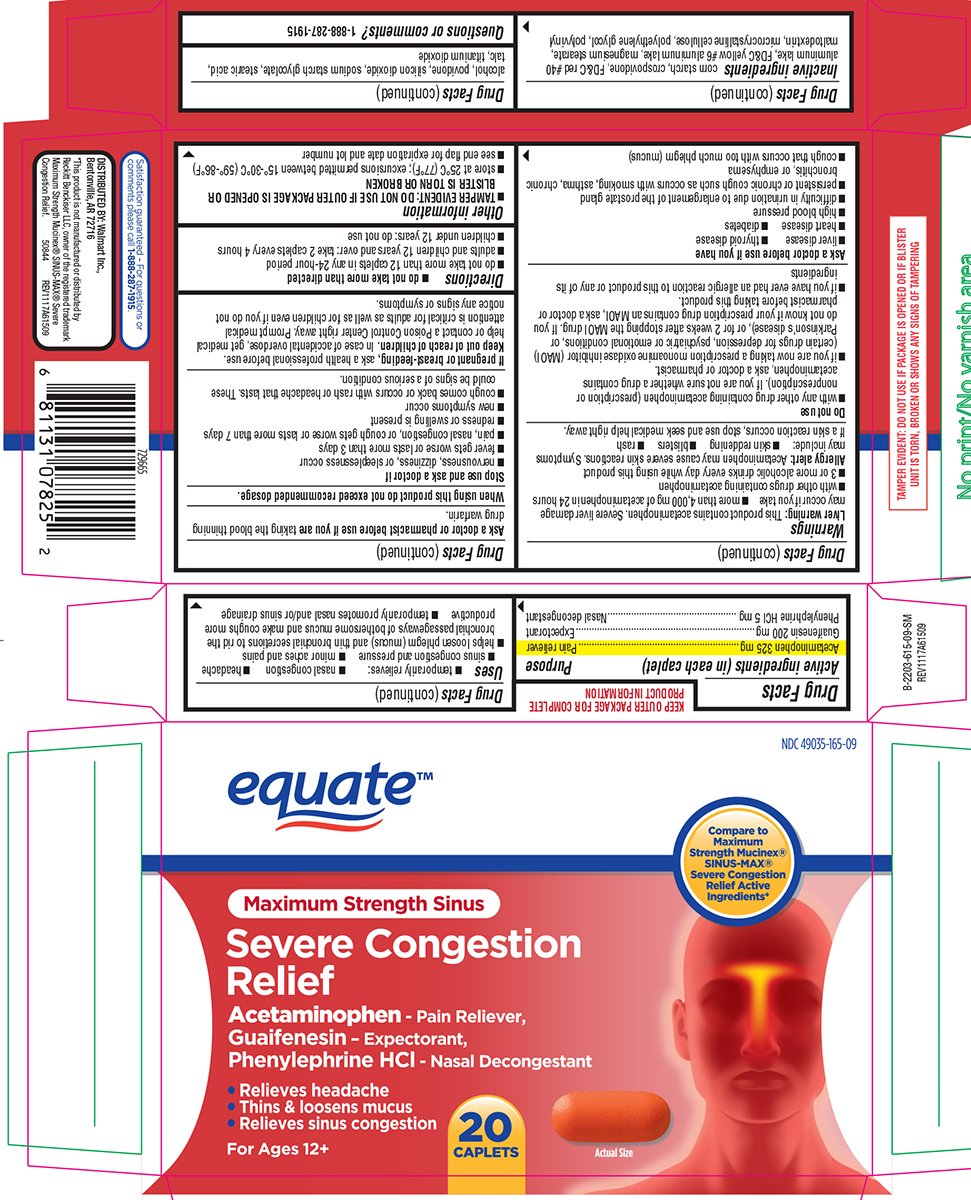
Severe Congestion Relief Maximum Strength Sinus
Dosage form: tablet, film coated
Ingredients: ACETAMINOPHEN 325mg, GUAIFENESIN 200mg, PHENYLEPHRINE HYDROCHLORIDE 5mg
Labeler: Wal-Mart Stores Inc
NDC code: 49035-165
Medically reviewed by Drugs.com. Last updated on Apr 11, 2025.
Acetaminophen 325 mg
Guaifenesin 200 mg
Phenylephrine HCl 5 mg
Pain reliever
Expectorant
Nasal decongestant
- temporarily relieves:
- nasal congestion
- headache
- sinus congestion and pressure
- minor aches and pains
- temporarily promotes nasal and/or sinus drainage
- helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
- with other drugs containing acetaminophen
- more than 4,000 mg of acetaminophen in 24 hours
- 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms
may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- if you have ever had an allergic reaction to this product or any of its ingredients
-
liver disease
-
thyroid disease
-
diabetes
-
heart disease
-
high blood pressure
-
difficulty in urination due to enlargement of the prostate gland
-
persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
-
cough that occurs with too much phlegm (mucus)
taking the blood thinning drug warfarin.
do not exceed recommended dosage.
- nervousness, dizziness, or sleeplessness occur
- fever gets worse or lasts more than 3 days
- pain, nasal congestion, or cough gets worse or lasts more than 7days
- redness or swelling is present
- new symptoms occur
- cough comes back or occurs with rash or headache that lasts. These could be signs of a serious condition.
ask a health professional before use.
In case of accidental overdose, get medical help or contact a Poison Control Center right away. Prompt medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
- do not take more than directed
- do not take more than 12 caplets in any 24-hour period
- adults and children 12 years and older: take 2 caplets every 4 hours
- children under 12 years of age: do not use
- TAMPER EVIDENT: DO NOT USE IF OUTER PACKAGE IS OPENED OR BLISTER IS TORN OR BROKEN
- see end flap for expiration date and lot number
- store at 25ºC (77ºF); excursions permitted between 15º-30ºC (59º-86ºF)
corn starch, crospovidone, FD&C red #40 aluminum lake, FD&C yellow #6 aluminum lake, magnesium stearate, maltodextrin, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, silicon dioxide, sodium starch glycolate, stearic acid, talc, titanium dioxide
1-888-287-1915
equate™ NDC 49035-165-09
Maximum Strength Sinus Compare to Maximum Strength Mucinex®SINUS-MAX®
Severe Congestion Relief Active Ingredients*
Severe Congestion Relief
Acetaminophen - Pain Reliever,
Guaifenesin - Expectorant,
Phenylephrine HCl - Nasal Decongestant
- Relieves headache
- Thins & loosens mucus
- Relieves sinus congestion
For Ages 12+ 20 CAPLETS Actual Size
TAMPER EVIDENT: DO NOT USE IF PACKAGE IS OPENED OR IF BLISTER UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING
Satisfaction guaranteed - For questions or comments please call 1-800-287-1915
DISTRIBUTED BY: Walmart Inc.,
Bentonville, AR 72716
*This product is not manufactured or distributed by Reckitt Benckiser LLC, owner of the registered trademark Maximum Strength Mucinex® SINUS-MAX® Severe Congestion Relief.
50844 REV1117A61509 729665
44-615
| SEVERE CONGESTION RELIEF
MAXIMUM STRENGTH SINUS
acetaminophen, guaifenesin, phenylephrine hcl tablet, film coated |
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
| Labeler - Wal-Mart Stores Inc (051957769) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| LNK International, Inc. | 038154464 | PACK(49035-165) | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| LNK International, Inc. | 832867894 | MANUFACTURE(49035-165) | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| LNK International, Inc. | 967626305 | PACK(49035-165) | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| LNK International, Inc. | 832867837 | PACK(49035-165) | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| LNK International, Inc. | 868734088 | PACK(49035-165) | |
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
