Prenate Pixie: Package Insert / Prescribing Info
Package insert / product label
Generic name: prenatal vitamin
Dosage form: softgel
Medically reviewed by Drugs.com. Last updated on May 19, 2025.
On This Page
WARNING:Ingestion of more than 3 grams of omega-3 fatty acids (such as DHA) per day has been shown to have potential antithrombotic effects, including an increased bleeding time and International Normalized Ratio (INR). Administration of omega-3 fatty acids should be avoided in patients taking anticoagulants and in those known to have an inherited or acquired predisposition to bleeding.
Rx Only Dietary Supplement
DESCRIPTION:PRENATE PIXIE ®is a prescription prenatal/postnatal multivitamin/mineral/essential fatty acid softgel. Each softgel is purple in color, opaque, and imprinted with “PIX”. Prenate Pixie ®contains no ingredient made from gluten-containing grain (wheat, barley, or rye)
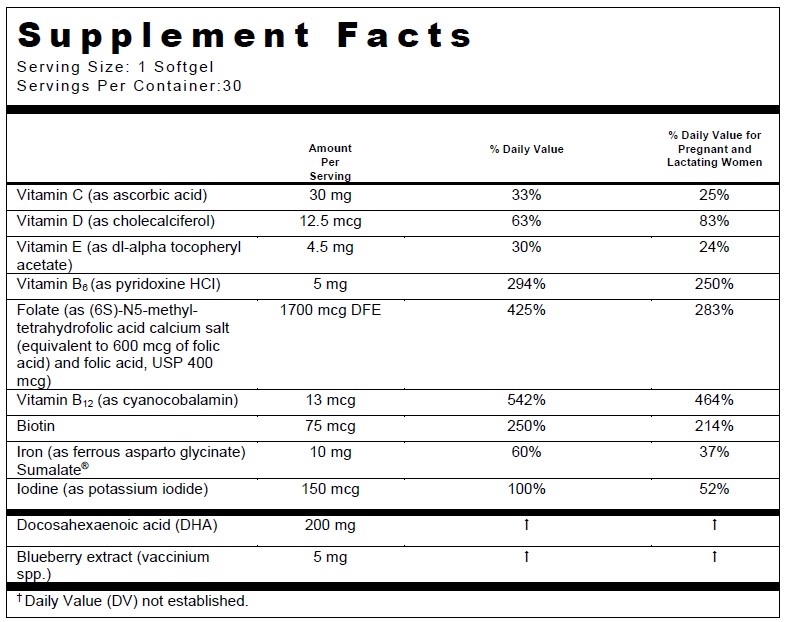
INDICATIONS:PRENATE PIXIE ®is a multivitamin/multimineral fatty acid dietary supplement indicated for use in improving the nutritional status of women throughout pregnancy and in the postnatal period for both lactating and nonlactating mothers.
CONTRAINDICATIONS:PRENATE PIXIE ®is contraindicated in patients with a known hypersensitivity to any of the ingredients.
PRECAUTIONS:Folic acid alone is improper therapy in the treatment of pernicious anemia and other megaloblastic anemias where Vitamin B 12is deficient. Folic acid in doses above 1.0 mg daily may obscure pernicious anemia in that hematologic remission can occur while neurological manifestations progress.
Biotin levels higher than the recommended daily allowance may cause interference with some laboratory tests, including cardiovascular diagnostic test (e.g. troponin) and hormone tests, and may lead to incorrect test results. Tell your healthcare provider about all prescription and over-the-counter medicines, vitamins, and dietary supplements that you take, including biotin.
WARNING:Accidental overdose of iron-containing products is a leading cause of fatal poisoning in children under 6. KEEP THIS PRODUCT OUT OF REACH OF CHILDREN. In case of accidental overdose, call a doctor or poison control center immediately.
ADVERSE REACTIONS:Allergic sensitization has been reported following both oral and parenteral administration of folic acid.
DOSAGE AND ADMINISTRATION:Before, during and/or after pregnancy, one softgel daily or as directed by a physician.
HOW SUPPLIED:Bottles of 30 softgels (75854-0316-30). The listed product number is not a National Drug Code. Instead, Avion Pharmaceuticals has assigned a product code formatted according to standard industry practice to meet the formatting requirements of pharmacy and healthcare insurance computer systems.
STORAGE:Store at 20° - 25°C (68° - 77°F); excursions permitted to 15° - 30°C (59° - 86°F) [See USP Controlled Room Temperature.]
THESE STATEMENTS HAVE NOT BEEN EVALUATED BY THE FOOD AND DRUG ADMINISTRATION. THIS PRODUCT IS NOT INTENDED TO DIAGNOSE, TREAT, CURE OR PREVENT ANY DISEASE.
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
Sumalate® is a registered trademark of Albion Laboratories, Inc., covered by one or more claims of U.S. Patent Nos. 5,516,925, 6,716,814, 8,007,846, and 8,425,956.
MANUFACTURED FOR:
Avion Pharmaceuticals, LLC
Alpharetta, Georgia 30005
1-888-61-AVION
L-0244 Rev. 0325-03
| PRENATE PIXIE
ascorbic acid, biotin, cholecalciferol, cyanocobalamin, levomefolate calcium, folic acid, ferrous asparto glycinate, potassium iodide, pyridoxine hydrochloride, .alpha.-tocopherol acetate, dl-, docosahexaenoic acid, and blueberry tablet, film coated |
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
| Labeler - Avion Pharmaceuticals, LLC (040348516) |
Related/similar drugs
More about Prenate Pixie (multivitamin, prenatal)
Patient resources
Professional resources
Other brands
Prenatal 19, M-Natal Plus, PreNexa, CitraNatal Assure, ... +35 more

