Glizigen Skin Protectant
Dosage form: spray
Ingredients: GLYCERIN 0.5mg in 1mL
Labeler: Catalysis, SL
NDC code: 64539-004
Medically reviewed by Drugs.com. Last updated on Jun 18, 2025.
Glycerin 0.5%................Skin Protectant
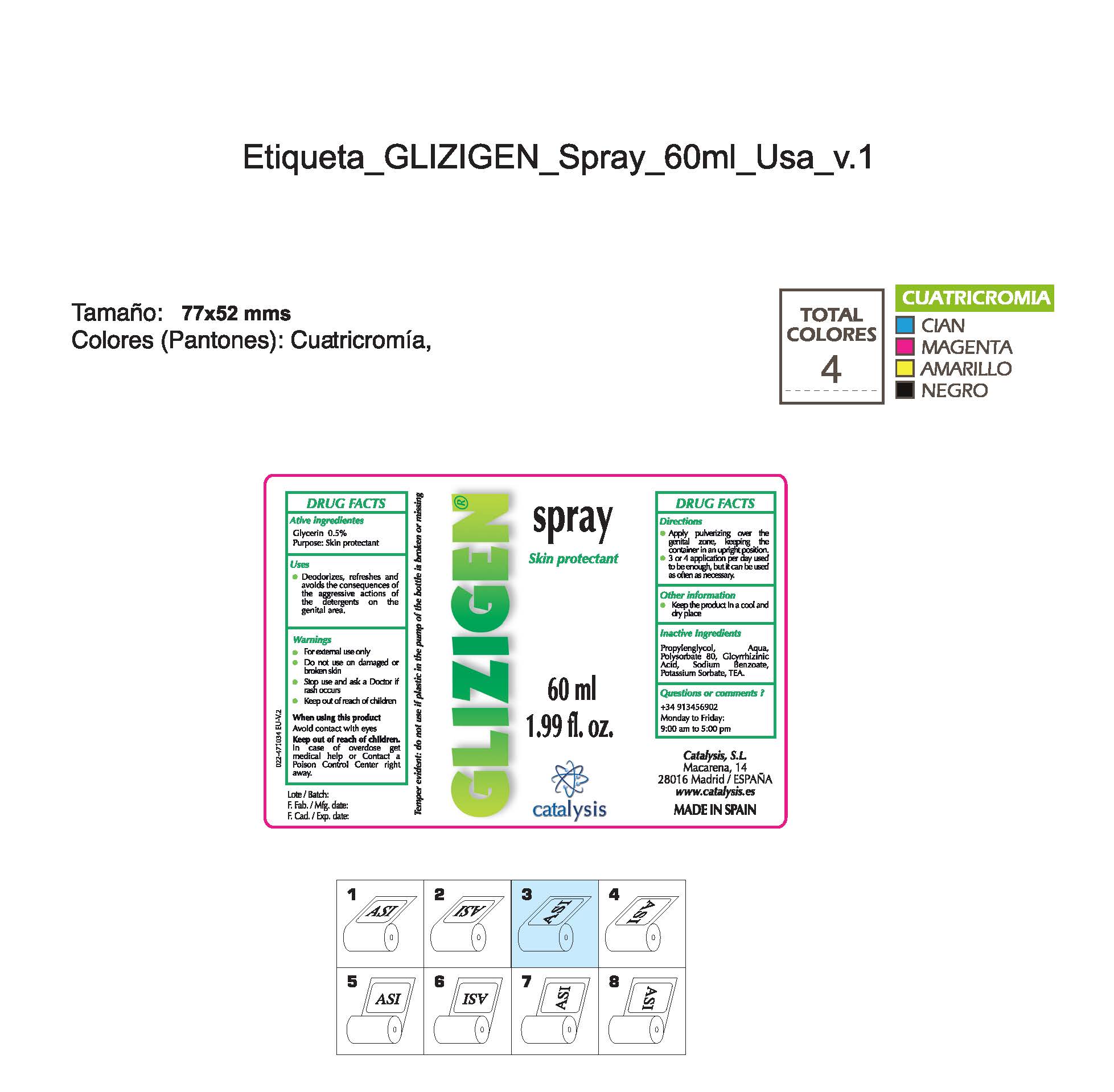
- Stop use and ask a doctor if rash occurs
- Children under 6 months: ask a doctor
- Do not use on damaged or broken skin.
- Keep out of reach of children
- For external use only.
- Do not use on damaged or broken skin.
- When using this product keep our of the eyes. Rinse with water to remove.
- Stop use and ask a doctor if rash occurs.
- Keep out of reach of children
- Children under 6 months: as a doctor
- + 34 913456902 M-F 9:00 am to 5:00 pm
- deodorizes, refreshes and avoids the consequences of the agressive actions of the detergents in the genital areas
- apply pulverizing over the genital zone keeping the container in an upright position
- 3 or 4 applications per day used to be anough, but it can be used as often as necessary
- apply pulverizing over the genital zone keeping the container in an upright position
- 3 or 4 applications per day used to be anough, but it can be used as often as necessary
Propyleneglycol, water, Polysorbate 80, Glycyrrhizinic Acid, Sodium Benzoate, Potassium Sorbate, TEA

| GLIZIGEN SKIN PROTECTANT
glycerin spray |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Catalysis, SL (862795119) |
| Registrant - Catalysis, SL (862795119) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Catalysis, SL | 862795119 | manufacture(64539-004) | |
Document Id: 530af5d6-debd-487a-e054-00144ff8d46c
Set id: 4ef6dae3-dbf2-25bc-e054-00144ff88e88
Version: 2
Catalysis, SL
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
