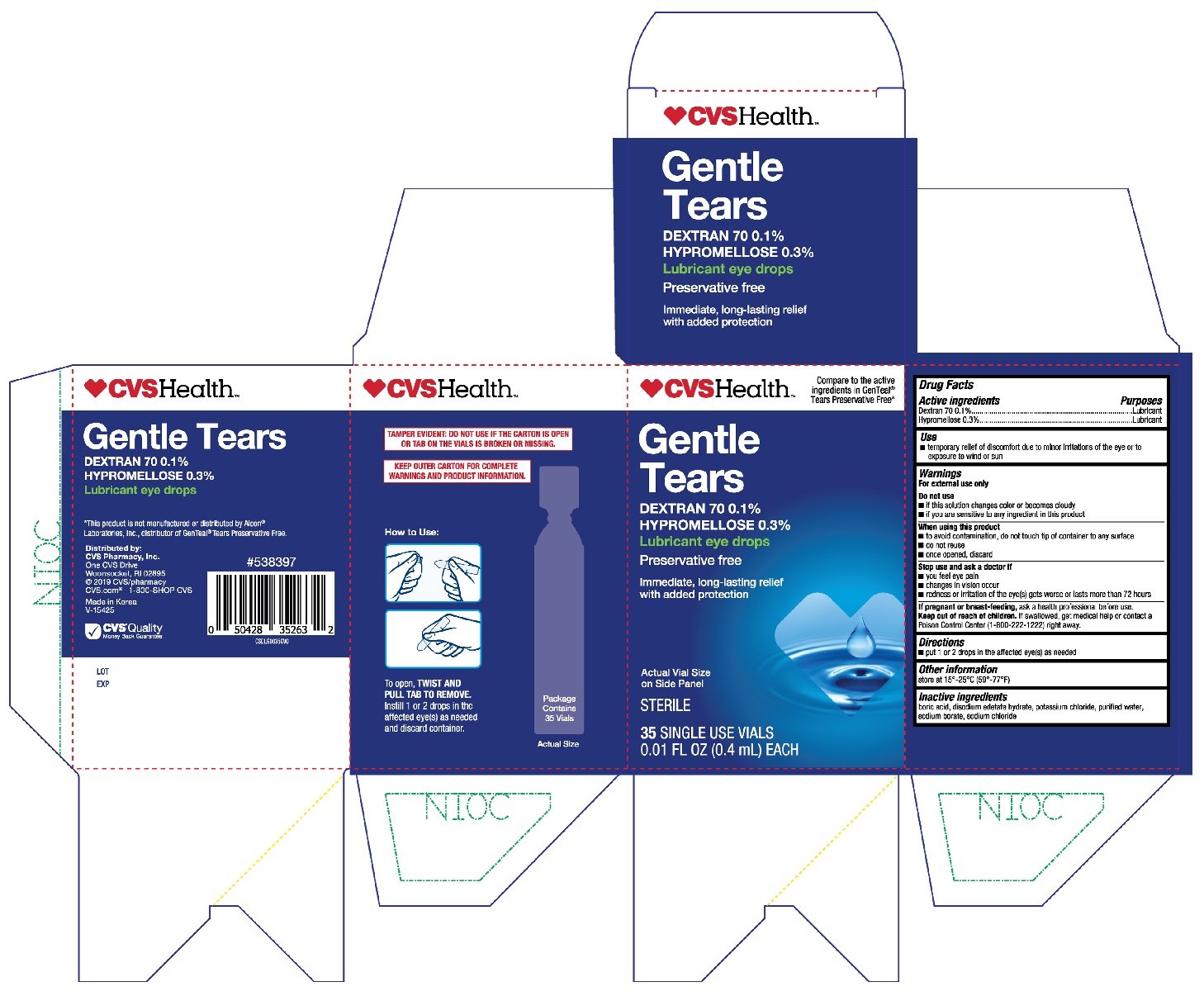
CVS Gentle Tears
Dosage form: liquid
Ingredients: DEXTRAN 70 0.1g in 100mL, HYPROMELLOSE, UNSPECIFIED 0.3g in 100mL
Labeler: CVS Pharmacy, Inc.
NDC code: 69842-474
Medically reviewed by Drugs.com. Last updated on Dec 23, 2024.
Active ingredients
Dextran 70 0.1%
Hypromellose 0.3%
Purposes
Lubricant
Lubricant
Use
- temporary relief of discomfort due to minor irritations of the eye or to exposure to wind or sun
Warnings
For external use only
Do not use
- if this solution changes color or becomes cloudy
- if you are sensitive to any ingredient in this product
When using this product
- to avoid contamination, do not touch tip of container to any surface
- do not reuse
- once opened, discard
Stop use and ask a doctor if
- you feel eye pain
- changes in vision occur
- redness or irritation of the eye(s) gets worse or lasts more than 72 hours
If pregnant or breast-feeding,
ask a health professional before use.
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Directions
- put 1 or 2 drops in the affected eye(s) as needed
Other information
store at 15°-25°C (59°-77°F)
Inactive ingredients
boric acid, disodium edetate hydrate, potassium chloride, purified water, sodium borate, sodium chloride
CVS Gentle Tears 35ct
| CVS GENTLE TEARS
dextran 70, hypromellose liquid |
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
| Labeler - CVS Pharmacy, Inc. (062312574) |
| Registrant - KC Pharmaceuticals, Inc. (174450460) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| KC Pharmaceuticals, Inc. | 174450460 | pack(69842-474), label(69842-474) | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Unimed | 689852052 | manufacture(69842-474) | |
Document Id: 9b1ea0eb-c350-6603-e053-2995a90a6a95
Set id: 9b1ea0eb-c34f-6603-e053-2995a90a6a95
Version: 1
CVS Pharmacy, Inc.
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
