Novoeight Dosage
Generic name: ANTIHEMOPHILIC FACTOR, HUMAN RECOMBINANT 62.5[iU] in 1mL;
Dosage form: injection
Drug class: Miscellaneous coagulation modifiers
Medically reviewed by Drugs.com. Last updated on Jul 21, 2025.
For intravenous injection after reconstitution only.
Dose
- •
- Dosage and duration of treatment depend on the severity of the factor VIII deficiency, on the location and extent of bleeding, and the patient’s clinical condition. Careful monitoring of replacement therapy is necessary in cases of major surgery or life-threatening bleeding episodes.
- •
- Each vial of Novoeight contains the labeled amount of recombinant factor VIII in international units (IU). One IU of factor VIII activity corresponds to the quantity of factor VIII in one milliliter of normal human plasma. The calculation of the required dosage of factor VIII is based on the empirical finding that one IU of factor VIII per kg body weight raises the plasma factor VIII activity by two IU/dL. This relationship causes a factor of 0.5 to be present in the dose calculation formula shown below.
- •
- The required dosage can be determined using the following formula:
- Dosage (IU) = Body Weight (kg) × Desired Factor VIII Increase (IU/dL or % normal) × 0.5
The final dose calculated is expressed as IU
- •
- Base the dose and frequency of Novoeight on the individual clinical response. Patients may vary in their pharmacokinetic and clinical responses.
On-demand Treatment and Control of Bleeding Episodes
A guide for dosing Novoeight for on-demand treatment and control of bleeding episodes is provided in Table 1. Dose to maintain a plasma factor VIII activity level at or above the plasma levels (in % of normal or in IU/dL) outlined in Table 1.
Table 1: Dosing for On-demand Treatment and Control of Bleeding Episodes
|
Type of Bleeding Episodes |
Factor VIII Level Required (IU/dL or % of normal) |
Frequency of Doses (hours) |
Duration of Therapy (days) |
|
Minor Early hemarthrosis, minor muscle or oral bleeding |
20-40 |
12-24 |
At least 1 day until bleeding resolution is achieved |
|
Moderate Muscle bleeding, bleeding into the oral cavity or mild head trauma |
30-60 |
12-24 |
Until pain and acute disability are resolved (approximately 3-4 days) |
|
Major Life or limb threatening hemorrhage, Gastrointestinal bleeding, intracranial, intra-abdominal or intrathoracic bleeding, fractures |
60-100 |
8-24 |
Until resolution of bleed (approximately 7-10 days) |
Perioperative Management
A guide for dosing Novoeight during surgery (perioperative management) is provided in Table 2. Consider maintaining a plasma factor VIII activity level at or above the plasma levels (in % of normal or in IU/dL) outlined in Table 2.
Table 2: Dosing for Perioperative Management
|
Type of Surgery |
Factor VIII Level Required (IU/dL or % of normal) |
Frequency of Doses (hours) |
Duration of Therapy (days) |
|
Minor Including tooth extraction |
30-60 |
24 |
At least 1 day until healing is achieved |
|
Major Intracranial, intra-abdominal, intrathoracic, or joint replacement surgery |
80-100 (pre-and post-operative) |
8-24 |
Until adequate wound healing, then continue therapy for at least 7 days to maintain a factor VIII activity of 30% to 60% (IU/dL) |
Routine Prophylaxis
A guide for dosing Novoeight for routine prophylaxis is included below in Table 3.
Table 3: Dosing for Routine Prophylaxis
|
Patient Population |
Factor VIII Dose Required (IU/kg) |
Frequency of Doses |
|
Adults and adolescents (≥ 12 years) |
20-50 |
3 times weekly |
|
20-40 |
Every other day |
|
|
Children (<12 years) |
25-60 |
3 times weekly |
|
25-50 |
Every other day |
Preparation and Reconstitution
- •
- Always wash hands and ensure that the area is clean before performing the procedures.
- •
- Use aseptic technique during the reconstitution procedures.
- •
- If the dose requires more than one vial of Novoeight per injection, reconstitute each vial according to the following instructions:
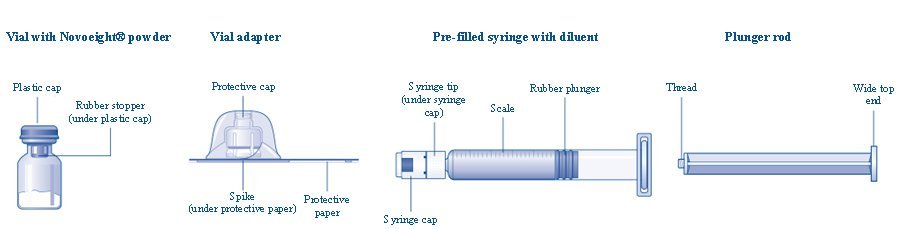
Overview of Novoeight Package
Reconstitution
- 1.
- Bring the Novoeight vial and the pre-filled diluent syringe to room temperature.
- 2.
- Remove the plastic cap from the Novoeight vial.
- 3.
- Wipe the rubber stopper on the vial with a sterile alcohol swab and allow it to dry prior to use.
- 4.
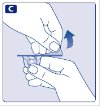
- Remove the protective paper from the vial adapter. Do not remove the vial adapter from the protective cap.
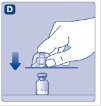
- 5.
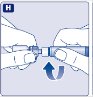
- Place the vial on a flat and solid surface. While holding the protective cap, place the vial adapter over the Novoeight vial and press down firmly on the protective cap until the vial adapter spike penetrates the rubber stopper.
- 6.
- Carefully remove the protective cap from the vial adapter.
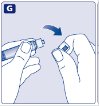
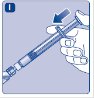
- 7.
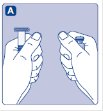
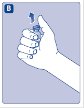
- Grasp the plunger rod as shown in the diagram. Attach the plunger rod to the syringe by holding the plunger rod by the wide top end. Turn the plunger rod clockwise into the rubber plunger inside the pre-filled diluent syringe until resistance is felt.
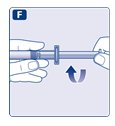
- 8.
- Break off the syringe cap from the pre-filled diluent syringe by snapping the perforation of the cap.
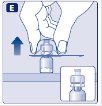
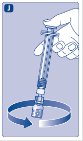
- 9.
- Connect the pre-filled diluent syringe to the vial adapter by turning it clockwise until it is secured.
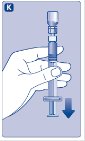
- 10.
- Push the plunger rod to slowly inject all the diluent into the vial.
- 11.
- Without removing the syringe, gently swirl the Novoeight vial until all of the powder is dissolved.
- 12.
- Use the Novoeight solution immediately. If not, store the solution in the vial with the vial adapter and the syringe attached. Use Novoeight within 4 hours after reconstitution when stored at <86°F (30°C) or within 2 hours when stored between 86°F (30°C) to 104°F (40°C).
Administration
For intravenous injection only.
- •
- Inspect the reconstituted Novoeight solution visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not use if particulate matter or discoloration is observed.
- •
- Do not administer Novoeight in the same tubing or container with other medicinal products.
- 1.
- Invert the Novoeight vial and slowly draw the solution into the syringe.
- 2.
- Detach the syringe from the vial adapter by turning the syringe counterclockwise.
- 3.
- Attach the syringe to the luer end of an infusion needle set.
- 4.
- Inject the reconstituted Novoeight intravenously slowly over 2 to 5 minutes.
- 5.
- After injection, safely dispose of the syringe with the infusion set, the vial with the vial adapter, any unused Novoeight and other waste materials. Accidental needle stick with a needle contaminated with blood can transmit infectious viruses including HIV (AIDS) and hepatitis. Obtain immediate medical attention if injury occurs. Place needles in a sharps container after single-use.
Caution:
The pre-filled diluent syringe is made of glass with an internal tip diameter of 0.037 inches, and is compatible with a standard Luer-lock connector.
Some needleless connectors for intravenous catheters are incompatible with the glass diluent syringes (for example, certain connectors with an internal spike, such as Clave® /MicroClave®, InVision-Plus®, InVision-Plus CS®, Invision-Plus Junior®, Bionector®), and their use can damage the connector and affect administration. To administer Novoeight through incompatible needleless connectors, withdraw the reconstituted product into a standard 10 mL sterile Luer-lock plastic syringe.
More about NovoEight (antihemophilic factor)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Side effects
- During pregnancy
- FDA approval history
- Drug class: miscellaneous coagulation modifiers
- Breastfeeding
- En español
Patient resources
Other brands
Advate, Altuviiio, Eloctate, Esperoct, ... +14 more
Professional resources
- Novoeight prescribing information
- Antihemophilic Factor (recombinant), PEGylated-aucl (AHFS Monograph)
Other brands
Advate, Altuviiio, Eloctate, Esperoct, ... +12 more
Related treatment guides
See also:
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.