Fragmin: Package Insert / Prescribing Info
Package insert / product label
Generic name: dalteparin sodium
Dosage form: injection
Drug class: Heparins
J Code (medical billing code): J1645 (2500 intl units, injection)
Medically reviewed by Drugs.com. Last updated on Nov 17, 2024.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Drug Interactions
- Use In Specific Populations
- Overdosage
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
FRAGMIN® (dalteparin sodium) injection, USP, for subcutaneous use
Initial U.S. Approval: 1994
WARNING: SPINAL/EPIDURAL HEMATOMAS
See full prescribing information for complete boxed warning.
Epidural or spinal hematomas may occur in patients who are anticoagulated with low molecular weight heparins (LMWH) or heparinoids and are receiving neuraxial anesthesia or undergoing spinal puncture. These hematomas may result in long-term or permanent paralysis. Consider these risks when scheduling patients for spinal procedures. Factors that can increase the risk of developing epidural or spinal hematomas in these patients include:
- •
- Use of indwelling epidural catheters
- •
- Concomitant use of other drugs that affect hemostasis, such as non-steroidal anti-inflammatory drugs (NSAIDs), platelet inhibitors, other anticoagulants.
- •
- A history of traumatic or repeated epidural or spinal punctures
- •
- A history of spinal deformity or spinal surgery
- •
- Optimal timing between the administration of FRAGMIN and neuraxial procedures is not known
Monitor patients frequently for signs and symptoms of neurological impairment. If neurological compromise is noted, urgent treatment is necessary.
Consider the benefits and risks before neuraxial intervention in patients anticoagulated or to be anticoagulated for thromboprophylaxis (5.1, 7).
Recent Major Changes
|
Indications and Usage, Treatment of Symptomatic Venous Thromboembolism (VTE) in Pediatric Patients (1.4) |
10/2024 |
|
Dosage and Administration, Treatment of Symptomatic Venous Thromboembolism (VTE) in Pediatric Patients (2.4) |
10/2024 |
|
Dosage and Administration, Dose Reductions for Thrombocytopenia in Adult Patients with Cancer and in Pediatric Patients with Symptomatic VTE (2.5) |
10/2024 |
Indications and Usage for Fragmin
FRAGMIN is a low molecular weight heparin (LMWH) indicated for
- •
- Prophylaxis of ischemic complications of unstable angina and non-Q-wave myocardial infarction (1.1)
- •
- Prophylaxis of deep vein thrombosis (DVT) in abdominal surgery, hip replacement surgery or medical patients with severely restricted mobility during acute illness (1.2)
- •
- Extended treatment of symptomatic venous thromboembolism (VTE) to reduce the recurrence in adult patients with cancer. In these patients, the FRAGMIN therapy begins with the initial VTE treatment and continues for six months (1.3)
- •
- Treatment of symptomatic venous thromboembolism (VTE) to reduce the recurrence in pediatric patients from birth (gestational age at least 35 weeks) (1.4)
- •
-
Limitations of Use
FRAGMIN is not indicated for the acute treatment of VTE (1.5)
Fragmin Dosage and Administration
|
Indication |
Dosing Regimen |
|
|
Unstable angina and non-Q-wave MI |
120 units/kg subcutaneous every 12 hours (with aspirin) (2.1) |
|
|
DVT prophylaxis in abdominal surgery |
2,500 units subcutaneous once daily or 5,000 units subcutaneous once daily or 2,500 units subcutaneous followed by 2,500 units subcutaneous 12 hours later and then 5,000 units subcutaneous once daily (2.2) |
|
|
DVT prophylaxis in hip replacement surgery |
Postoperative start – 2,500 units subcutaneous 4 hours to 8 hours after surgery, then 5,000 units subcutaneous once daily, or Preoperative start – day of surgery 2,500 units subcutaneous 2 hours before surgery followed by 2,500 units subcutaneous 4 hours to 8 hours after surgery, then 5,000 units subcutaneous once daily (2.2) |
|
|
DVT prophylaxis in medical patients |
5,000 units subcutaneous once daily (2.2) |
|
|
Extended treatment of VTE in adult patients with cancer |
Month 1: 200 units/kg subcutaneous once daily (2.3) |
|
|
Age Group |
Starting Dose |
|
|
Birth (gestational age at least 35 weeks) to less than 2 Years |
150 units/kg twice daily |
|
|
2 Years to less than 8 Years |
125 units/kg twice daily |
|
|
8 Years to less than 17 Years |
100 units/kg twice daily |
|
Do not use as intramuscular injection. FRAGMIN should not be mixed with other injections or infusions (2.7)
Dosage Forms and Strengths
- •
- Injection: 2,500 units/ 0.2 mL, 5,000 units/ 0.2 mL, 7,500 units/ 0.3 mL, 12,500 units/ 0.5 mL, 15,000 units/ 0.6 mL, and 18,000 units/ 0.72 mL single-dose prefilled syringes (3)
- •
- Injection: 10,000 units/mL single-dose graduated syringes (3)
- •
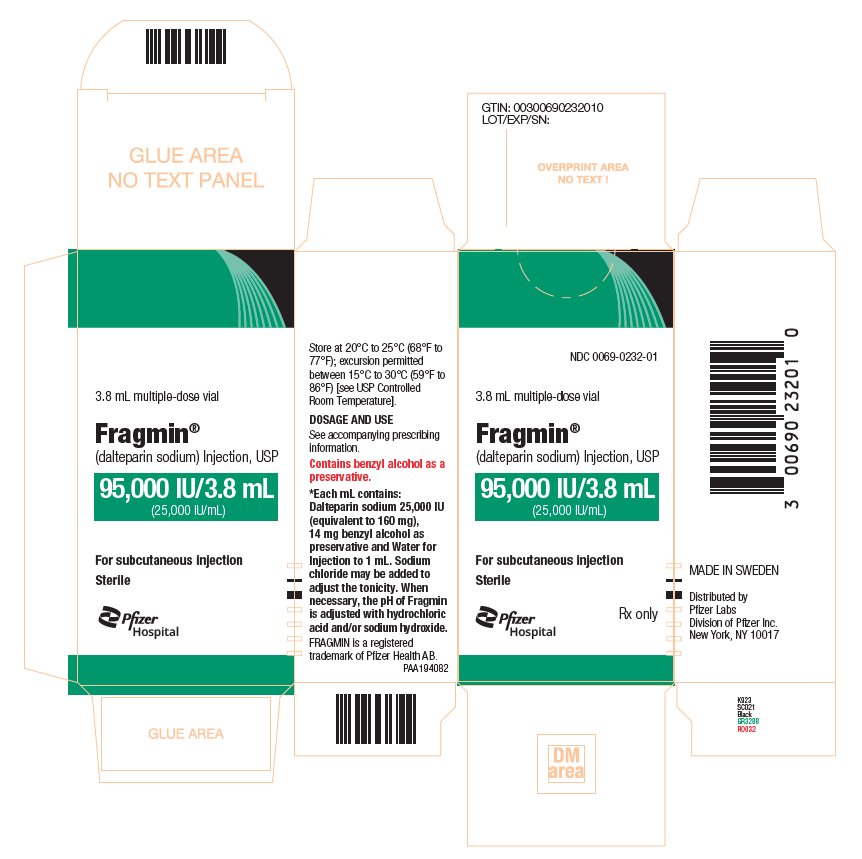
- Injection: 95,000 units/ 3.8 mL (25,000 units/mL) multiple-dose vials (3)
- •
- Injection: 10,000 units/ 4 mL (2,500 units/mL) single-dose vials (3)
Contraindications
- •
- Active major bleeding (4)
- •
- History of heparin induced thrombocytopenia or heparin induced thrombocytopenia with thrombosis (4)
- •
- Hypersensitivity to dalteparin sodium (4, 6.1)
- •
- In patients undergoing Epidural/Neuraxial anesthesia, do not administer FRAGMIN (5.1)
- o
- As a treatment for unstable angina and non-Q-wave MI
- o
- For prolonged VTE prophylaxis (4)
- •
- Hypersensitivity to heparin or pork products (4)
Warnings and Precautions
- •
- Hemorrhage: Use caution in conditions with increased risk of hemorrhage (5.1)
- •
- Thrombocytopenia: Monitor thrombocytopenia of any degree closely (5.2)
- •
- Benzyl Alcohol Preservative: Do not use multiple-dose formulations in neonates and infants as they contain benzyl alcohol (5.3)
- •
- Laboratory Tests: Periodic blood counts recommended (5.4)
Adverse Reactions/Side Effects
Most common adverse reactions (>1%) are: bleeding (including hemorrhage), thrombocytopenia (Type I), hematoma at the injection site, pain at the injection site, transient elevation of transaminases (6)
To report SUSPECTED ADVERSE REACTIONS, contact Pfizer Inc. at 1-800-438-1985 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
The use of FRAGMIN in patients receiving oral anticoagulants, platelet inhibitors, and thrombolytic agents may increase the risk of bleeding (7)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 10/2024
Full Prescribing Information
WARNING: SPINAL/EPIDURAL HEMATOMAS
Epidural or spinal hematomas may occur in patients who are anticoagulated with low molecular weight heparins (LMWH) or heparinoids and are receiving neuraxial anesthesia or undergoing spinal puncture. These hematomas may result in long-term or permanent paralysis. Consider these risks when scheduling patients for spinal procedures. Factors that can increase the risk of developing epidural or spinal hematomas in these patients include:
- •
- Use of indwelling epidural catheters
- •
- Concomitant use of other drugs that affect hemostasis, such as non-steroidal anti-inflammatory drugs (NSAIDs), platelet inhibitors, other anticoagulants.
- •
- A history of traumatic or repeated epidural or spinal punctures
- •
- A history of spinal deformity or spinal surgery
- •
- Optimal timing between the administration of FRAGMIN and neuraxial procedures is not known
Monitor patients frequently for signs and symptoms of neurological impairment. If neurological compromise is noted, urgent treatment is necessary.
Consider the benefits and risks before neuraxial intervention in patients anticoagulated or to be anticoagulated for thromboprophylaxis [see Warnings and Precautions (5.1) and Drug Interactions (7)].
1. Indications and Usage for Fragmin
1.1 Prophylaxis of Ischemic Complications in Unstable Angina and Non-Q-Wave Myocardial Infarction
FRAGMIN Injection is indicated for the prophylaxis of ischemic complications in unstable angina and non-Q-wave myocardial infarction, when concurrently administered with aspirin therapy [see Clinical Studies (14.1)].
1.2 Prophylaxis of Deep Vein Thrombosis
FRAGMIN is indicated for the prophylaxis of deep vein thrombosis (DVT), which may lead to pulmonary embolism (PE):
- •
- In patients undergoing hip replacement surgery [see Clinical Studies (14.2)];
- •
- In patients undergoing abdominal surgery who are at risk for thromboembolic complications [see Clinical Studies (14.3)];
- •
- In medical patients who are at risk for thromboembolic complications due to severely restricted mobility during acute illness [see Clinical Studies (14.4)].
1.3 Extended Treatment of Symptomatic Venous Thromboembolism (VTE) in Adult Patients with Cancer
FRAGMIN is indicated for the extended treatment of symptomatic venous thromboembolism (VTE) (proximal DVT and/or PE), to reduce the recurrence of VTE in adult patients with cancer. In these patients, the FRAGMIN therapy begins with the initial VTE treatment and continues for six months [see Clinical Studies (14.5)].
2. Fragmin Dosage and Administration
2.1 Recommended Dosage for Prophylaxis of Ischemic Complications in Unstable Angina and Non-Q-Wave Myocardial Infarction
In patients with unstable angina or non-Q-wave myocardial infarction, the recommended dose of FRAGMIN Injection is 120 units/kg of body weight, but not more than 10,000 units, subcutaneously every 12 hours with concurrent oral aspirin (75 mg to 165 mg once daily) therapy. Treatment should be continued until the patient is clinically stabilized. The usual duration of administration is 5 days to 8 days. Concurrent aspirin therapy is recommended except when contraindicated.
Table 1 lists the volume of FRAGMIN in mL (based on the 3.8 mL multiple-dose vial 25,000 units/mL) and quantity of FRAGMIN in units, to be administered for a range of patient weights.
|
Table 1 |
||||||
|
Quantity and Volume of FRAGMIN to be Administered by Patient Weight |
||||||
|
Patient weight (kg) |
<50 kg |
50 kg to 59 kg |
60 kg to 69 kg |
70 kg to 79 kg |
80 kg to 89 kg |
≥90 kg |
|
Quantity of FRAGMIN (units) |
5,500 units |
6,500 units |
7,500 units |
9,000 units |
10,000 units |
10,000 units |
|
Volume of FRAGMIN (mL) |
0.22 mL |
0.26 mL |
0.3 mL |
0.36 mL |
0.4 mL |
0.4 mL |
2.2 Prophylaxis of Deep Vein Thrombosis
Prophylaxis of VTE Following Hip Replacement Surgery: Table 2 presents the dosing options for patients undergoing hip replacement surgery. The usual duration of administration is 5 days to 10 days after surgery; up to 14 days of treatment with FRAGMIN have been well tolerated in clinical trials.
| Table 2 | ||||
|---|---|---|---|---|
| Dosing Options for Patients Undergoing Hip Replacement Surgery | ||||
|
||||
|
Timing of First Dose of FRAGMIN |
Dose of FRAGMIN to be Given Subcutaneously |
|||
|
10 Hours to 14 Hours Before Surgery |
Within 2 Hours Before Surgery |
4 Hours to 8 Hours After Surgery* |
Postoperative Period† |
|
|
Postoperative Start |
--- |
--- |
2,500 units‡ |
5,000 units once daily |
|
Preoperative Start - Day of Surgery |
--- |
2,500 units |
2,500 units‡ |
5,000 units once daily |
|
Preoperative Start - Evening Before Surgery§ |
5,000 units |
--- |
5,000 units |
5,000 units once daily |
Abdominal Surgery: In patients undergoing abdominal surgery with a risk of thromboembolic complications, the recommended dose of FRAGMIN is 2,500 units administered by subcutaneous injection once daily, starting 1 hour to 2 hours prior to surgery and repeated once daily postoperatively. The usual duration of administration is 5 days to 10 days.
In patients undergoing abdominal surgery associated with a high risk of thromboembolic complications, such as malignant disorder, the recommended dose of FRAGMIN is 5,000 units subcutaneously the evening before surgery, then once daily postoperatively. The usual duration of administration is 5 days to 10 days. Alternatively, in patients with malignancy, 2,500 units of FRAGMIN can be administered subcutaneously 1 hour to 2 hours before surgery followed by 2,500 units subcutaneously 12 hours later, and then 5,000 units once daily postoperatively. The usual duration of administration is 5 days to 10 days.
2.3 Extended Treatment of Symptomatic Venous Thromboembolism (VTE) in Adult Patients with Cancer
In adult patients with cancer and symptomatic VTE, the recommended dosing of FRAGMIN is as follows: for the first 30 days of treatment administer FRAGMIN 200 units/kg total body weight subcutaneously once daily. The total daily dose should not exceed 18,000 units. Table 3 lists the dose of FRAGMIN to be administered once daily during the first month for a range of patient weights.
Month 1
| Table 3 | |
|---|---|
| Dose of FRAGMIN to be Administered Subcutaneously by Patient Weight during the First Month | |
|
Body Weight (kg) |
FRAGMIN Dose (units) |
|
≤56 kg |
10,000 units |
|
57 kg to 68 kg |
12,500 units |
|
69 kg to 82 kg |
15,000 units |
|
83 kg to 98 kg |
18,000 units |
|
≥99 kg |
18,000 units |
Months 2 to 6
Administer FRAGMIN at a dose of approximately 150 units/kg, subcutaneously once daily during Months 2 through 6. The total daily dose should not exceed 18,000 units. Table 4 lists the dose of FRAGMIN to be administered once daily for a range of patient weights during months 2 to 6.
| Table 4 | |
|---|---|
| Dose of FRAGMIN to be Administered Subcutaneously by Patient Weight during Months 2 to 6 | |
|
Body Weight (kg) |
FRAGMIN Dose (units) |
|
≤56 kg |
7,500 units |
|
57 kg to 68 kg |
10,000 units |
|
69 kg to 82 kg |
12,500 units |
|
83 kg to 98 kg |
15,000 units |
|
≥99 kg |
18,000 units |
Safety and efficacy beyond six months have not been evaluated in patients with cancer and acute symptomatic VTE [see Warnings and Precaution (5) and Adverse Reactions (6.1)].
2.4 Treatment of Symptomatic Venous Thromboembolism (VTE) in Pediatric Patients
The recommended starting dose according to pediatric age is provided in Table 5.
| Table 5 | |
|---|---|
| Starting Doses for Pediatric Patients with Symptomatic VTE | |
|
Age Group |
Starting Dose |
|
Birth (gestational age at least 35 weeks) to less than 2 Years |
150 units/kg twice daily |
|
125 units/kg twice daily |
|
100 units/kg twice daily |
After initiation of FRAGMIN, measure anti-Xa level prior to the 4th dose. Samples for anti-Xa level should be drawn 4 hours after administration of FRAGMIN. Adjust doses in increments of 25 units/kg to achieve target anti-Xa level between 0.5 units/mL and 1 unit/mL. Individualize the maintenance dose of FRAGMIN based on the dose that achieves target anti-Xa level collected 4 hours after administration of FRAGMIN. Monitor anti-Xa level periodically in pediatric patients to maintain anti-Xa level between 0.5 units/mL and 1 unit/mL [see Clinical Pharmacology (12.3) and Clinical Studies (14.5)].
The 3.8 mL multiple-dose vials of FRAGMIN contain 14 mg/mL of benzyl alcohol [see Warnings and Precautions (5.3)].
Whenever possible, administer benzyl alcohol-free formulations (prefilled syringes) in pediatric patients [see Warnings and Precautions (5.3) and Use in Specific Populations (8.4)].
The 10,000 units/4 mL (2,500 units/mL) single-dose vials are preservative-free. For treatment of neonates, use this vial presentation.
2.5 Dose Reductions for Thrombocytopenia in Adult Patients with Cancer and in Pediatric Patients with Symptomatic VTE
Dose reductions recommended in patients receiving FRAGMIN who experience thrombocytopenia are presented below in Table 6.
| Table 6 | ||
|---|---|---|
|
Platelet Count less than or equal to 50,000/mm3 |
Platelet Count greater than 50,000/mm3 up to 100,000/mm3 |
|
|
Adults |
Discontinue FRAGMIN until platelet count recovers to above 50,000/mm3. |
Reduce the daily dose of FRAGMIN by 2,500 units until the platelet count recovers to greater or equal to 100,000/mm3. |
|
Children from birth (gestational age at least 35 weeks) to less than 17 Years |
Discontinue FRAGMIN until platelet count recovers to above 50,000/mm3. |
Reduce the daily dose of FRAGMIN by 50% until the platelet count recovers to greater or equal to 100,000/mm3. |
2.6 Dose Reductions for Renal Insufficiency in Extended Treatment of Acute Symptomatic VTE in Adult Patients with Cancer
In patients with severely impaired renal function (CrCl <30 mL/min), monitor anti-Xa levels to determine the appropriate FRAGMIN dose. Target anti-Xa range is 0.5 units/mL to 1.5 units/mL. When monitoring anti-Xa in these patients, perform sampling 4 hours to 6 hours after FRAGMIN dosing and only after the patient has received 3 to 4 doses [see Clinical Pharmacology (12.3)].
2.7 Administration
Latex Allergy: Persons with latex allergies should not handle the FRAGMIN prefilled syringe because the needle shield may contain natural rubber latex which may cause allergic reactions.
FRAGMIN is administered by subcutaneous injection. It must not be administered by intramuscular injection.
FRAGMIN Injection should not be mixed with other injections or infusions unless specific compatibility data are available that support such mixing.
Subcutaneous injection technique: Patients should be sitting or lying down and FRAGMIN administered by deep subcutaneous injection. FRAGMIN may be injected in a U-shape area around the navel, the upper outer side of the thigh or the upper outer quadrangle of the buttock. The injection site should be varied daily. When the area around the navel or the thigh is used, using the thumb and forefinger, you must lift up a fold of skin while giving the injection. The entire length of the needle should be inserted at a 45 to 90 degree angle.
Inspect FRAGMIN prefilled syringes and vials visually for particulate matter and discoloration prior to administration.
After first penetration of the rubber stopper, store the multiple-dose vials at room temperature for up to 2 weeks. Discard any unused solution after 2 weeks.
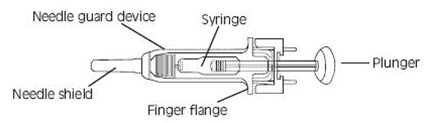
Instructions for using the prefilled single-dose syringes preassembled with needle guard devices
Fixed dose syringes: To ensure delivery of the full dose, do not expel the air bubble from the prefilled syringe before injection. Hold the syringe assembly by the open sides of the device. Remove the needle shield. Insert the needle into the injection area as instructed above. Depress the plunger of the syringe while holding the finger flange until the entire dose has been given. The needle guard will not be activated unless the entire dose has been given. Remove needle from the patient. Let go of the plunger and allow syringe to move up inside the device until the entire needle is guarded. Discard the syringe assembly in approved containers.
Graduated syringes: Hold the syringe assembly by the open sides of the device. Remove the needle shield. With the needle pointing up, prepare the syringe by expelling the air bubble and then continuing to push the plunger to the desired dose or volume, discarding the extra solution in an appropriate manner. Insert the needle into the injection area as instructed above. Depress the plunger of the syringe while holding the finger flange until the entire dose remaining in the syringe has been given. The needle guard will not be activated unless the entire dose has been given. Remove needle from the patient. Let go of the plunger and allow syringe to move up inside the device until the entire needle is guarded. Discard the syringe assembly in approved containers.
3. Dosage Forms and Strengths
FRAGMIN (dalteparin sodium) injection is a sterile, aqueous, clear, colorless or straw-colored solution for injection, available in the following dosage forms and strengths:
- •
- Injection: 2,500 units/ 0.2 mL, 5,000 units/ 0.2 mL, 7,500 units/ 0.3 mL, 12,500 units/ 0.5 mL, 15,000 units/ 0.6 mL, and 18,000 units/ 0.72 mL sterile, single-dose, prefilled syringes preassembled with a needle guard device.
- •
- Injection: 10,000 units/mL sterile, single-dose, graduated syringes preassembled with a needle guard device.
- •
- Injection: 95,000 units/ 3.8 mL (25,000 units/mL) sterile, multiple-dose vials.
- •
- Injection: 10,000 units/ 4 mL (2,500 units/mL) sterile, single-dose vials.
4. Contraindications
FRAGMIN is contraindicated in:
- •
- Patients with active major bleeding.
- •
- Patients with a history of heparin induced thrombocytopenia or heparin induced thrombocytopenia with thrombosis.
- •
- Patients with prior hypersensitivity to dalteparin sodium (e.g., pruritis, rash, anaphylactic reactions) [see Adverse Reactions (6.1)].
- •
- Patients undergoing Epidural/Neuraxial anesthesia, do not administer FRAGMIN [see Boxed Warning and Warnings and Precautions (5.1)];
- o
- As a treatment for unstable angina and non-Q-wave MI.
- o
- For prolonged VTE prophylaxis.
- •
- Patients with prior hypersensitivity to heparin or pork products.
5. Warnings and Precautions
5.1 Risk of Hemorrhage including Spinal/Epidural Hematomas
Spinal or epidural hemorrhage and subsequent hematomas can occur with the associated use of low molecular weight heparins or heparinoids and neuraxial (spinal/epidural) anesthesia or spinal puncture. The risk of these events is higher with the use of post-operative indwelling epidural catheters, with the concomitant use of additional drugs affecting hemostasis such as NSAIDs, with traumatic or repeated epidural or spinal puncture, or in patients with a history of spinal surgery or spinal deformity [see Boxed Warning, Adverse Reactions (6.2), and Drug Interactions (7)].
To reduce the potential risk of bleeding associated with the concurrent use of FRAGMIN and epidural or spinal anesthesia/analgesia or spinal puncture, consider the pharmacokinetic profile of FRAGMIN [see Clinical Pharmacology (12.3)].
Placement or removal of an epidural catheter or lumbar puncture is best performed when the anticoagulant effect of FRAGMIN is low; however, the exact timing to reach a sufficiently low anticoagulant effect in each patient is not known. No additional hemostasis-altering medications should be administered due to the additive effects.
Patients on preoperative FRAGMIN thromboprophylaxis can be assumed to have altered coagulation. The first postoperative FRAGMIN thromboprophylaxis dose (2,500 units) should be administered 6 hours to 8 hours postoperatively. The second postoperative dose (2,500 units or 5,000 units) should occur no sooner than 24 hours after the first dose. Placement or removal of a catheter should be delayed for at least 12 hours after administration of 2,500 units once daily of FRAGMIN, at least 15 hours after the administration of 5,000 units once daily of FRAGMIN, and at least 24 hours after the administration of higher doses (200 units/kg once daily, 120 units/kg twice daily) of FRAGMIN. Anti-Xa levels are still detectable at these time points, and these delays are not a guarantee that neuraxial hematoma will be avoided.
Although a specific recommendation for timing of a subsequent FRAGMIN dose after catheter removal cannot be made, consider delaying this next dose for at least 4 hours, based on a benefit-risk assessment considering both the risk for thrombosis and the risk for bleeding in the context of the procedure and patient risk factors. For patients with creatinine clearance <30 mL/minute, additional considerations are necessary because elimination of FRAGMIN may be more prolonged; consider doubling the timing of removal of a catheter, at least 24 hours for the lower prescribed dose of FRAGMIN (2,500 units or 5,000 units once daily) and at least 48 hours for the higher dose (200 units/kg once daily, 120 units/kg twice daily) [see Clinical Pharmacology (12.3)].
Should the physician decide to administer anticoagulation in the context of epidural or spinal anesthesia/analgesia or lumbar puncture, frequent monitoring must be exercised to detect any signs and symptoms of neurological impairment such as midline back pain, sensory and motor deficits (numbness or weakness in lower limbs), bowel and/or bladder dysfunction. Instruct patients to report immediately if they experience any of the above signs or symptoms. If signs or symptoms of spinal hematoma are suspected, initiate urgent diagnosis and treatment including consideration for spinal cord decompression even though such treatment may not prevent or reverse neurological sequelae.
Use FRAGMIN with extreme caution in patients who have an increased risk of hemorrhage, such as those with severe uncontrolled hypertension, bacterial endocarditis, congenital or acquired bleeding disorders, active ulceration and angiodysplastic gastrointestinal disease, hemorrhagic stroke, or shortly after brain, spinal or ophthalmological surgery. FRAGMIN may enhance the risk of bleeding in patients with thrombocytopenia or platelet defects; severe liver or kidney insufficiency, hypertensive or diabetic retinopathy, and recent gastrointestinal bleeding. Bleeding can occur at any site during therapy with FRAGMIN.
5.2 Thrombocytopenia
Heparin-induced thrombocytopenia can occur with the administration of FRAGMIN. The incidence of this complication is unknown at present. In clinical practice, cases of thrombocytopenia with thrombosis, amputation and death have been observed [see Contraindications (4)]. Closely monitor thrombocytopenia of any degree.
In FRAGMIN clinical trials supporting non-cancer indications, platelet counts of <50,000/mm3 occurred in <1% of patients.
In the clinical trial of adult patients with cancer and acute symptomatic VTE treated for up to 6 months in the FRAGMIN treatment arm, platelet counts of <100,000/mm3 occurred in 13.6% of patients, including 6.5% who also had platelet counts less than 50,000/mm3. In the same clinical trial, thrombocytopenia was reported as an adverse event in 10.9% of patients in the FRAGMIN arm and 8.1% of patients in the Oral Anti-Coagulant (OAC) arm. FRAGMIN dose was decreased or interrupted in patients whose platelet counts fell below 100,000/mm3.
In the clinical trial of pediatric patients with or without cancer with acute symptomatic VTE treated for up to 3 months with FRAGMIN, platelet counts of <100,000/mm3 occurred in 37% of patients, including 21% who also had platelet counts less than 50,000/mm3. In the same clinical trial, thrombocytopenia was reported as an adverse reaction in 21% of patients. FRAGMIN dose was interrupted in patients whose platelet counts fell below 50,000/mm3.
5.3 Risk of Serious Adverse Reactions in Neonates and Infants due to Benzyl Alcohol Preservative
Use preservative-free FRAGMIN in neonates and infants.
Serious and fatal adverse reactions including "gasping syndrome" can occur in neonates and low-birth weight infants treated with medications that contain the preservative benzyl alcohol. The "gasping syndrome" is characterized by central nervous system depression, metabolic acidosis, and gasping respirations. When prescribing FRAGMIN in infants, consider the combined daily metabolic load of benzyl alcohol from all sources including FRAGMIN multiple-dose vial (contains 14 mg of benzyl alcohol per mL) and other drugs containing benzyl alcohol. The minimum amount of benzyl alcohol at which serious adverse reactions may occur is not known [see Use in Specific Populations (8.4)]. Because benzyl alcohol may cross the placenta, use caution when administering FRAGMIN preserved with benzyl alcohol to pregnant women. If anticoagulation with FRAGMIN is needed during pregnancy, use preservative-free formulations where possible [see Use in Specific Populations (8.1)].
5.4 Laboratory Tests
Periodic routine complete blood counts, including platelet count, blood chemistry, and stool occult blood tests are recommended during the course of treatment with FRAGMIN. When administered at recommended prophylaxis doses, routine coagulation tests such as Prothrombin Time (PT) and Activated Partial Thromboplastin Time (APTT) are relatively insensitive measures of FRAGMIN activity and, therefore, unsuitable for monitoring the anticoagulant effect of FRAGMIN. Monitor anti-Xa level periodically in pediatric patients. Anti-Xa may be used to monitor the anticoagulant effect of FRAGMIN, such as in patients with severe renal impairment or if abnormal coagulation parameters or bleeding occurs during FRAGMIN therapy.
6. Adverse Reactions/Side Effects
The following clinically significant adverse reactions are described in more detail in other sections of the prescribing information.
- •
- Risk of Hemorrhage including Spinal/Epidural Hematomas [see Warnings and Precautions (5.1)]
- •
- Thrombocytopenia [see Warnings and Precautions (5.2)]
- •
- Benzyl Alcohol Preservative Risk to Premature Infants [see Warnings and Precautions (5.3)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not accurately reflect the rates observed in practice.
Hemorrhage
The most commonly reported adverse reactions are hematoma at the injection site and hemorrhagic complications. The risk for bleeding varies with the indication and may increase with higher doses.
Unstable Angina and Non-Q-Wave Myocardial Infarction
Table 7 summarizes major bleeding reactions that occurred with FRAGMIN, heparin, and placebo in clinical trials of unstable angina and non-Q-wave myocardial infarction.
| Table 7 | |||
|---|---|---|---|
| Major Bleeding Reactions in Unstable Angina and Non-Q-Wave Myocardial Infarction | |||
|
|||
|
Indication |
Dosing Regimen |
||
|
Unstable Angina and Non-Q-Wave MI |
FRAGMIN
|
Placebo
|
|
|
15/1497 (1.0) |
7/731 (1.0) |
4/760 (0.5) |
|
Hip Replacement Surgery
Table 8 summarizes: 1) all major bleeding reactions and, 2) other bleeding reactions possibly or probably related to treatment with FRAGMIN (preoperative dosing regimen), warfarin sodium, or heparin in two hip replacement surgery clinical trials.
| Table 8 | ||||
|---|---|---|---|---|
| Bleeding Reactions Following Hip Replacement Surgery | ||||
|
||||
|
Indication |
FRAGMIN vs Warfarin Sodium |
FRAGMIN vs Heparin |
||
|
Dosing Regimen |
Dosing Regimen |
|||
|
Hip Replacement Surgery |
FRAGMIN*
|
Warfarin
|
FRAGMIN‡
|
Heparin
|
|
n (%) |
n (%) |
n (%) |
n (%) |
|
|
Major Bleeding Reactions§ |
7/274 (2.6) |
1/279 (0.4) |
0 |
3/69 (4.3) |
|
Other Bleeding Reactions¶
|
8/274 (2.9) |
5/279 (1.8) |
0 |
0 |
|
Wound Hematoma |
6/274 (2.2) |
0 |
0 |
0 |
|
Injection Site Hematoma |
3/274 (1.1) |
NA |
2/69 (2.9) |
7/69 (10.1) |
Six of the patients treated with FRAGMIN experienced seven major bleeding reactions. Two of the reactions were wound hematoma (one requiring reoperation), three were bleeding from the operative site, one was intraoperative bleeding due to vessel damage, and one was gastrointestinal bleeding.
In the third hip replacement surgery clinical trial, the incidence of major bleeding reactions was similar in all three treatment groups: 3.6% (18/496) for patients who started FRAGMIN before surgery; 2.5% (12/487) for patients who started FRAGMIN after surgery; and 3.1% (15/489) for patients treated with warfarin sodium.
Abdominal Surgery
Table 9 summarizes bleeding reactions that occurred in clinical trials which studied FRAGMIN 2,500 units and 5,000 units administered once daily to abdominal surgery patients.
| Table 9 | ||||
|---|---|---|---|---|
| Bleeding Reactions Following Abdominal Surgery | ||||
|
Indication |
FRAGMIN vs Placebo |
FRAGMIN vs FRAGMIN |
||
|
Dosing Regimen |
Dosing Regimen |
|||
|
Abdominal Surgery |
FRAGMIN |
Placebo |
FRAGMIN |
FRAGMIN |
|
Postoperative Transfusions |
14/182 |
13/182 |
89/1,025 |
125/1,033 |
|
Wound Hematoma |
2/79 |
2/77 |
1/1,030 |
4/1,039 |
|
Reoperation Due to Bleeding |
1/79 |
1/78 |
2/1,030 |
13/1,038 |
|
Injection Site Hematoma |
8/172 |
2/174 |
36/1,026 |
57/1,035 |
| Table 9 Cont. | ||||
|---|---|---|---|---|
| Bleeding Reactions Following Abdominal Surgery | ||||
|
Indication |
FRAGMIN vs Heparin |
|||
|
Dosing Regimen |
||||
|
Abdominal Surgery |
FRAGMIN |
Heparin |
FRAGMIN |
Heparin |
|
Postoperative Transfusions |
26/459 |
36/454 |
81/508 |
63/498 |
|
Wound Hematoma |
16/467 |
18/467 |
12/508 |
6/498 |
|
Reoperation Due to Bleeding |
2/392 |
3/392 |
4/508 |
2/498 |
|
Injection Site Hematoma |
1/466 |
5/464 |
36/506 |
47/493 |
In a trial comparing FRAGMIN 5,000 units once daily to FRAGMIN 2,500 units once daily in patients undergoing surgery for malignancy, the incidence of bleeding reactions was 4.6% and 3.6%, respectively (n.s.). In a trial comparing FRAGMIN 5,000 units once daily to heparin 5,000 units twice daily, in the malignancy subgroup the incidence of bleeding reactions was 3.2% and 2.7%, respectively for FRAGMIN and Heparin (n.s.).
Medical Patients with Severely Restricted Mobility During Acute Illness
Table 10 summarizes major bleeding reactions that occurred in a clinical trial of medical patients with severely restricted mobility during acute illness.
| Table 10 | ||
|---|---|---|
| Bleeding Reactions in Medical Patients with Severely Restricted Mobility During Acute Illness | ||
|
||
|
Indication |
Dosing Regimen |
|
|
Medical Patients with Severely Restricted Mobility |
FRAGMIN
|
Placebo
|
|
Major Bleeding Reactions* at Day 14 |
8/1,848 (0.4) |
0/1,833 (0) |
|
Major Bleeding Reactions* at Day 21 |
9/1,848 (0.5) |
3/1,833 (0.2) |
Three of the major bleeding reactions that occurred by Day 21 were fatal, all due to gastrointestinal hemorrhage (two patients in the group treated with FRAGMIN and one in the group receiving placebo).
Adult Patients with Cancer and Acute Symptomatic VTE
Table 11 summarizes the number of patients with bleeding reactions that occurred in the clinical trial of adult patients with cancer and acute symptomatic VTE. A bleeding event was considered major if it: 1) was accompanied by a decrease in hemoglobin of ≥2 g/dL in connection with clinical symptoms; 2) occurred at a critical site (intraocular, spinal/epidural, intracranial, retroperitoneal, or pericardial bleeding); 3) required transfusion of ≥2 units of blood products; or 4) led to death. Minor bleeding was classified as clinically overt bleeding that did not meet criteria for major bleeding.
At the end of the six-month study, a total of 46 (13.6%) patients in the FRAGMIN arm and 62 (18.5%) patients in the OAC arm experienced any bleeding event. One bleeding event (hemoptysis in a patient in the FRAGMIN arm at Day 71) was fatal.
| Table 11 | ||||||
|---|---|---|---|---|---|---|
| Bleeding Reactions (Major and Any) (As treated population)* | ||||||
|
||||||
|
Study period |
FRAGMIN 200 units/kg (max. 18,000 units) subcutaneous once daily x 1 month, then 150 units/kg (max. 18,000 units) subcutaneous once daily x 5 months |
OAC FRAGMIN 200 units/kg (max 18,000 units) subcutaneous once daily x 5 to 7 days and OAC for 6 months (target INR 2 to 3) |
||||
|
Number at risk |
Patients with Major Bleeding n (%) |
Patients with Any Bleeding n (%) |
Number at risk |
Patients with Major Bleeding n (%) |
Patients with Any Bleeding n (%) |
|
|
Total during study |
338 |
19 (5.6) |
46 (13.6) |
335 |
12 (3.6) |
62 (18.5) |
|
Week 1 |
338 |
4 (1.2) |
15 (4.4) |
335 |
4 (1.2) |
12 (3.6) |
|
Weeks 2 to 4 |
332 |
9 (2.7) |
17 (5.1) |
321 |
1 (0.3) |
12 (3.7) |
|
Weeks 5 to 28 |
297 |
9 (3.0) |
26 (8.8) |
267 |
8 (3) |
40 (15) |
Elevations of Serum Transaminases
In FRAGMIN clinical trials supporting non-cancer indications, where hepatic transaminases were measured, asymptomatic increases in transaminase levels (SGOT/AST and SGPT/ALT) greater than three times the upper limit of normal of the laboratory reference range were seen in 4.7% and 4.2%, respectively, of patients during treatment with FRAGMIN.
In the FRAGMIN clinical trial of patients with cancer and acute symptomatic venous thromboembolism treated with FRAGMIN for up to 6 months, asymptomatic increases in transaminase levels, AST and ALT, greater than three times the upper limit of normal of the laboratory reference range were reported in 8.9% and 9.5% of patients, respectively. The frequencies of Grades 3 and 4 increases in AST and ALT, as classified by the National Cancer Institute, Common Toxicity Criteria (NCI-CTC) Scoring System, were 3% and 3.8%, respectively. Grades 2, 3 & 4 combined have been reported in 12% and 14% of patients, respectively.
Other
Allergic Reactions: Allergic reactions (i.e., pruritus, rash, fever, injection site reaction, bullous eruption) have occurred. Cases of anaphylactoid reactions have been reported.
Local Reactions: Pain at the injection site was reported in 4.5% of patients treated with FRAGMIN 5,000 units once daily vs 11.8% of patients treated with heparin 5,000 units twice daily in the abdominal surgery trials. In the hip replacement trials, pain at injection site was reported in 12% of patients treated with FRAGMIN 5,000 units once daily vs 13% of patients treated with heparin 5,000 units three times a day.
Pediatric Patients with Symptomatic VTE
The data below reflect exposure to FRAGMIN from two studies in pediatric patients from newborn to less than 18 years of age with or without cancer and symptomatic VTE (n = 50). Patients were started on FRAGMIN using age and weight-based dosing via subcutaneous injection twice daily. Anti-Xa levels were measured prior to the 4th dose and then periodically to determine whether dose adjustments were required, using 25 units/kg increments, to achieve a target anti-Xa level of 0.5 units/mL to 1.0 units/mL. The median time on treatment with FRAGMIN was 86 days (range 2 days to 170 days).
In pediatric patients with symptomatic VTE, the most common (greater than 10%) adverse reactions were injection site bruising (30%), contusion (12%), and epistaxis (10%).
Major bleeding was defined as any fatal bleeding, clinically overt bleeding with a decrease in hemoglobin of ≥2g/dL in 24 hours, overt bleeding deemed by the attending physician to be unrelated to the subject's underlying condition and accompanied by blood product administration, overt bleeding that was retroperitoneal, intracranial, intraspinal, intraocular, or intraarticular, or overt bleeding deemed by the attending physician to necessitate permanent discontinuation of trial medication. Major bleeding (intestinal hematoma) occurred in one patient (2%). Discontinuation due to adverse reactions occurred in 12% of patients, most often due to thrombocytopenia (4%).
Neonatal Patients
No controlled clinical trials have been conducted with FRAGMIN in neonatal patients with VTE; however, data are available from a non‑interventional retrospective medical chart review study of 16 neonates with VTE in the UK who were admitted to hospital between January 2010 and December 2021 and were treated with FRAGMIN. The mean (±SD) duration of dalteparin treatment was 62±30 days. For these 16 neonatal patients, the mean (±SD) daily dose of dalteparin at initiation was 365±196 units/kg (median:309 units/kg). The mean (±SD) daily dose of dalteparin including dose at initiation and dose changes was 575±320 units/kg (median: 450 units/kg). After the initial dose, all patients required one or more dose changes; 15 of 16 patients required dose increases due to anti-Xa level being initially below the target range of 0.5 units/mL to 1 units/mL.
The safety profile in neonates was similar to other pediatric patients.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of FRAGMIN. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Since first international market introduction in 1985, there have been more than 15 reports of epidural or spinal hematoma formation with concurrent use of FRAGMIN and spinal/epidural anesthesia or spinal puncture. The majority of patients had postoperative indwelling epidural catheters placed for analgesia or received additional drugs affecting hemostasis. In some cases, the hematoma resulted in long-term or permanent paralysis (partial or complete) [see Boxed Warning].
Musculoskeletal system: Osteoporosis
Skin or subcutaneous tissues disorders: Skin necrosis, cases of alopecia reported that improved on drug discontinuation
Metabolism and nutrition disorders: Hyperkalemia
Related/similar drugs
7. Drug Interactions
The use of FRAGMIN in patients receiving oral anticoagulants, platelet inhibitors, and thrombolytic agents may increase the risk of bleeding [see Warnings and Precautions (5)].
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
Available data from published literature and postmarketing reports have not reported a clear association with FRAGMIN and adverse developmental outcomes. There are risks to the mother associated with untreated VTE in pregnancy, and a potential for adverse effects on the preterm infant when FRAGMIN is used in pregnancy (see Clinical Considerations). In animal reproduction studies, there was no evidence of embryo-fetal toxicity or teratogenicity when dalteparin sodium was administered to pregnant rats and rabbits during organogenesis at doses 2 to 4 times (rats) and 4 times (rabbits) the human dose of 100 units/kg dalteparin based on the body surface area (see Data). Because animal reproduction studies are not always predictive of human response, FRAGMIN should be used during pregnancy only if clearly needed.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Disease-Associated Maternal and/or Embryo/Fetal Risk
Published data describe that women with a previous history of VTE in pregnancy are at higher risk for recurrence during subsequent pregnancies compared to those with no risk factor for VTE (4.5% versus 2.7% respectively, relative risk 1.7, 95% CI: 1.0–2.8).
Fetal/Neonatal Adverse Reactions
Cases of "gasping syndrome" have occurred in premature infants when large amounts of benzyl alcohol have been administered (99 mg/kg/day to 404 mg/kg/day). The 3.8 mL multiple-dose vials of FRAGMIN contain 14 mg/mL of benzyl alcohol [see Warnings and Precautions (5.3)].
Animal Data
In reproductive and developmental toxicity studies, pregnant rats and rabbits received dalteparin sodium during organogenesis at intravenous doses up to 2,400 units/kg (14,160 units/m2) (rats) and 4,800 units/kg (40,800 units/m2) (rabbits). These exposures were 2 to 4 times (rats) and 4 times (rabbits) the human dose of 100 units/kg dalteparin based on the body surface area. These studies revealed no evidence of teratogenicity or embryo-fetal toxicity.
8.2 Lactation
Risk Summary
Limited published data indicate that dalteparin is present in human milk in small amounts (see Data). No adverse effects on the breastfed infant have been reported. There are no data on the effects of the drug on milk production. Oral absorption of dalteparin is expected to be low, but the clinical implications, if any, of this small amount of anticoagulant activity on a breastfed infant are unknown. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for FRAGMIN and any potential adverse effects on the breastfed child from FRAGMIN or from the underlying maternal condition.
A study evaluated samples of maternal blood and breast milk in 15 lactating women receiving prophylactic doses of dalteparin in the immediate postpartum period (days 4 to 8 after Cesarean-section). The samples were collected before and 3 hours to 4 hours after daily injections of 2500 units dalteparin. Small amounts of anti-Xa activity (range <0.005 to 0.037 units/mL) in breast milk were detected in 11 of the 15 women. Because this study evaluated colostrum or transitional milk at a single timepoint during the 24 hours dosing interval, the clinical relevance of this data is unclear in regard to passage of drug into mature milk and the quantification of drug exposure to the infant over the full dosing interval.
8.4 Pediatric Use
The safety and effectiveness of FRAGMIN for the treatment of symptomatic venous thromboembolism (VTE) in patients have been established in pediatric patients from birth (gestational age at least 35 weeks) to less than 17 years of age.
Use of FRAGMIN for this indication is supported by evidence from well-controlled studies in adults with additional pharmacokinetic, pharmacodynamic, efficacy, and safety data from two separate prospective studies in pediatric patients aged 1 month and older with symptomatic VTE, and a retrospective study in neonatal patients aged birth (gestational age at least 35 weeks) to 1 month with VTE [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.5]. The frequency, type and severity of adverse reactions observed were generally consistent with those observed in adults.
Use preservative-free FRAGMIN in neonates and infants.
Serious adverse reactions including fatal reactions and the “gasping syndrome” occurred in premature neonates and low-birth weight infants in the neonatal intensive care unit who received benzyl alcohol preserved medications. In these cases, benzyl alcohol dosages of 99 mg/kg/day to 234 mg/kg/day produced high levels of benzyl alcohol and its metabolites in the blood and urine (blood levels of benzyl alcohol were 0.61 mmol/L to 1.378 mmol/L). Additional adverse reactions included gradual neurological deterioration, seizures, intracranial hemorrhage, hematologic abnormalities, skin breakdown, hepatic and renal failure, hypotension, bradycardia, and cardiovascular collapse. Preterm, low-birth weight infants may be more likely to develop these reactions because they may be less able to metabolize benzyl alcohol.
When prescribing FRAGMIN multiple-dose vials in infants consider the combined daily metabolic load of benzyl alcohol from all sources including FRAGMIN multiple-dose vials (FRAGMIN contains 14 mg of benzyl alcohol per mL) and other drugs containing benzyl alcohol. The minimum amount of benzyl alcohol at which serious adverse reactions may occur is not known [see Warnings and Precautions (5.3)].
The long-term effects of treatment with FRAGMIN in pediatric patients, including effects on growth and bone metabolism, are unknown.
8.5 Geriatric Use
Of the total number of patients in clinical studies of FRAGMIN, 5,516 patients were 65 years of age or older and 2,237 were 75 or older. No overall differences in effectiveness were observed between these subjects and younger subjects. Some studies suggest that the risk of bleeding increases with age. Postmarketing surveillance and literature reports have not revealed additional differences in the safety of FRAGMIN between elderly and younger patients. Give careful attention to dosing intervals and concomitant medications (especially antiplatelet medications) in geriatric patients, particularly in those with low body weight (<45 kg) and those predisposed to decreased renal function [see Warnings and Precautions (5) and Clinical Pharmacology (12)].
10. Overdosage
An excessive dosage of FRAGMIN Injection may lead to hemorrhagic complications. These may generally be stopped by slow intravenous injection of protamine sulfate (1% solution), at a dose of 1 mg protamine for every 100 anti-Xa units of FRAGMIN given. If the APTT measured 2 hours to 4 hours after the first infusion remains prolonged, a second infusion of 0.5 mg protamine sulfate per 100 anti-Xa units of FRAGMIN may be administered. Even with these additional doses of protamine, the APTT may remain more prolonged than would usually be found following administration of unfractionated heparin. In all cases, the anti-Xa activity is never completely neutralized (maximum about 60% to 75%).
Take particular care to avoid overdosage with protamine sulfate. Administration of protamine sulfate can cause severe hypotensive and anaphylactoid reactions. Because fatal reactions, often resembling anaphylaxis, have been reported with protamine sulfate, give protamine sulfate only when resuscitation techniques and treatment for anaphylactic shock are readily available. For additional information, consult the labeling of Protamine Sulfate Injection, USP, products.
11. Fragmin Description
FRAGMIN Injection (dalteparin sodium injection) is a sterile, low molecular weight heparin. It is available in single-dose, prefilled syringes preassembled with a needle guard device, and multiple-dose vials. With reference to the W.H.O. First International Low Molecular Weight Heparin Reference Standard, each syringe contains either 2,500 units, 5,000 units, 7,500 units, 10,000 units, 12,500 units, 15,000 units or 18,000 anti-Xa international units (units), equivalent to 16 mg, 32 mg, 48 mg, 64 mg, 80 mg, 96 mg or 115.2 mg dalteparin sodium, respectively. Each multiple-dose vial contains 25,000 anti-Xa units per 1 mL (equivalent to 160 mg dalteparin sodium), for a total of 95,000 anti-Xa units per vial. Each single-dose vial contains 2,500 anti-Xa units per 1 mL (equivalent to 16 mg dalteparin sodium) for a total of 10,000 anti-Xa units per vial.
Each prefilled syringe also contains Water for Injection and sodium chloride, when required, to maintain physiologic ionic strength. The prefilled syringes are preservative-free. Each multiple-dose vial also contains Water for Injection and 14 mg of benzyl alcohol per mL as a preservative. The pH of both formulations is 5.0 to 7.5. When necessary, the pH of FRAGMIN is adjusted with hydrochloric acid and/or sodium hydroxide [see Dosage and Administration (2.7) and How Supplied/Storage and Handling (16)].
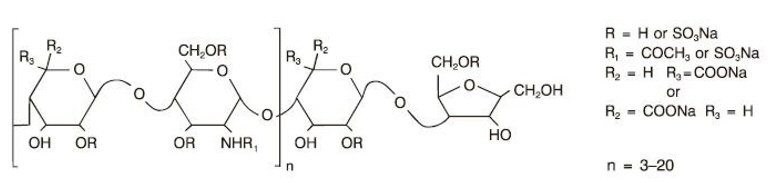
Dalteparin sodium is produced through controlled nitrous acid depolymerization of sodium heparin from porcine intestinal mucosa followed by a chromatographic purification process. It is composed of strongly acidic sulfated polysaccharide chains (oligosaccharide, containing 2,5-anhydro-D-mannitol residues as end groups) with an average molecular weight of 5,000 and about 90% of the material within the range 2,000–9,000. The molecular weight distribution is:
|
<3000 daltons |
3.0–15% |
|
|
3,000 to 8,000 daltons |
65.0–78.0% |
|
|
>8,000 daltons |
14.0–26.0% |
Structural Formula
12. Fragmin - Clinical Pharmacology
12.1 Mechanism of Action
Dalteparin is a low molecular weight heparin with antithrombotic properties. It acts by enhancing the inhibition of Factor Xa and thrombin by antithrombin. In humans, dalteparin potentiates preferentially the inhibition of coagulation Factor Xa, while only slightly affecting the activated partial thromboplastin time (APTT).
12.2 Pharmacodynamics
Doses of FRAGMIN Injection of up to 10,000 anti-Xa units administered subcutaneously as a single dose or two 5,000 units doses 12 hours apart to healthy subjects did not produce a significant change in platelet aggregation, fibrinolysis, or global clotting tests such as prothrombin time (PT), thrombin time (TT) or APTT. Subcutaneous administration of doses of 5,000 units twice daily of FRAGMIN for seven consecutive days to patients undergoing abdominal surgery did not markedly affect APTT, Platelet Factor 4 (PF4), or lipoprotein lipase.
12.3 Pharmacokinetics
Adults
Mean peak levels of plasma anti-Xa activity following single subcutaneous doses of 2,500 units, 5,000 units and 10,000 units were 0.19 ± 0.04 units/mL, 0.41 ± 0.07 units/mL and 0.82 ± 0.10 units/mL, respectively, and were attained in about 4 hours in most subjects. Absolute bioavailability in healthy volunteers, measured as the anti-Xa activity, was 87 ± 6%. Increasing the dose from 2,500 units to 10,000 units resulted in an overall increase in anti-Xa AUC that was greater than proportional by about one-third.
Peak anti-Xa activity increased more or less linearly with dose over the same dose range. There appeared to be no appreciable accumulation of anti-Xa activity with twice-daily dosing of 100 units/kg subcutaneously for up to 7 days.
The volume of distribution for dalteparin anti-Xa activity was 40 mL/kg to 60 mL/kg. The mean plasma clearances of dalteparin anti-Xa activity in normal volunteers following single intravenous bolus doses of 30 and 120 anti-Xa units/kg were 24.6 ± 5.4 mL/hr/kg and 15.6 ± 2.4 mL/hr/kg, respectively. The corresponding mean disposition half-lives were 1.47 ± 0.3 hours and 2.5 ± 0.3 hours.
Following intravenous doses of 40 units/kg and 60 units/kg, mean terminal half-lives were 2.1 ± 0.3 hours and 2.3 ± 0.4 hours, respectively. Longer apparent terminal half-lives (3 hours to 5 hours) are observed following subcutaneous dosing, possibly due to delayed absorption. In patients with chronic renal insufficiency requiring hemodialysis, the mean terminal half-life of anti-Xa activity following a single intravenous dose of 5,000 units FRAGMIN was 5.7 ± 2.0 hours, i.e., considerably longer than values observed in healthy volunteers, therefore, greater accumulation can be expected in these patients.
Pediatric Patients
The pharmacokinetics of twice-daily subcutaneous dalteparin, measured as anti-Xa activity, was characterized in 89 pediatric patients with or without cancer from two clinical studies and 1 observational study. Dalteparin pharmacokinetics (PK) were described by a 1-compartment model with linear absorption and elimination and PK parameters are shown in Table 12. After correcting for the body weight, clearance (CL/F) decreased with increasing age, while volume of distribution at steady-state (Vd/F) remained similar. The mean elimination half-life increased with age.
| Table 12 | |||||
|---|---|---|---|---|---|
| Pharmacokinetic Parameters of Dalteparin in Pediatric Population | |||||
| Abbreviations: CL=clearance; F=absolute bioavailability; SD=standard deviation; t½β=elimination half-life; Vd=volume of distribution. | |||||
|
Parameter |
3 Weeks to <8 Weeks |
≥8 Weeks to <2 Years |
≥2 Years to <8 Years |
≥8 Years to <12 Years |
≥12 Years to <20 Years |
|
Median Age (range) (years) |
0.06 (0.04 – 0.14) |
0.5 (0.2 – 1.91) |
4.47 (2.01 – 7.6) |
9.62 (8.01 – 10.5) |
15.9 (12.0 – 19.5) |
|
Derived Mean (SD) CL/F (mL/h/kg) |
55.8 (3.91) |
40.4 (8.49) |
26.7 (4.75) |
22.4 (3.40) |
18.8 (3.01) |
|
Derived Mean (SD) Vd/F (mL/kg) |
181 (15.3) |
175 (55.3) |
160 (25.6) |
165 (27.3) |
171 (38.9) |
|
Derived Mean (SD) t½β (h) |
2.25 (0.173) |
3.02 (0.688) |
4.27 (1.05) |
5.11 (0.509) |
6.28 (0.937) |
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Dalteparin sodium has not been tested for its carcinogenic potential in long-term animal studies. It was not mutagenic in the in vitro Ames Test, mouse lymphoma cell forward mutation test and human lymphocyte chromosomal aberration test and in the in vivo mouse micronucleus test. Dalteparin sodium at subcutaneous doses up to 1,200 units/kg (7,080 units/m2) did not affect the fertility or reproductive performance of male and female rats.
14. Clinical Studies
14.1 Prophylaxis of Ischemic Complications in Unstable Angina and Non-Q-Wave Myocardial Infarction
In a double-blind, randomized, placebo-controlled clinical trial, patients who recently experienced unstable angina with EKG changes or non-Q-wave myocardial infarction (MI) were randomized to FRAGMIN Injection 120 units/kg or placebo every 12 hours subcutaneously. In this trial, unstable angina was defined to include only angina with EKG changes. All patients, except when contraindicated, were treated concurrently with aspirin (75 mg once daily) and beta blockers. Treatment was initiated within 72 hours of the event (the majority of patients received treatment within 24 hours) and continued for 5 days to 8 days. A total of 1,506 patients were enrolled and treated; 746 received FRAGMIN and 760 received placebo. The mean age of the study population was 68 years (range 40 years to 90 years) and the majority of patients were white (99.7%) and male (63.9%). The combined incidence of the endpoint of death or myocardial infarction was lower for FRAGMIN compared with placebo at 6 days after initiation of therapy. These results were observed in an analysis of all-randomized and all-treated patients. The combined incidence of death, MI, need for intravenous heparin or intravenous. nitroglycerin, and revascularization was also lower for FRAGMIN than for placebo (see Table 13).
| Table 13 | ||
|---|---|---|
| Efficacy of FRAGMIN in the Prophylaxis of Ischemic Complications in Unstable Angina and Non-Q-Wave Myocardial Infarction | ||
|
||
|
Indication |
Dosing Regimen |
|
|
FRAGMIN
|
Placebo
|
|
|
All Treated Unstable Angina and Non-Q-Wave MI Patients |
746 |
760 |
|
Primary Endpoints - 6 day timepoint |
13/741 (1.8)* |
36/757 (4.8) |
|
Secondary Endpoints - 6 day timepoint |
59/739 (8)* |
106/756 (14) |
In a second randomized, controlled trial designed to evaluate long-term treatment with FRAGMIN (6 days to 45 days), data were also collected comparing 1 week (5 days to 8 days) treatment of FRAGMIN 120 units/kg every 12 hours subcutaneously with heparin at an APTT-adjusted dosage. All patients, except when contraindicated, were treated concurrently with aspirin (100 mg per day to 165 mg per day). Of the 1,499 patients enrolled, 1,482 patients were treated; 751 received FRAGMIN and 731 received heparin. The mean age of the study population was 64 years (range 25 years to 92 years) and the majority of patients were white (96.0%) and male (64.2%). The incidence of the combined endpoint of death, myocardial infarction, or recurrent angina during this 1-week treatment period (5 days to 8 days) was 9.3% for FRAGMIN and 7.6% for heparin (p = 0.323).
14.2 Prophylaxis of Deep Vein Thrombosis in Patients Following Hip Replacement Surgery
In an open-label randomized study, FRAGMIN 5,000 units administered once daily subcutaneously was compared with warfarin sodium, administered orally, in patients undergoing hip replacement surgery. Treatment with FRAGMIN was initiated with a 2,500 units dose subcutaneously within 2 hours before surgery, followed by a 2,500 units dose subcutaneously the evening of the day of surgery. Then, a dosing regimen of FRAGMIN 5,000 units subcutaneously once daily was initiated on the first postoperative day. The first dose of warfarin sodium was given the evening before surgery, then continued daily at a dose adjusted for INR 2 to 3. Treatment in both groups was then continued for 5 days to 9 days postoperatively. Of the 580 patients enrolled, 553 were treated and 550 underwent surgery. Of those who underwent surgery, 271 received FRAGMIN and 279 received warfarin sodium. The mean age of the study population was 63 years (range 20 years to 92 years) and the majority of patients were white (91.1%) and female (52.9%). The incidence of deep vein thrombosis (DVT), as determined by evaluable venography, was significantly lower for the group treated with FRAGMIN compared with patients treated with warfarin sodium (see Table 14).
| Table 14 | ||
|---|---|---|
| Efficacy of FRAGMIN in the Prophylaxis of Deep Vein Thrombosis Following Hip Replacement Surgery | ||
|
||
|
Indication |
Dosing Regimen |
|
|
FRAGMIN
|
Warfarin Sodium
|
|
|
All Treated Hip Replacement Surgery Patients |
271 |
279 |
|
Treatment Failures in Evaluable Patients |
28/192 (14.6)‡ |
49/190 (25.8) |
|
Proximal DVT |
10/192 (5.2)§ |
16/190 (8.4) |
|
PE |
2/271 (0.7) |
2/279 (0.7) |
In a second single-center, double-blind study of patients undergoing hip replacement surgery, FRAGMIN 5,000 units once daily subcutaneously starting the evening before surgery, was compared with heparin 5,000 units subcutaneously three times a day, starting the morning of surgery. Treatment in both groups was continued for up to 9 days postoperatively. Of the 140 patients enrolled, 139 were treated and 136 underwent surgery. Of those who underwent surgery, 67 received FRAGMIN and 69 received heparin. The mean age of the study population was 69 years (range 42 years to 87 years) and the majority of patients were female (58.8%). In the intent-to-treat analysis, the incidence of proximal DVT was significantly lower for patients treated with FRAGMIN compared with patients treated with heparin (6/67 vs 18/69; p = 0.012). The incidence of pulmonary embolism detected by lung scan was also significantly lower in the group treated with FRAGMIN (9/67 vs 19/69; p = 0.032).
A third multi-center, double-blind, randomized study evaluated a postoperative dosing regimen of FRAGMIN for thromboprophylaxis following total hip replacement surgery. Patients received either FRAGMIN or warfarin sodium, randomized into one of three treatment groups. One group of patients received the first dose of FRAGMIN 2,500 units subcutaneous within 2 hours before surgery, followed by another dose of FRAGMIN 2,500 units subcutaneous at least 4 hours (6.6 ± 2.3 hours) after surgery. Another group received the first dose of FRAGMIN 2,500 units subcutaneous at least 4 hours (6.6 ± 2.4 hours) after surgery. Then, both of these groups began a dosing regimen of FRAGMIN 5,000 units once daily subcutaneous on postoperative day 1. The third group of patients received warfarin sodium the evening of the day of surgery, then continued daily at a dose adjusted to maintain INR 2 to 3. Treatment for all groups was continued for 4 days to 8 days postoperatively, after which time all patients underwent bilateral venography.
In the total enrolled study population of 1,501 patients, 1,472 patients were treated; 496 received FRAGMIN (first dose before surgery), 487 received FRAGMIN (first dose after surgery) and 489 received warfarin sodium. The mean age of the study population was 63 years (range 18 years to 91 years) and the majority of patients were white (94.4%) and female (51.8%).
Administration of the first dose of FRAGMIN after surgery was as effective in reducing the incidence of thromboembolic reactions as administration of the first dose of FRAGMIN before surgery (44/336 vs 37/338; p =0.448). Both dosing regimens of FRAGMIN were more effective than warfarin sodium in reducing the incidence of thromboembolic reactions following hip replacement surgery.
14.3 Prophylaxis of Deep Vein Thrombosis Following Abdominal Surgery in Patients at Risk for Thromboembolic Complications
Abdominal surgery patients at risk include those who are over 40 years of age, obese, undergoing surgery under general anesthesia lasting longer than 30 minutes, or who have additional risk factors such as malignancy or a history of deep vein thrombosis or pulmonary embolism.
FRAGMIN administered once daily subcutaneously beginning prior to surgery and continued for 5 days to 10 days after surgery, reduced the risk of DVT in patients at risk for thromboembolic complications in two double-blind, randomized, controlled clinical trials performed in patients undergoing major abdominal surgery. In the first study, a total of 204 patients were enrolled and treated; 102 received FRAGMIN and 102 received placebo. The mean age of the study population was 64 years (range 40 years to 98 years) and the majority of patients were female (54.9%). In the second study, a total of 391 patients were enrolled and treated; 195 received FRAGMIN and 196 received heparin. The mean age of the study population was 59 years (range 30 years to 88 years) and the majority of patients were female (51.9%). FRAGMIN 2,500 units were superior to placebo and similar to heparin in reducing the risk of DVT (see Tables 15 and 16).
| Table 15 | ||
|---|---|---|
| Efficacy of FRAGMIN in the Prophylaxis of Deep Vein Thrombosis Following Abdominal Surgery | ||
|
Indication |
Dosing Regimen |
|
|
FRAGMIN
|
Placebo
|
|
|
All Treated Abdominal Surgery Patients |
102 |
102 |
|
Treatment Failures in Evaluable Patients |
4/91 (4.4)* |
16/91 (17.6) |
|
Proximal DVT |
0 |
5/91 (5.5) |
|
Distal DVT |
4/91 (4.4) |
11/91 (12.1) |
|
PE |
0 |
2/91 (2.2)† |
| Table 16 | ||
|---|---|---|
| Efficacy of FRAGMIN in the Prophylaxis of Deep Vein Thrombosis Following Abdominal Surgery | ||
|
||
|
Indication |
Dosing Regimen |
|
|
FRAGMIN
|
Heparin
|
|
|
All Treated Abdominal Surgery Patients |
195 |
196 |
|
Treatment Failures in Evaluable Patients |
7/178 (3.9)* |
7/174 (4) |
|
Proximal DVT |
3/178 (1.7) |
4/174 (2.3) |
|
Distal DVT |
3/178 (1.7) |
3/174 (1.7) |
|
PE |
1/178 (0.6) |
0 |
In a third double-blind, randomized study performed in patients undergoing major abdominal surgery with malignancy, FRAGMIN 5,000 units subcutaneous once daily was compared with FRAGMIN 2,500 units subcutaneous once daily. Treatment was continued for 6 days to 8 days. A total of 1,375 patients were enrolled and treated; 679 received FRAGMIN 5,000 units and 696 received 2,500 units. The mean age of the combined groups was 71 years (range 40 years to 95 years). The majority of patients were female (51.0%). FRAGMIN 5,000 units once daily was more effective than FRAGMIN 2,500 units once daily in reducing the risk of DVT in patients undergoing abdominal surgery with malignancy (see Table 17).
| Table 17 | ||
|---|---|---|
| Efficacy of FRAGMIN in the Prophylaxis of Deep Vein Thrombosis Following Abdominal Surgery | ||
|
Indication |
Dosing Regimen |
|
|
FRAGMIN
|
FRAGMIN
|
|
|
All Treated Abdominal Surgery Patients* |
696 |
679 |
|
Treatment Failures in Evaluable Patients |
99/656 (15.1)† |
60/645 (9.3) |
|
Proximal DVT |
18/657 (2.7) |
14/646 (2.2) |
|
Distal DVT |
80/657 (12.2) |
41/646 (6.3) |
|
PE | ||
|
Fatal |
1/674 (0.1) |
1/669 (0.1) |
|
Non-fatal |
2 |
4 |
14.4 Prophylaxis of Deep Vein Thrombosis in Medical Patients at Risk for Thromboembolic Complications Due to Severely Restricted Mobility During Acute Illness
In a double-blind, multi-center, randomized, placebo-controlled clinical trial, general medical patients with severely restricted mobility who were at risk of VTE were randomized to receive either FRAGMIN 5,000 units or placebo subcutaneously once daily during Days 1 to 14 of the study. These patients had an acute medical condition requiring a projected hospital stay of at least 4 days, and were confined to bed during waking hours. The study included patients with congestive heart failure (NYHA Class III or IV), acute respiratory failure not requiring ventilatory support, and the following acute conditions with at least one risk factor occurring in >1% of treated patients: acute infection (excluding septic shock), acute rheumatic disorder, acute lumbar or sciatic pain, vertebral compression, or acute arthritis of the lower extremities. Risk factors include >75 years of age, cancer, previous DVT/PE, obesity, and chronic venous insufficiency. A total of 3,681 patients were enrolled and treated: 1,848 received FRAGMIN and 1,833 received placebo. The mean age of the study population was 69 years (range 26 years to 99 years), 92.1% were white and 51.9% were female. The primary efficacy endpoint was evaluated at Day 21 and was defined as at least one of the following within Days 1 to 21 of the study: asymptomatic DVT (diagnosed by compression ultrasound), a confirmed symptomatic DVT, a confirmed pulmonary embolism or sudden death. The follow-up extended through Day 90.
When given at a dose of 5,000 units once a day subcutaneously, FRAGMIN significantly reduced the incidence of thromboembolic reactions including verified DVT by Day 21 (see Table 18). The prophylactic effect was sustained through Day 90.
| Table 18 | ||
|---|---|---|
| Efficacy of FRAGMIN in the Prophylaxis of Deep Vein Thrombosis in Medical Patients with Severely Restricted Mobility During Acute Illness | ||
|
Indication |
Dosing Regimen |
|
|
FRAGMIN
|
Placebo
|
|
|
All Treated Medical Patients During Acute Illness |
1,848 |
1,833 |
|
Treatment failure in evaluable patients |
42/1,518 (2.8)† |
73/1,473 (5) |
|
Total Thromboembolic Reactions |
37/1,513 (2.5) |
70/1,470 (4.8) |
|
Total DVT |
32/1,508 (2.1) |
64/1,464 (4.4) |
|
Proximal DVT |
29/1,518 (1.9) |
60/1,474 (4.1) |
|
Symptomatic VTE |
10/1,759 (0.6) |
17/1,740 (1) |
|
PE |
5/1,759 (0.3) |
6/1,740 (0.3) |
|
Sudden Death |
5/1,829 (0.3) |
3/1,807 (0.2) |
14.5 Patients with Cancer and Acute Symptomatic VTE
Adult Patients
In a prospective, multi-center, open-label, clinical trial, 676 patients with cancer and newly diagnosed, objectively confirmed acute deep vein thrombosis (DVT) and/or pulmonary embolism (PE) were studied. Patients were randomized to either FRAGMIN 200 units/kg subcutaneous (max 18,000 units subcutaneous daily for one month) then 150 units/kg subcutaneous (max 18,000 units) subcutaneous daily for five months (FRAGMIN arm) or FRAGMIN 200 units/kg subcutaneous (max 18,000 units) subcutaneous daily for 5 days to 7 days and oral anticoagulant for six months (OAC arm). In the OAC arm, oral anticoagulation was adjusted to maintain an INR of 2 to 3. Patients were evaluated for recurrence of symptomatic VTE every two weeks for six months.
The median age of patients was 64 years (range: 22 years to 89 years); 51.5% of patients were females; 95.3% of patients were Caucasians. Types of tumors were: gastrointestinal tract (23.7%), genito-urinary (21.5%), breast (16%), lung (13.3%), hematological tumors (10.4%), and other tumors (15.1%).
A total of 27 (8.0%) and 53 (15.7%) patients in the FRAGMIN and OAC arms, respectively, experienced at least one episode of an objectively confirmed, symptomatic DVT and/or PE during the 6-month study period. Most of the difference occurred during the first month of treatment (see Table 19). The benefit was maintained over the 6-month study period.
| Table 19 | ||||||
|---|---|---|---|---|---|---|
| Recurrent VTE in Patients with Cancer (Intention to treat population)* | ||||||
|
||||||
|
Study Period |
FRAGMIN arm |
OAC arm |
||||
|
FRAGMIN 200 units/kg (max. 18,000 units) subcutaneous once daily × 1 month, then 150 units/kg (max. 18,000 units) subcutaneous once daily × 5 months |
FRAGMIN 200 units/kg (max 18,000 units) subcutaneous once daily × 5 days to 7 days and OAC for 6 months (target INR 2 to 3) |
|||||
|
Number at Risk |
Patients with VTE |
% |
Number at Risk |
Patients with VTE |
% |
|
|
Total |
338 |
27 |
8 |
338 |
53 |
15.7 |
|
Week 1 |
338 |
5 |
1.5 |
338 |
8 |
2.4 |
|
Weeks 2 to 4 |
331 |
6 |
1.8 |
327 |
25 |
7.6 |
|
Weeks 5 to 28 |
307 |
16 |
5.2 |
284 |
20 |
7 |
In the intent-to-treat population that included all randomized patients, the primary comparison of the cumulative probability of the first VTE recurrence over the 6 months study period was statistically significant (p <0.01) in favor of the FRAGMIN arm, with most of the treatment difference evident in the first month.
Pediatric Patients
The efficacy of FRAGMIN is based on a single-arm, open-label, multi-center clinical trial in 38 pediatric patients with or without cancer and symptomatic deep vein thrombosis and/or pulmonary embolism. This study included 26 patients with an active malignancy and 12 patients without cancer. Of the 38 total patients, 3 patients were less than 2 years, 8 patients were 2 years to less than 8 years, 7 patients were 8 years to less than 12 years, and 17 patients were 12 years to less than or equal to 18 years. Patients were treated with FRAGMIN for up to 3 months, with starting doses by age and weight.
The efficacy of FRAGMIN was established by the achievement of therapeutic anti-Xa levels by Day 7 of therapy during the dose adjustment period and supported by the number of patients with lack of VTE progression or new VTE.
The efficacy population included pediatric patients who achieved a therapeutic anti-Xa level (0.5 mg per day to 1 units/mL) in the 7-day dose adjustment period (N = 34). The median doses of FRAGMIN (units/kg) required to achieve a therapeutic anti-Xa level during the dose adjustment period are presented in Table 20. Therapeutic anti-Xa levels were achieved within a mean of 2.6 days (range: 1 days to 7 days).
At study completion, 21 patients (62%) achieved resolution of the qualifying VTE, 7 patients (21%) showed regression, 2 patients (6%) showed no change, and no patients showed progression of the qualifying VTE. One patient (3%) experienced a new VTE during the study while on treatment.
| Table 20 | ||
|---|---|---|
| Median Doses of FRAGMIN (units/kg) Associated with Therapeutic Anti-Xa Level (0.5 to 1 units/mL) by Age Cohort (N = 34) | ||
|
Age Cohort |
N |
Median Dose (units/kg) |
|
Greater than or equal to 8 weeks to less than 2 years |
2 |
208 units/kg |
|
Greater than or equal to 2 years to less than 8 years |
8 |
128 units/kg |
|
Greater than or equal to 8 years to less than 12 years |
7 |
125 units/kg |
|
Greater than or equal to 12 years to less than or equal to 18 years |
17 |
117 units/kg |
14.6 Neonatal Patients
The efficacy of FRAGMIN in neonatal patients is based on a retrospective, non-interventional, multi-center study in 16 neonatal patients with deep vein thrombosis. Of the 16 total patients, the median gestational age was 39.1 weeks and median age at first FRAGMIN dose was 13.5 days. Patients were treated with FRAGMIN for up to 3.5 months, with starting doses determined by weight.
The efficacy of FRAGMIN was established by the achievement of VTE resolution and/or completion of FRAGMIN treatment and was supported by the number of patients with lack of VTE progression or new VTE.
The efficacy population included neonatal patients who completed a treatment course of FRAGMIN and/or achieved VTE resolution. The median starting dose of FRAGMIN and the median dose (units/kg) required to achieve a therapeutic anti-Xa level during the dose adjustment period are presented in Table 21. Therapeutic anti-Xa levels were documented for 8 of the 16 patients.
At the end of follow-up, resolution of the baseline VTE was documented for 4 patients (25%). No patients were reported to have either partial regression of the VTE or a new VTE. Of the remaining 12 patients, 1 patient (6%) switched to another anticoagulant after 3 days for an unknown reason (no adverse events related to FRAGMIN were documented) and 11 patients (69%) were reported to have completed their course of therapy.
|
Table 21 |
||
|
Median Starting and Maintenance Doses of FRAGMIN (units/kg) in Neonatal Cohort (N = 16) |
||
|
All study patients (N=16) |
Start Dose (units/kg); given BID |
Maintenance Dose (units/kg); given BID |
|
Median (Range) |
154.5 (100, 475) units/kg |
345 (96, 723) units/kg |
16. How is Fragmin supplied
FRAGMIN (dalteparin sodium) injection, USP, is a sterile, aqueous, clear, colorless or straw-colored solution available as follows:
|
|||
|
Presentation |
Strength |
Package Size |
NDC Number |
|
Single-dose prefilled syringe* |
2,500 units / 0.2 mL |
10 Syringes |
0069-0195-02 |
|
5,000 units / 0.2 mL |
10 Syringes |
0069-0196-02 |
|
|
7,500 units / 0.3 mL |
10 Syringes |
0069-0206-02 |
|
|
Single-dose graduated syringe† |
10,000 units / mL |
10 Syringes |
0069-0217-02 |
|
Single-dose prefilled syringe* |
12,500 units / 0.5 mL |
10 Syringes |
0069-0220-02 |
|
15,000 units / 0.6 mL |
10 Syringes |
0069-0223-02 |
|
|
18,000 units / 0.72 mL |
10 Syringes |
0069-0228-02 |
|
|
Multiple-dose vial |
95,000 units / 3.8 mL |
3.8 mL vial |
0069-0232-01 |
|
Single-dose vial |
10,000 units / 4 mL |
10 vials |
0069-0253-10 |
Store at 20°C to 25°C (68°F to 77°F); excursion permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Latex Allergy: The needle shield of the prefilled syringe may contain natural rubber latex [see Dosage and Administration (2.7)].
17. Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Risk of Hemorrhage including Spinal/Epidural Hematomas
If patients have had neuraxial anesthesia or spinal puncture, and particularly, if they are taking concomitant NSAIDs, platelet inhibitors, or other anticoagulants, inform the patients to watch for signs and symptoms of spinal or epidural hematoma, such as tingling, numbness (especially in the lower limbs) and muscular weakness. If any of these symptoms occur the patient should contact his or her physician immediately.
Additionally, the use of aspirin and other NSAIDs may enhance the risk of hemorrhage. Discontinue their use prior to FRAGMIN therapy whenever possible; if co-administration is essential, the patient's clinical and laboratory status should be closely monitored [see Drug Interactions (7)].
Inform Patients:
- •
- of the instructions for injecting FRAGMIN if their therapy is to continue after discharge from the hospitals.
- •
- it may take them longer than usual to stop bleeding.
- •
- they may bruise and/or bleed more easily when they are treated with FRAGMIN.
- •
- they should report any unusual bleeding, bruising, or signs of thrombocytopenia (such as a rash of dark red spots under the skin) to their physician [see Warnings and Precautions (5.1, 5.2)].
- •
- to tell their physicians and dentists they are taking FRAGMIN and/or any other product known to affect bleeding before any surgery is scheduled and before any new drug is taken [see Warnings and Precautions (5.1)].
- •
- to tell their physicians and dentists of all medications they are taking, including those obtained without a prescription, such as aspirin or other NSAIDs [see Drug Interactions (7)].
- •
- Risks are associated with benzyl alcohol in neonates, infants, and pregnant women [see Warnings and Precautions (5.3) and Use in Specific Populations (8.1)].
- •
- to tell their physicians and care givers if they are allergic to natural rubber latex [see Dosage and Administration (2.7)].
This product's labeling may have been updated. For the most recent prescribing information, please visit www.pfizer.com.
FRAGMIN is a registered trademark of Pfizer Health AB.
LAB-0058-25.0
PRINCIPAL DISPLAY PANEL - 0.2 mL Syringe Label - 0195
0.2 mL
NDC 0069-0195-01
Fragmin®
(dalteparin sodium) Injection, USP
2,500 IU per 0.2 mL
For subcutaneous injection
Single-dose only
Pfizer Labs
Division of Pfizer Inc.
NY, NY 10017
Rx only
PAA193770
PRINCIPAL DISPLAY PANEL - 0.2 mL Syringe Blister Pack Label - 0195
0.2 mL Single-dose prefilled syringe
NDC 0069-0195-01
Fragmin® (dalteparin sodium) Injection, USP
2,500 IU per 0.2 mL
For subcutaneous injection
Rx only
PAA194311
PRINCIPAL DISPLAY PANEL - 0.2 mL Syringe Carton - 0195
NDC 0069-0195-02
Contains 10 of NDC 0069-0195-01
10 x 0.2 mL Single-dose prefilled syringes,
preassembled with needle guards
Fragmin®
(dalteparin sodium) Injection, USP
2,500 IU per 0.2 mL*
For subcutaneous injection
Sterile
Pfizer Injectables
Rx only
PRINCIPAL DISPLAY PANEL - 0.2 mL Syringe Label - 0196
0.2 mL
NDC 0069-0196-01
Fragmin®
(dalteparin sodium) Injection, USP
5,000 IU per 0.2 mL
For subcutaneous injection
Single-dose only
Pfizer Labs
Division of Pfizer Inc.
NY, NY 10017
Rx only
PAA194333
PRINCIPAL DISPLAY PANEL - 0.2 mL Syringe Blister Pack Label - 0196
0.2 mL Single-dose prefilled syringe
NDC 0069-0196-01
Fragmin® (dalteparin sodium) Injection, USP
5,000 IU per 0.2 mL
For subcutaneous injection
Rx only
PAA194306
PRINCIPAL DISPLAY PANEL - 0.2 mL Syringe Carton - 0196
NDC 0069-0196-02
Contains 10 of NDC 0069-0196-01
10 x 0.2 mL Single-dose prefilled syringes,
preassembled with needle guards
Fragmin®
(dalteparin sodium) Injection, USP
5,000 IU per 0.2 mL*
For subcutaneous injection
Sterile
Pfizer Injectables
Rx only
PRINCIPAL DISPLAY PANEL - 0.3 mL Syringe Label - 0206
0.3 mL
NDC 0069-0206-01
Fragmin®
(dalteparin sodium) Injection, USP
7,500 IU per 0.3 mL
For subcutaneous injection
Single-dose only
Pfizer Labs
Division of Pfizer Inc.
NY, NY 10017
Rx only
PAA194716
PRINCIPAL DISPLAY PANEL - 0.3 mL Syringe Blister Pack Label - 0206
0.3 mL Single-dose prefilled syringe
NDC 0069-0206-01
Fragmin® (dalteparin sodium) Injection, USP
7,500 IU per 0.3 mL
For subcutaneous injection
Rx only
PAA194312
PRINCIPAL DISPLAY PANEL - 0.3 mL Syringe Carton - 0206
NDC 0069-0206-02
Contains 10 of NDC 0069-0206-01
10 x 0.3 mL Single-dose prefilled syringes,
preassembled with needle guards
Fragmin®
(dalteparin sodium) Injection, USP
7,500 IU per 0.3 mL*
For subcutaneous injection
Sterile
Pfizer Injectables
Rx only
PRINCIPAL DISPLAY PANEL - 1 mL Syringe Label - 0217
PAA194310
Fragmin® 1 mL
(dalteparin sodium) Injection, USP
10,000 IU
per mL
NDC 0069-0217-01
For subcutaneous
injection
Single-dose only.
Pfizer Labs
Division of Pfizer Inc.
NY, NY 10017
NDC 0069-0217-01
Rx only
PRINCIPAL DISPLAY PANEL - 1 mL Syringe Blister Pack Label - 0217
1 mL Single-dose graduated syringe
NDC 0069-0217-01
Fragmin® (dalteparin sodium) Injection, USP
10,000 IU per mL
For subcutaneous injection
Rx only
PAA194270
PRINCIPAL DISPLAY PANEL - 1 mL Syringe Carton - 0217
NDC 0069-0217-02
Contains 10 of NDC 0069-0217-01
10 x 1 mL Single-dose graduated syringes,
preassembled with needle guards
Fragmin®
(dalteparin sodium) Injection, USP
10,000 IU per mL*
For subcutaneous injection
Sterile
Pfizer Injectables
Rx only
PRINCIPAL DISPLAY PANEL - 0.5 mL Syringe Label - 0220
0.5 mL NDC 0069-0220-01
Fragmin®
(dalteparin sodium) Injection, USP
12,500 IU per 0.5 mL
For subcutaneous injection
Single-dose only
Pfizer Labs
Division of Pfizer Inc.
NY, NY 10017
Rx only
PAA194717
PRINCIPAL DISPLAY PANEL - 0.5 mL Syringe Blister Pack Label - 0220
0.5 mL Single-dose prefilled syringe
NDC 0069-0220-01
Fragmin® (dalteparin sodium) Injection, USP
12,500 IU per 0.5 mL
For subcutaneous injection
Rx only
PAA194307
PRINCIPAL DISPLAY PANEL - 0.5 mL Syringe Carton - 0220
NDC 0069-0220-02
Contains 10 of NDC 0069-0220-01
10 x 0.5 mL Single-dose prefilled syringes,
preassembled with needle guards
Fragmin®
(dalteparin sodium) Injection, USP
12,500 IU per 0.5 mL*
For subcutaneous injection
Sterile
Pfizer Injectables
Rx only
PRINCIPAL DISPLAY PANEL - 0.6 mL Syringe Label-0223
0.6 mL NDC 0069-0223-01
Fragmin®
(dalteparin sodium) Injection, USP
15,000 IU per 0.6 mL
For subcutaneous injection
Single-dose only
Pfizer Labs
Division of Pfizer Inc.
NY, NY 10017
Rx only
PAA194331
PRINCIPAL DISPLAY PANEL - 0.6 mL Syringe Blister Pack Label - 0223
0.6 mL Single-dose prefilled syringe
NDC 0069-0223-01
Fragmin® (dalteparin sodium) Injection, USP
15,000 IU per 0.6 mL
For subcutaneous injection
Rx only
PAA194305
PRINCIPAL DISPLAY PANEL - 0.6 mL Syringe Carton - 0223
NDC 0069-0223-02
Contains 10 of NDC 0069-0223-01
10 x 0.6 mL Single-dose prefilled syringes,
preassembled with needle guards
Fragmin®
(dalteparin sodium) Injection,USP
15,000 IU per 0.6 mL*
For subcutaneous injection
Sterile
Pfizer Injectables
Rx only
PRINCIPAL DISPLAY PANEL - 0.72 mL Syringe Label - 0228
0.72 mL
NDC 0069-0228-01
Fragmin®
(dalteparin sodium) Injection, USP
18,000 IU per 0.72 mL
For subcutaneous injection
Single-dose only
Pfizer Labs
Division of Pfizer Inc.
NY, NY 10017
Rx only
PAA194332
PRINCIPAL DISPLAY PANEL - 0.72 mL Syringe Blister Pack Label - 0228
0.72 mL Single-dose prefilled syringe
NDC 0069-0228-01
Fragmin® (dalteparin sodium) Injection, USP
18,000 IU per 0.72 mL
For subcutaneous injection
Rx only
PAA194308
PRINCIPAL DISPLAY PANEL - 0.72 mL Syringe Carton - 0228
NDC 0069-0228-02
Contains 10 of NDC 0069-0228-01
10 x 0.72 mL Single-dose prefilled syringes,
preassembled with needle guards
Fragmin®
(dalteparin sodium) Injection, USP
18,000 IU per 0.72 mL*
For subcutaneous injection
Sterile
Pfizer Injectables
Rx only
PRINCIPAL DISPLAY PANEL - 95,000 IU Vial Label - 0232
NDC 0069-0232-01
3.8 mL multiple-dose vial
Fragmin®
(dalteparin sodium) Injection, USP
95,000 IU/3.8 mL
(25,000 IU/mL)
For subcutaneous injection
Rx only
PRINCIPAL DISPLAY PANEL - 95,000 IU Vial Carton - 0232
NDC 0069-0232-01
3.8 mL multiple-dose vial
Fragmin®
(dalteparin sodium) Injection, USP
95,000 IU/3.8 mL
(25,000 IU/mL)
For subcutaneous injection
Sterile
Pfizer Injectables
Rx only
| FRAGMIN
dalteparin sodium injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| FRAGMIN
dalteparin sodium injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| FRAGMIN
dalteparin sodium injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| FRAGMIN
dalteparin sodium injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| FRAGMIN
dalteparin sodium injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| FRAGMIN
dalteparin sodium injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| FRAGMIN
dalteparin sodium injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| FRAGMIN
dalteparin sodium injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| FRAGMIN
dalteparin sodium injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Pfizer Laboratories Div Pfizer Inc (134489525) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Pfizer Manufacturing Belgium NV | 370156507 | ANALYSIS(0069-0195, 0069-0196, 0069-0206, 0069-0217, 0069-0220, 0069-0223, 0069-0228, 0069-0232, 0069-0253) , MANUFACTURE(0069-0195, 0069-0196, 0069-0206, 0069-0217, 0069-0220, 0069-0223, 0069-0228, 0069-0232, 0069-0253) , PACK(0069-0195, 0069-0196, 0069-0206, 0069-0217, 0069-0220, 0069-0223, 0069-0228, 0069-0232, 0069-0253) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Pfizer Health AB | 354433591 | ANALYSIS(0069-0195, 0069-0196, 0069-0206, 0069-0217, 0069-0220, 0069-0223, 0069-0228, 0069-0232, 0069-0253) , API MANUFACTURE(0069-0195, 0069-0196, 0069-0206, 0069-0217, 0069-0220, 0069-0223, 0069-0228, 0069-0232, 0069-0253) | |
More about Fragmin (dalteparin)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (5)
- Side effects
- Dosage information
- During pregnancy
- Drug class: heparins
- Breastfeeding
- En español