Acetazolamide Tablets: Package Insert / Prescribing Info
Package insert / product label
Dosage form: tablet
Drug classes: Carbonic anhydrase inhibitor anticonvulsants, Carbonic anhydrase inhibitors
Medically reviewed by Drugs.com. Last updated on Sep 2, 2025.
On This Page
Acetazolamide Tablets Description
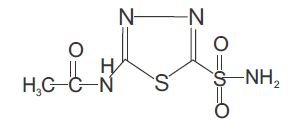
Acetazolamide, an inhibitor of the enzyme carbonic anhydrase is a white to faintly yellowish white crystalline, odorless powder, very slightly soluble in water, sparingly soluble in practically boiling water and slightly soluble in alcohol. The chemical name for acetazolamide is N-(5-Sulfamoyl-1,3,4-thiadiazol-2-yl) acetamide and has the following chemical structure:
Molecular Formula: C4H6N4O3S2
Acetazolamide is available as oral tablets containing 125 mg and 250 mg of acetazolamide, respectively, and the following inactive ingredients: corn starch, gelatin, glycerin, lactose monohydrate, magnesium stearate, sodium starch glycolate (Potato) and talc.
Acetazolamide Tablets - Clinical Pharmacology
Acetazolamide is a potent carbonic anhydrase inhibitor, effective in the control of fluid secretion (eg, some types of glaucoma), in the treatment of certain convulsive disorders (eg, epilepsy) and in the promotion of diuresis in instances of abnormal fluid retention (eg, cardiac edema).
Acetazolamide is not a mercurial diuretic. Rather, it is a nonbacteriostatic sulfonamide possessing a chemical structure and pharmacological activity distinctly different from the bacteriostatic sulphonamides.
Acetazolamide is an enzyme inhibitor that acts specifically on carbonic anhydrase, the enzyme that catalyzes the reversible reaction involving the hydration of carbon dioxide and the dehydration of carbonic acid. In the eye, this inhibitory action of acetazolamide decreases the secretion of aqueous humor and results in a drop in intraocular pressure, a reaction considered desirable in cases of glaucoma and even in certain nonglaucomatous conditions. Evidence seems to indicate that acetazolamide has utility as an adjuvant in the treatment of certain dysfunctions of the central nervous system (eg, epilepsy). Inhibition of carbonic anhydrase in this area appears to retard abnormal, paroxysmal, excessive discharge from central nervous system neurons. The diuretic effect of acetazolamide is due to its action in the kidney on the reversible reaction involving hydration of carbon dioxide and dehydration of carbonic acid. The result is renal loss of HCO3 ion, which carries out sodium, water, and potassium. Alkalinization of the urine and promotion of diuresis are thus affected. Alteration in ammonia metabolism occurs due to increased reabsorption of ammonia by the renal tubules as a result of urinary alkalinisation.
Placebo-controlled clinical trials have shown that prophylactic administration of acetazolamide at a dose of 250 mg every eight to 12 hours (or a 500 mg controlled-release capsule once daily) before and during rapid ascent to altitude results in fewer and/or less severe symptoms (such as headache, nausea, shortness of breath, dizziness, drowsiness, and fatigue) of acute mountain sickness (AMS). Pulmonary function (eg, minute ventilation, expired vital capacity, and peak flow) is greater in the acetazolamide treated group, both in subjects with AMS and asymptomatic subjects. The acetazolamide treated climbers also had less difficulty in sleeping.
Indications and Usage for Acetazolamide Tablets
For adjunctive treatment of: edema due to congestive heart failure; drug-induced edema; centrencephalic epilepsies (petit mal, unlocalized seizures); chronic simple (open-angle) glaucoma, secondary glaucoma, and preoperatively in acute angle-closure glaucoma where delay of surgery is desired in order to lower intraocular pressure. Acetazolamide Tablets are also indicated for the prevention or amelioration of symptoms associated with acute mountain sickness in climbers attempting rapid ascent and in those who are very susceptible to acute mountain sickness despite gradual ascent.
Contraindications
Hypersensitivity to acetazolamide or any excipients in the formulation. Since acetazolamide is a sulfonamide derivative, cross sensitivity between acetazolamide, sulfonamides and other sulfonamide derivatives is possible.
Acetazolamide therapy is contraindicated in situations in which sodium and/or potassium blood serum levels are depressed, in cases of marked kidney and liver disease or dysfunction, in suprarenal gland failure, and in hyperchloremic acidosis. It is contraindicated in patients with cirrhosis because of the risk of development of hepatic encephalopathy.
Long-term administration of acetazolamide is contraindicated in patients with chronic noncongestive angle-closure glaucoma since it may permit organic closure of the angle to occur while the worsening glaucoma is masked by lowered intraocular pressure.
Warnings
Fatalities have occurred, although rarely, due to severe reactions to sulfonamides including Stevens-Johnson syndrome, toxic epidermal necrolysis, fulminant hepatic necrosis, agranulocytosis, aplastic anemia, and other blood dyscrasias, and anaphylaxis. Sensitizations may recur when a sulfonamide is readministered irrespective of the route of administration. If signs of hypersensitivity or other serious reactions occur, discontinue use of this drug.
Caution is advised for patients receiving concomitant high-dose aspirin and acetazolamide, as anorexia, tachypnea, lethargy, metabolic acidosis, coma, and death have been reported.
Adverse Reactions/Side Effects
Body as a whole: Headache, malaise, fatigue, fever, pain at injection site, flushing, growth retardation in children, flaccid paralysis, anaphylaxis
Digestive: Gastrointestinal disturbances such as nausea, vomiting, diarrhea
Hematological/Lymphatic: Blood dyscrasias such as aplastic anemia, agranulocytosis, leukopenia, thrombocytopenia, thrombocytopenic purpura, melena
Hepato-biliary disorders: Abnormal liver function, cholestatic jaundice, hepatic insufficiency, fulminant hepatic necrosis
Metabolic/Nutritional: Metabolic acidosis, electrolyte imbalance, including hypokalemia, hyponatremia, osteomalacia with long-term phenytoin therapy, loss of appetite, taste alteration, hyper/hypoglycaemia
Nervous: Drowsiness, paraesthesia (including numbness and tingling of extremities and face), depression, excitement, ataxia, confusion, convulsions, dizziness
Skin: Allergic skin reactions including urticaria, photosensitivity, Stevens-Johnson syndrome, toxic epidermal necrolysis
Otologic: Hearing disturbances, tinnitus
Eye Disorders: choroidal effusion, choroidal detachment, transient myopia. Transient myopia is the result of forward movement of the ciliary body leading to a narrowing of the angle.
Urogenital: Crystalluria, increased risk of nephrolithiasis with long-term therapy, hematuria, glycosuria, renal failure, polyuria
To report SUSPECTED ADVERSE REACTIONS, contact Strides Pharma Inc. at 1-877-244-9825 or go to www.strides.com or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Related/similar drugs
Overdosage
No specific antidote is known. Treatment should be symptomatic and supportive.
Electrolyte imbalance, development of an acidotic state, and central nervous effects might be expected to occur. Serum electrolyte levels (particularly potassium) and blood pH levels should be monitored.
Supportive measures are required to restore electrolyte and pH balance. The acidotic state can usually be corrected by the administration of bicarbonate.
Despite its high intraerythrocytic distribution and plasma protein binding properties, acetazolamide is dialyzable. This may be particularly important in the management of acetazolamide overdosage when complicated by the presence of renal failure.
Acetazolamide Tablets Dosage and Administration
Glaucoma: Acetazolamide should be used as an adjunct to the usual therapy. The dosage employed in the treatment of chronic simple(open-angle) glaucoma ranges from 250 mg to 1 g of acetazolamide per 24 hours, usually in divided doses for amounts over 250 mg. It has usually been found that a dosage in excess of 1 g per 24 hours does not produce an increased effect. In all cases, the dosage should be adjusted with careful individual attention both to symptomatology and ocular tension. Continuous supervision by a physician is advisable.
In treatment of secondary glaucoma and in the preoperative treatment of some cases of acute congestive (closed-angle) glaucoma, the preferred dosage is 250 mg every four hours, although some cases have responded to 250 mg twice daily on short-term therapy. In some acute cases, it may be more satisfactory to administer an initial dose of 500 mg followed by 125 mg or 250 mg every four hours depending on the individual case. Intravenous therapy may be used for rapid relief of ocular tension in acute cases. A complementary effect has been noted when acetazolamide has been used in conjunction with miotics or mydriatics as the case demanded.
Epilepsy: It is not clearly known whether the beneficial effects observed in epilepsy are due to direct inhibition of carbonic anhydrase in the central nervous system or whether they are due to the slight degree of acidosis produced by the divided dosage. The best results to date have been seen in petit mal in pediatric patients. Good results, however, have been seen in patients, both pediatric patients and adult, in other types of seizures such as grand mal, mixed seizure patterns, myoclonic jerk patterns, etc. The suggested total daily dose is 8 to 30 mg per kg in divided doses. Although some patients respond to a low dose, the optimum range appears to be from 375 to 1000 mg daily. However, some investigators feel that daily doses in excess of 1 g do not produce any better results than a 1 g dose. When acetazolamide tablets are given in combination with other anticonvulsants, it is suggested that the starting dose should be 250 mg once daily in addition to the existing medications. This can be increased to levels as indicated above.
The change from other medications to acetazolamide should be gradual and in accordance with usual practice in epilepsy therapy.
Congestive Heart Failure: For diuresis in congestive heart failure, the starting dose is usually 250 to 375 mg once daily in the morning (5 mg/kg). If, after an initial response, the patient fails to continue to lose edema fluid, do not increase the dose but allow for kidney recovery by skipping medication for a day. Acetazolamide tablets yield best diuretic results when given on alternate days, or for two days alternating with a day of rest.
Failures in therapy may be due to overdosage or too frequent dosage. The use of acetazolamide does not eliminate the need for other therapy such as digitalis, bed rest, and salt restriction.
Drug-Induced Edema: Recommended dosage is 250 to 375 mg of acetazolamide once a day for one or two days, alternating with a day of rest.
Acute Mountain Sickness: Dosage is 500 mg to 1000 mg daily, in divided doses using tablets or sustained-release capsules as appropriate. In circumstances of rapid ascent, such as in rescue or military operations, the higher dose level of 1000 mg is recommended. It is preferable to initiate dosing 24 to 48 hours before ascent and to continue for 48 hours while at high altitude, or longer as necessary to control symptoms.
Note: The dosage recommendations for glaucoma and epilepsy differ considerably from those for congestive heart failure, since the first two conditions are not dependent upon carbonic anhydrase inhibition in the kidney which requires intermittent dosage if it is to recover from the inhibitory effect of the therapeutic agent.
Interference with Laboratory Tests
Sulfonamides may give false negative or decreased values for urinary phenolsulfonphthalein and phenol red elimination values for urinary protein, serum non-protein and for serum uric acid. Acetazolamide may produce an increased level of crystals in the urine.
Acetazolamide interferes with the HPLC method of assay for theophylline. Interference with the theophylline assay by acetazolamide depends on the solvent used in the extraction; acetazolamide may not interfere with other assay methods for theophylline.
How is Acetazolamide Tablets supplied
Acetazolamide Tablets USP are supplied as follows:
125 mg – White to off white, round, flat, uncoated tablet, debossed as "S;571" on one side and scored in half on other side
NDC 64380-833-06 - Bottle of 100
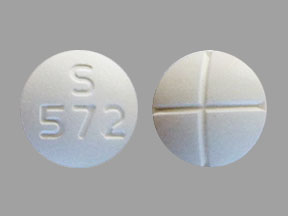
250 mg – White to off white, round, biconvex, uncoated tablet, debossed as "S;572" on one side and scored in quarters on other side
NDC 64380-834-06 - Bottle of 100
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Manufactured by:
Strides Pharma Science Ltd.
Puducherry - 605 014, India.
Distributed by:
Strides Pharma Inc
East Brunswick, NJ 08816
Revised: 10/2024
| ACETAZOLAMIDE
acetazolamide tablet |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| ACETAZOLAMIDE
acetazolamide tablet |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Strides Pharma Science Limited (650738743) |
More about acetazolamide
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (192)
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Drug class: carbonic anhydrase inhibitor anticonvulsants
- Breastfeeding
- En español