Khapzory Injection: Package Insert / Prescribing Info
Package insert / product label
Generic name: levoleucovorin
Dosage form: injection, powder, lyophilized, for solution
Drug class: Antidotes
J Code (medical billing code): J0642 (0.5 mg, injection)
Medically reviewed by Drugs.com. Last updated on Dec 15, 2024.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Drug Interactions
- Use In Specific Populations
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
Highlights of Prescribing Information
KHAPZORY (levoleucovorin) for injection, for intravenous use
Initial U.S. Approval: 1952 (d,l-leucovorin)
Indications and Usage for Khapzory Injection
KHAPZORY is a folate analog indicated for:
- Rescue after high-dose methotrexate therapy in adults and pediatric patients with osteosarcoma. (1)
- Diminishing the toxicity associated with overdosage of folic acid antagonists or impaired methotrexate elimination in adult and pediatric patients. (1)
- Treatment of adults with metastatic colorectal cancer in combination with fluorouracil. (1)
Limitations of Use
KHAPZORY is not indicated for pernicious anemia and megaloblastic anemia secondary to lack of vitamin B12 because of the risk of progression of neurologic manifestations despite hematologic remission.(1)
Khapzory Injection Dosage and Administration
For intravenous administration only. Do not administer intrathecally. (2.1)
Rescue After High-Dose Methotrexate Therapy
- Rescue recommendations are based on a methotrexate dose of 12 grams/ m2 administered by intravenous infusion over 4 hours. Initiate rescue at a dose of 7.5 mg (approximately 5 mg/m2) every 6 hours, 24 hours after the beginning of the methotrexate infusion. (2.2)
- Continue until the methotrexate level is below 5 x 10-8 M (0.05 micromolar). Adjust dose if necessary, based on methotrexate elimination; refer to Full Prescribing Information. (2.2)
Overdosage of Folic Acid Antagonists or Impaired Methotrexate Elimination
- Start as soon as possible after methotrexate overdosage or within 24 hours of delayed methotrexate elimination. (2.3)
- Administer KHAPZORY 7.5 mg (approximately 5 mg/m2) intravenously every 6 hours until methotrexate level is less than 5 x 10-8 M (0.05 micromolar). (2.3)
Metastatic Colorectal Cancer in Combination with Fluorouracil
- The following regimens have been used for the treatment of colorectal cancer.
Dosage Forms and Strengths
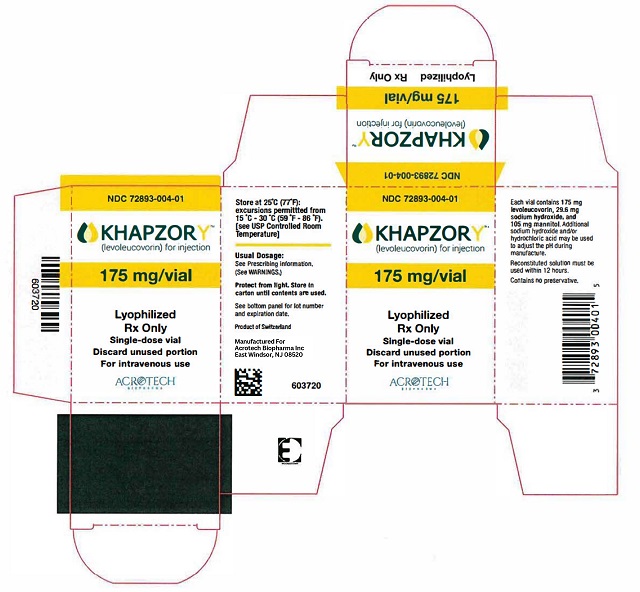
For Injection: 175 mg of levoleucovorin lyophilized powder in a single-dose vial for reconstitution. (3)
Contraindications
Patients who have had severe hypersensitivity reactions to leucovorin products, folic acid, or folinic acid. (4)
Warnings and Precautions
- Increased Gastrointestinal Toxicities with Fluorouracil: Do not initiate or continue therapy with KHAPZORY and fluorouracil in patients with symptoms of gastrointestinal toxicity until symptoms have resolved. Monitor patients with diarrhea until it has resolved as rapid deterioration leading to death can occur. (5.1,7)
-
Drug Interaction with Trimethoprim-Sulfamethoxazole: Increased rates of treatment failure and morbidity with concomitant use of d,l-leucovorin with trimethoprim-sulfamethoxazole for Pneumocystis jiroveci pneumonia in patients with HIV. (5.2)
Adverse Reactions/Side Effects
- The most common adverse reactions (≥ 20%) in patients receiving high-dose methotrexate therapy with levoleucovorin rescue are stomatitis and vomiting. (6.1)
- The most common adverse reactions (>50%) in patients receiving levoleucovorin in combination with fluorouracil for metastatic colorectal cancer are stomatitis, diarrhea, and nausea. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Acrotech Biopharma Inc at 1-888-292-9617 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Revised: 12/2024
Full Prescribing Information
1. Indications and Usage for Khapzory Injection
KHAPZORY is indicated for:
- rescue after high-dose methotrexate therapy in adult and pediatric patients with osteosarcoma.
- diminishing the toxicity associated with overdosage of folic acid antagonists or impaired methotrexate elimination adult and pediatric patients.
- The treatment of adults with metastatic colorectal cancer in combination with fluorouracil.
Limitations of Use
KHAPZORY is not indicated for pernicious anemia and megaloblastic anemia secondary to lack the of vitamin B12 because of the risk of progression of neurologic manifestations despite hematologic remission.
2. Khapzory Injection Dosage and Administration
2.1 Important Use Information
KHAPZORY is indicated for intravenous administration only. Do not administer intrathecally.
2.2 Recommended Dosage for Rescue After High-Dose Methotrexate Therapy
The recommended dosage for KHAPZORY is based on a methotrexate dose of 12 grams/m2 administered as intravenous infusion over 4 hours. Twenty-four hours after starting the methotrexate infusion, initiate KHAPZORY at a dose of 7.5 mg (approximately 5 mg/m2) as an intravenous infusion every 6 hours.
Monitor serum creatinine and methotrexate levels at least once daily. Continue KHAPZORY, hydration, and urinary alkalinization (pH of 7 or greater) until the methotrexate level is below 5 x 10-8 M (0.05 micromolar). Adjust the dose KHAPZORY or extend the duration as recommended in Table 1.
| Clinical Situation
| Laboratory Findings
| Recommendation
|
| Normal methotrexate elimination | Serum methotrexate level approximately 10 micromolar at 24 hours after administration, 1 micromolar at 48 hours, and less than 0.2 micromolar at 72 hours. | Administer 7.5 mg by intravenous infusion every 6 hours for 60 hours (10 doses starting at 24 hours after start of methotrexate infusion). |
| Delayed late methotrexate elimination | Serum methotrexate level remaining above 0.2 micromolar at 72 hours, and more than 0.05 micromolar at 96 hours after administration. | Continue 7.5 mg by intravenous infusion every 6 hours, until methotrexate level is less than 0.05 micromolar. |
| Delayed early methotrexate elimination and/or evidence of acute renal injury* | Serum methotrexate level of 50 micromolar or more at 24 hours, or 5 micromolar or more at 48 hours after administration, OR 100% or greater increase in serum creatinine level at 24 hours after methotrexate administration (e.g., an increase from 0.5 mg/dL to a level of 1 mg/dL or more). | Administer 75 mg by intravenous infusion every 3 hours until methotrexate level is less than 1 micromolar; then 7.5 mg by intravenous infusion every 3 hours until methotrexate level is less than 0.05 micromolar. |
*These patients are likely to develop reversible renal failure. In addition to appropriate KHAPZORY therapy, continue hydration and urinary alkalinization, and monitoring fluid and electrolyte status, until the serum methotrexate level has fallen to below 0.05 micromolar and the renal failure has resolved.
Impaired Methotrexate Elimination or Renal Impairment
Decreased methotrexate elimination or renal impairment which are clinically important but less severe than the abnormalities described in Table 1 can occur following methotrexate administration. If toxicity associated with methotrexate is observed, in subsequent courses extend KHAPZORY rescue for an additional 24 hours (total of 14 doses over 84 hours).
Third-Space Fluid Collection and Other Causes of Delayed Methotrexate Elimination
Accumulation in a third space fluid collection (i.e., ascites, pleural effusion), renal insufficiency, or inadequate hydration can delay methotrexate elimination. Under such circumstances, higher doses of KHAPZORY or prolonged administration may be indicated.
2.3 Recommended Dosage for Overdosage of Folic Acid Antagonists or Impaired Methotrexate Elimination
Start KHAPZORY as soon as possible after an overdosage of methotrexate or within 24 hours of methotrexate administration when methotrexate elimination is impaired. As the time interval between methotrexate administration and KHAPZORY increases, the effectiveness of KHAPZORY to diminish methotrexate toxicity may decrease. Administer KHAPZORY 7.5 mg (approximately 5 mg/m2) as an intravenous infusion every 6 hours until the serum methotrexate level is less than 5 x 10-8 M (0.05 micromolar).
Monitor serum creatinine and methotrexate levels at least every 24 hours. Increase the dosage of KHAPZORY to 50 mg/m2 intravenously every 3 hours and continue KHAPZORY at this dosage until the methotrexate level is less than 5 x 10-8 M for the following:
- •
- if the serum creatinine at 24-hours increases 50% or more compared to baseline
- •
- if the methotrexate level at 24-hours is greater than 5 x 10-6 M
- •
- if the methotrexate level at 48-hours is greater than 9 x 10-7 M
Continue concomitant hydration (3 L per day) and urinary alkalinization with sodium bicarbonate. Adjust the bicarbonate dose to maintain urine pH at 7 or greater.
2.4 Dosage in Combination with Fluorouracil for Metastatic Colorectal Cancer
The following regimens have been used for the treatment of colorectal cancer:
- •
- KHAPZORY 100 mg/m2 by intravenous injection over a minimum of 3 minutes, followed by fluorouracil at 370 mg/m2 by intravenous injection , once daily for 5 consecutive days
- •
- KHAPZORY at 10 mg/m2 by intravenous injection, followed by fluorouracil at 425 mg/m2,by intravenous injection once daily for 5 consecutive days
This five-day course may be repeated every 4 weeks for 2 courses, then every 4 to 5 weeks, if the patient has recovered from toxicity from the prior course. Do not adjust KHAPZORY dosage for toxicity.
Refer to fluorouracil prescribing information for information on fluorouracil dosage and dosage modifications for adverse reactions.
2.5 Preparation
Reconstitute the 175 mg vial contents with 3.6 mL of 0.9% Sodium Chloride Injection, USP, to obtain a clear, colorless to yellowish solution (resultant concentration 50 mg per mL levoleucovorin). Reconstitution with a sodium chloride solution with preservatives (e.g., benzyl alcohol) has not been studied. Do not store reconstituted solution for more than 12 hours at room temperature. Protect from light.
Dilute reconstituted solution immediately (if possible), to concentrations of 0.5 mg/mL to 5 mg/mL in 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP. Do not store the diluted reconstituted solution for more than 12 hours at room temperature. Protect from light.
Visually inspect the product for particulate matter and discoloration prior to administration. Discard if particulate matter or discoloration is observed.
3. Dosage Forms and Strengths
For Injection: 175 mg of levoleucovorin as a sterile, white to yellowish lyophilized powder in a single-dose vial for reconstitution.
4. Contraindications
KHAPZORY is contraindicated in patients who have had severe hypersensitivity to leucovorin products, folic acid, or folinic acid [see Adverse Reactions (6.2)].
5. Warnings and Precautions
5.1 Increased Gastrointestinal Toxicities with Fluorouracil
Leucovorin products increase the toxicities of fluorouracil [see Drug Interactions (7)]. Gastrointestinal toxicities, including stomatitis and diarrhea, occur more commonly and may be of greater severity and of prolonged duration. Deaths from severe enterocolitis, diarrhea, and dehydration have occurred in elderly patients receiving weekly d,l-leucovorin and fluorouracil.
Monitor patients for gastrointestinal toxicities. Do not initiate or continue therapy with KHAPZORY and fluorouracil in patients with symptoms of gastrointestinal toxicity until those symptoms have resolved. Monitor patients with diarrhea until it has resolved as rapid deterioration leading to death can occur.
5.2 Drug Interaction with Trimethoprim-Sulfamethoxazole
The concomitant use of d,l-leucovorin with trimethoprim-sulfamethoxazole for the acute treatment of Pneumocystis jiroveci pneumonia in patients with HIV infection was associated with increased rates of treatment failure and morbidity [ see Drug Interation (7)].
6. Adverse Reactions/Side Effects
The following clinical significant adverse reactions are described elsewhere in the labeling:
- •
- Increased gastrointestinal toxicities with fluorouracil [see Warnings and Precautions (5.1)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
High-Dose Methotrexate Therapy
Table 2 presents the frequency of adverse reactions which occurred during the administration of 58 courses of high-dose methotrexate 12 grams/m2 followed by levoleucovorin rescue, for osteosarcoma, in 16 patients aged 6 to 21 years. Most patients received levoleucovorin 7.5 mg every 6 hours for 60 hours or longer, beginning 24 hours after completion of methotrexate administration.
| Adverse Reactions
| Levoleucovorin
n=16 |
|
| All Grades (%)
| Grade 3-4 (%)
|
|
| Gastrointestinal
|
||
| Stomatitis | 38 | 6 |
| Vomiting | 38 | 0 |
| Nausea | 19 | 0 |
| Diarrhea | 6 | 0 |
| Dyspepsia | 6 | 0 |
| Typhlitis | 6 | 6 |
| Respiratory
|
||
| Dyspnea | 6 | 0 |
| Skin and Appendages
|
||
| Dermatitis | 6 | 0 |
| Other
|
||
| Confusion | 6 | 0 |
| Neuropathy | 6 | 0 |
| Renal function abnormal | 6 | 0 |
| Taste perversion | 6 | 0 |
Combination with Fluorouracil in Colorectal Cancer
Table 3 presents the frequency of adverse reactions which occurred in 2 arms of a randomized controlled trial conducted by the North Central Cancer Treatment Group (NCCTG) in patients with metastatic colorectal cancer. The trial failed to show superior overall survival with fluorouracil + levoleucovorin compared to fluorouracil + d,l-leucovorin. Patients were randomized to fluorouracil 370 mg/m2 intravenously and levoleucovorin 100 mg/m2 intravenously, both daily for 5 days, or to fluorouracil 370 mg/m2 intravenously and d,l-leucovorin 200 mg/m2 intravenously, both daily for 5 days. Treatment was repeated week 4 and week 8, and then every 5 weeks until disease progression or unacceptable toxicity.
| Adverse Reactions
| Levoleucovorin/
fluorouracil n=318 | d,l-Leucovorin/
fluorouracil n=307 |
||
| Grades 1-4 (%)
| Grades 3-4 (%)
| Grades 1-4 (%)
| Grades 3-4 (%)
|
|
| Gastrointestinal Disorders
|
||||
| Stomatitis | 72 | 12 | 72 | 14 |
| Diarrhea | 70 | 19 | 65 | 17 |
| Nausea | 62 | 8 | 61 | 8 |
| Vomiting | 40 | 5 | 37 | 6 |
| Abdominal Pain* | 14 | 3 | 19 | 3 |
| General Disorders
|
||||
| Asthenia/Fatigue/Malaise | 29 | 5 | 32 | 11 |
| Skin Disorders
|
||||
| Dermatitis | 29 | 1 | 28 | 1 |
| Alopecia | 26 | 0.3 | 28 | 1 |
| Metabolism and Nutrition | ||||
| Anorexia/Decreased Appetite |
24 |
4 |
25 |
2 |
*Includes abdominal pain, upper abdominal pain, lower abdominal pain, and abdominal tenderness
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of levoleucovorin products. Because these adverse reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. The following have been reported:
- •
- Dermatologic: pruritus, rash
- •
- Respiratory: dyspnea
- •
- Other Clinical Events: temperature change, rigors, allergic reactions
Related/similar drugs
7. Drug Interactions
7.1 Effects of Leucovorin Products on Other Drugs
Antiepileptic Drugs
Folic acid in large amounts may counteract the antiepileptic effect of phenobarbital, phenytoin and primidone and increase the frequency of seizures in susceptible children. It is not known whether folinic acid has the same effects; however, both folic and folinic acids share some common metabolic pathways. Monitor patients taking folinic acid in combination with antiepileptic drugs.
Fluorouracil
Leucovorin products increase the toxicity of fluorouracil. Do not initiate or continue therapy with Fusilev and fluorouracil in patients with symptoms of gastrointestinal toxicity until those symptoms have resolved. Monitor patients with diarrhea until the diarrhea has resolved, as rapid deterioration leading to death can occur [see Warnings and Precautions (5.2)].
Trimethoprim-Sulfamethoxazole
The concomitant use of d,l-leucovorin with trimethoprim-sulfamethoxazole for the acute treatment of Pneumocystis jiroveci pneumonia in patients with HIV infection was associated with increased rates of treatment failure and morbidity in a placebo-controlled study [see Warnings and Precautions (5)].
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
There are limited data with levoleucovorin use in pregnant women. Animal reproduction studies have not been conducted with levoleucovorin.
Levoleucovorin is administered in combination with methotrexate or fluorouracil, which can cause embryo-fetal harm. Refer to methotrexate or fluorouracil prescribing information for additional information.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
8.2 Lactation
Risk Summary
There are no data on the presence of levoleucovorin in human milk or its effects on the breastfed infant or on milk production.
Levoleucovorin is administered in combination with methotrexate or fluorouracil. Refer to methotrexate or fluorouracil prescribing information for additional information.
8.4 Pediatric Use
The safety and effectiveness of KHAPZORY have been established in pediatric patients for rescue after high-dose methotrexate therapy in osteosarcoma and diminishing the toxicity associated with overdosage of folic acid antagonists or impaired methotrexate elimination. Use of levoleucovorin in pediatric patients is supported by open-label clinical trial data in 16 pediatric patients 6 years of age and older, with additional supporting evidence from literature [see Clinical Studies (14.1)].
The safety and effectiveness of KHAPZORY have not been established for the treatment of pediatric patients with advanced metastatic colorectal cancer.
8.5 Geriatric Use
Clinical studies of levoleucovorin in the treatment of osteosarcoma did not include patients aged 65 years and over to determine whether they respond differently from younger patients.
In the NCCTG clinical trial of levoleucovorin in combination with fluorouracil in the treatment of metastatic colorectal cancer, adverse reactions were consistent with fluorouracil related toxicity and were similar for patients age 65 years and older and patients younger than 65 [see Clinical Studies (14.2)].
11. Khapzory Injection Description
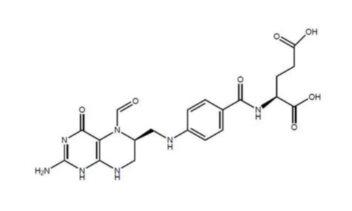
Levoleucovorin is a folate analog and the pharmacologically active levo-isomer of d,l-leucovorin. The chemical name is (2S)-2-[[4-[[(6S)-2-amino-5-formyl-4-oxo-1,6,7,8-tetrahydropteridin-6- yl] methylamino] benzoyl] amino] pentanedioate. The molecular formula is C20H23N7O7 and the molecular weight is 473.45. The molecular structure is:
Levoleucovorin is a slightly hygroscopic, crystalline, yellow powder which is soluble in water when pH is at or above 8.
KHAPZORY 175 mg is a sterile lyophilized powder consisting of 175 mg levoleucovorin, 29.6 mg sodium hydroxide, and 105 mg mannitol in each vial. Additional sodium hydroxide and/or hydrochloric acid may be used to adjust the pH during manufacture. It is intended for intravenous administration after reconstitution with 3.6 mL of sterile 0.9% Sodium Chloride Injection, USP.
12. Khapzory Injection - Clinical Pharmacology
12.1 Mechanism of Action
High-Dose Methotrexate Therapy
Levoleucovorin is the pharmacologically active isomer of 5-formyl tetrahydrofolic acid. Levoleucovorin does not require reduction by dihydrofolate reductase to participate in reactions utilizing folates as a source of “one-carbon” moieties. Administration of levoleucovorin counteracts the therapeutic and toxic effects of folic acid antagonists such as methotrexate, which act by inhibiting dihydrofolate reductase.
Combination with Fluorouracil in Colorectal Cancer
Levoleucovorin enhances the therapeutic and toxic effects of fluorouracil. Fluorouracil is metabolized to 5-fluoro-2'-deoxyuridine-5'-monophosphate (FdUMP), which binds to and inhibits thymidylate synthase (an enzyme important in DNA repair and replication). Levoleucovorin is converted to another reduced folate, 5,10-methylenetetrahydrofolate, which then acts to stabilize the binding of FdUMP to thymidylate synthase, thereby enhancing the inhibition of thymidylate synthase.
12.3 Pharmacokinetics
The pharmacokinetics of levoleucovorin after intravenous injection of a 15 mg dose was studied in healthy subjects. The mean maximum serum total tetrahydrofolate (total-THF) concentration was 1722 ng/mL (CV 39%) and the mean maximum serum (6S)-5-methyl-5,6,7,8-tetrahydrofolate concentration was 275 ng/mL (CV 18%) observed around 0.9 hours post injection.
Distribution
Exploratory studies show that small quantities of systemically administered leucovorin enter the cerebrospinal fluid (CSF), primarily as its major metabolite 5-methyltetrahydrofolate (5-MTHFA). In humans, the CSF levels of 5-MTHFA remain 1-3 orders of magnitude lower than the usual methotrexate concentrations following intrathecal administration.
Elimination
The mean terminal half-life was 5.1 hours for total-THF and 6.8 hours for (6S)-5-methyl-5,6,7,8-tetrahydrofolate.
Drug Interaction Studies
The mean dose-normalized steady-state plasma concentrations for both levoleucovorin and 5-methyl-THF were comparable whether fluorouracil (370 mg/m2/day as an intravenous bolus) was given in combination with levoleucovorin (250 mg/m2 and 1000 mg/m2 as a continuous intravenous infusion for 5.5 days) or in combination with d,l-leucovorin (500 mg/m2 as a continuous intravenous infusion for 5.5 days).
14. Clinical Studies
14.1 Rescue after High-Dose Methotrexate Therapy in Patients with Osteosarcoma
The efficacy of levoleucovorin rescue following high-dose methotrexate were evaluated in 16 patients aged 6 to 21 years, who received 58 courses of therapy for osteogenic sarcoma. High-dose methotrexate was one component of several different combination chemotherapy regimens evaluated across several trials. Methotrexate 12 g/m2 as an intravenous infusion over 4 hours was administered to 13 patients, who received levoleucovorin 7.5 mg every 6 hours for 60 hours or longer beginning 24 hours after completion of methotrexate. Three patients received methotrexate 12.5 g/m2 intravenously over 6 hours, followed by levoleucovorin 7.5 mg by intravenous infusion every 3 hours for 18 doses beginning 12 hours after completion of methotrexate. The mean number of levoleucovorin doses per course was 18.2 and the mean total dose per course was 350 mg. The efficacy of levoleucovorin rescue following high-dose methotrexate was based on adverse reaction profile [see Adverse Reactions (6.1)].
14.2 Metastatic Colorectal Cancer
In a randomized clinical study conducted by the Mayo Clinic and the North Central Cancer Treatment Group (NCCTG) in patients with metastatic colorectal cancer comparing d,l leucovorin 200 mg/m2 and fluorouracil 370 mg/m2 versus leucovorin 20 mg/m2 and fluorouracil 425 mg/m2 versus fluorouracil 500 mg/m2, with all drugs administered by intravenous infusion daily for 5 days every 28 to 35 days, response rates were 26% (p=0.04 versus fluorouracil alone), 43% (p=0.001 versus fluorouracil alone), and 10%, respectively. Respective median survival times were 12.2 months (p=0.037), 12 months (p=0.050), and 7.7 months. The low dose d,l-leucovorin regimen was associated with a statistically significant improvement in weight gain of more than 5%, relief of symptoms, and improvement in performance status. The high dose d,l-leucovorin regimen was associated with a statistically significant improvement in performance status and trended toward improvement in weight gain and in relief of symptoms but these were not statistically significant.
In a second randomized clinical study conducted by Mayo and NCCTG, the fluorouracil alone arm was replaced by sequentially administered methotrexate, fluorouracil, and d,l leucovorin . Response rates with d,l leucovorin 200 mg/m2 and fluorouracil 370 mg/m2 versus d,l leucovorin 20 mg/m2 and fluorouracil 425 mg/m2 versus sequential methotrexate and fluorouracil and d,l leucovorin were 31% (p≤0.01), 42% (p≤0.01), and 14%, respectively. Respective median survival times were 12.7 months (p≤0.04), 12.7 months (p≤0.01), and 8.4 months. There was no statistically significant difference in weight gain of more than 5% or in improvement in performance status between the treatment arms.
A randomized controlled study conducted by the NCCTG in patients with metastatic colorectal cancer failed to show superiority of a regimen of fluorouracil + levoleucovorin to fluorouracil + d,l-leucovorin in overall survival. Patients were randomized to fluorouracil 370 mg/m2 intravenously and levoleucovorin 100 mg/m2 intravenously, both daily for 5 days, or to fluorouracil 370 mg/m2 intravenously and d,l-leucovorin 200 mg/m2 intravenously, both daily for 5 days. Treatment was repeated week 4 and week 8, then every 5 weeks until disease progression or unacceptable toxicity.
16. How is Khapzory Injection supplied
KHAPZORY (levoleucovorin) for injection is a sterile, preservative-free, white to yellowish lyophilized powder in a single-dose vial. It is available as:
175 mg vial –NDC 72893-004-01.
Store at 20°C to 25°C (68°F to 77°F) in original carton until contents are used. Excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature]. Protect solutions from light.
Distributed by:
Acrotech Biopharma Inc
East Windsor, NJ 08520
KHAPZORY is a registered trademark of Acrotech Biopharma Inc.
| KHAPZORY
levoleucovorin injection, powder, lyophilized, for solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Acrotech Biopharma Inc (116965616) |
| Registrant - Acrotech Biopharma Inc (116965616) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Bora Pharmaceuticals Injectables Inc | 119352788 | MANUFACTURE(72893-004) | |
More about Khapzory (levoleucovorin)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Side effects
- Dosage information
- During pregnancy
- FDA approval history
- Drug class: antidotes
- En español