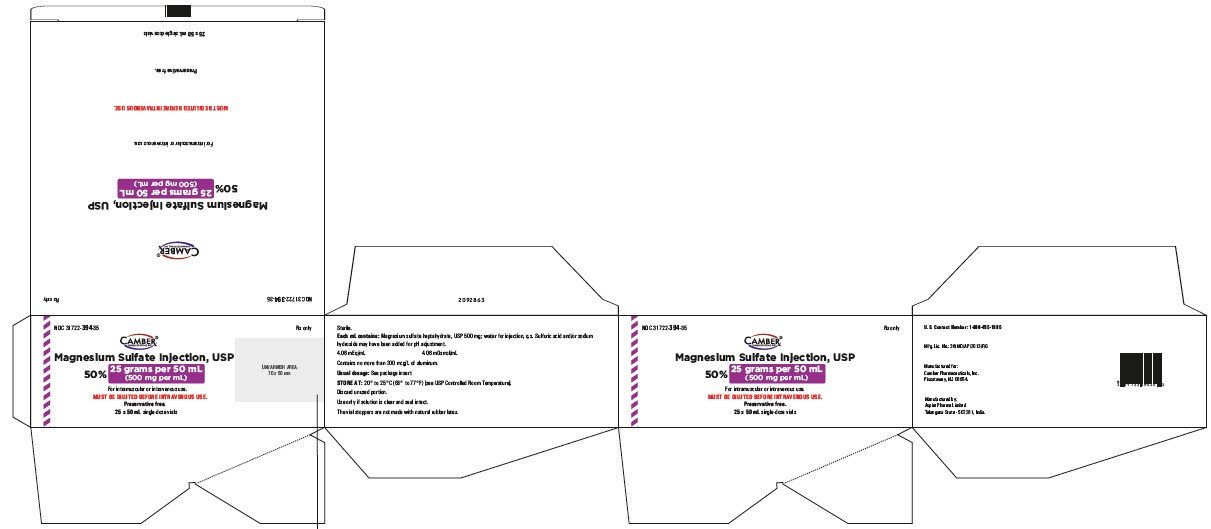
Magnesium Sulfate Injection 25 gram per 50 mL Carton Label

Magnesium Sulfate Injection 25 gram per 50 mL Container Label
| MAGNESIUM SULFATE
magnesium sulfate heptahydrate injection, solution |
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Camber Pharmaceuticals, Inc. (826774775) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Aspiro Pharma Limited | 872341023 | manufacture(31722-394) | |
Frequently asked questions
- When is the best time to take magnesium, morning or night?
- Should I take magnesium with or without food?
- Does magnesium help with restless leg syndrome (RLS)?
- Does magnesium help with headaches and migraines?
- Does magnesium help with weight loss?
More about magnesium sulfate
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (6)
- Side effects
- Dosage information
- During pregnancy
- Drug class: laxatives
- Breastfeeding
Patient resources
- Magnesium sulfate drug information
- Magnesium sulfate injection
- Magnesium Sulfate Granules (Epsom Salt)
Professional resources
- Magnesium Sulfate monograph
- Magnesium (FDA)
- Magnesium Sulfate in Dextrose Injection (FDA)
- Magnesium Water (FDA)
