Prenate DHA: Package Insert / Prescribing Info
Package insert / product label
Generic name: ascorbic acid, cholecalciferol, .alpha.-tocopherol, dl-, folic acid, pyridoxine hydrochloride, cyanocobalamin, calcium formate, ferrous asparto glycinate, magnesium oxide and doconexent
Dosage form: capsule, gelatin coated
Drug classes: Iron products, Vitamin and mineral combinations
Medically reviewed by Drugs.com. Last updated on Jan 24, 2024.
On This Page
WARNING: Accidental overdose of iron-containing products is a leading cause of fatal poisoning in children under 6. Keep this product out of reach of children. In case of accidental overdose, call a doctor or poison control center immediately.
DESCRIPTION: PRENATE DHA ® is a prescription prenatal/postnatal multivitamin/mineral/essential fatty acid softgel. Each softgel is blue in color, opaque, and imprinted with “DHA” on one side and blank on the other.
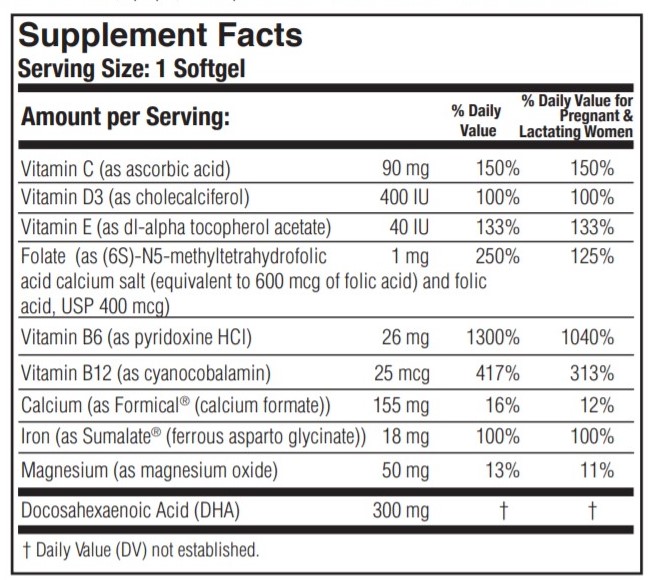
PRENATE DHA ® contains fish oil and soy.
OTHER INGREDIENTS: Gelatin capsule (FD&C Blue #1, FD&C Red #3, gelatin, glycerin, purified water, sorbitol and titanium dioxide), soybean oil, soy lecithin, and white beeswax.
INDICATIONS: PRENATE DHA ® is a multivitamin/multimineral fatty acid dietary supplement indicated for use in improving the nutritional status of women throughout pregnancy and in the postnatal period for both lactating and nonlactating mothers. PRENATE DHA ® can also be beneficial in improving the nutritional status of women prior to conception.
CONTRAINDICATIONS: PRENATE DHA ® is contraindicated in patients with a known hypersensitivity to any of the ingredients.
WARNING: Ingestion of more than 3 grams of omega-3 fatty acids (such as DHA) per day has been shown to have potential antithrombotic effects, including an increased bleeding time and International Normalized Ratio (INR). Administration of omega-3 fatty acids should be avoided in patients taking anticoagulants and in those known to have an inherited or acquired predisposition to bleeding.
PRECAUTIONS: Folic acid alone is improper therapy in the treatment of pernicious anemia and other megaloblastic anemias where Vitamin B 12 is deficient. Folic acid in doses above 1.0 mg daily may obscure pernicious anemia in that hematologic remission can occur while neurological manifestations progress.
ADVERSE REACTIONS: Allergic sensitization has been reported following both oral and parenteral administration of folic acid.
DOSAGE AND ADMINISTRATION: Before, during and/or after pregnancy, one softgel daily or as directed by a physician.
HOW SUPPLIED: Bottles of 30 softgels (75854-312-30).
The listed product number is not a National Drug Code. Instead, Avion Pharmaceuticals has assigned a product code formatted according to standard industry practice to meet the formatting requirements of pharmacy and healthcare insurance computer systems.
Store at 20° - 25°C (68° - 77°F); excursions permitted to 15° - 30°C (59° - 86°F) [See USP Controlled Room Temperature.]
Formical ® is a registered trademark of Nephro-Tech 1, LLC, covered by one or more claims of U.S. Patent No. 6,528,542.
Sumalate ® is a registered trademark of Albion Laboratories, Inc., covered by one or more claims of U.S. Patent Nos. 5,516,925, 6,716,814, 8,007,846, and 8,425,956.
PRENATE ® is a registered trademark of Avion Pharmaceuticals, LLC. Under license from Avion Pharmaceuticals, LLC.
All rights reserved.
Rev. 0519-02
| PRENATE DHA
ascorbic acid, cholecalciferol, .alpha.-tocopherol, dl-, folic acid, pyridoxine hydrochloride, cyanocobalamin, calcium formate, ferrous asparto glycinate, magnesium oxide and doconexent capsule, gelatin coated |
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
| Labeler - Avion Pharmaceuticals, LLC (040348516) |
| Registrant - Avion Pharmaceuticals, LLC (965450542) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Avion Pharmaceuticals, LLC | 040348516 | manufacture(75854-312) | |
Related/similar drugs
More about Prenate DHA (multivitamin, prenatal)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Drug images
- Side effects
- Dosage information
- Drug class: iron products
Patient resources
Professional resources
Other brands
Prenatal 19, M-Natal Plus, PreNexa, CitraNatal Assure, ... +35 more


