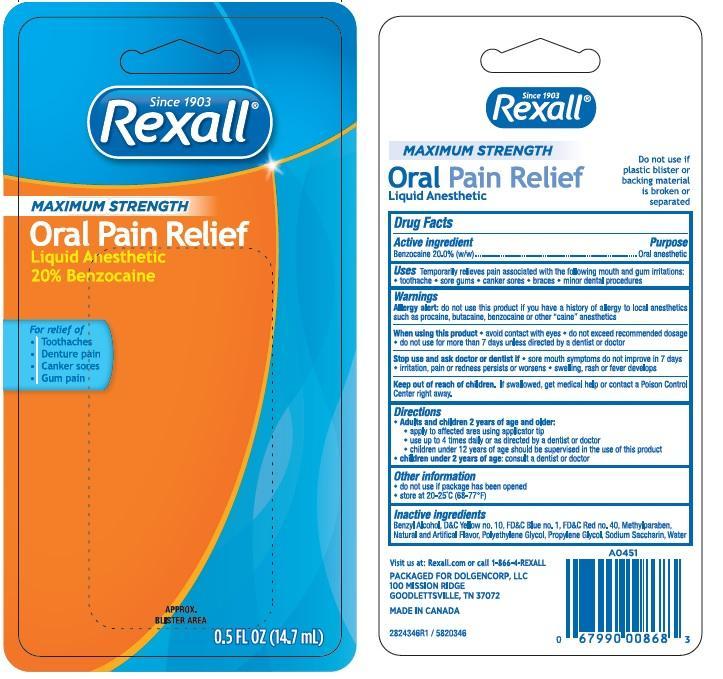
Rexall Maximum Strength
Dosage form: liquid
Ingredients: BENZOCAINE 20g in 100mL
Labeler: Rexall
NDC code: 55910-346
Medically reviewed by Drugs.com. Last updated on Dec 30, 2024.
Benzocaine 20.0% ..................................Purpose: Oral pain reliever
Temporarily relieves pain associated with the following mouth irritations toothache sore gums canker sores braces minor dental procedures
Allergy alert: do not use this product if you have a history of allergy to local anesthetics such as procaine, butacaine, benzocaine or other “caine” anesthetics
When using this product
- avoid contact with eyes
- do not exceed recommended dosage
- Do not use more than directed for more than 7 days unless directed by a dentist or doctor.
Stop use and ask a doctor if sore mouth symptoms do not improve in 7 days, swelling, rash or fever develops, irritation, pain or redness persists or worsens swelling rash or fever developes
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Adults and children 2 years of age and older:
- apply to affect4ed area using applicator tip
- use up to 4 times daily or as directed by a dentist or doctor.
- Children under 12 years of age should be supervised in the use of this product.
Children under 2 years of age: ask a dentist or doctor.
Do not use if package has been opened
Store at 20-25°C (68-77°F)
Benzyl Alcohol, D&C Yellow 10, FD&C Blue 1, FD&C Red 40, FD&C Yellow 5, Methylparaben, Flavor, Polyethylene Glycol, Propylene Glycol, Sodium Saccharin, Water
Adults and children 2 years of age and older:
apply to affect4ed area using applicator tip
use up to 4 times daily or as directed by a dentist or doctor.
Children under 12 years of age should be supervised in the use of this product.
Children under 2 years of age: ask a dentist or doctor.
mm1.jpg
mm2.jpg

| REXALL
MAXIMUM STRENGTH
benzocaine liquid |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Rexall (068331990) |
| Registrant - Lornamead (126440440) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| CSR Cosmetic Solutions | 243501959 | manufacture(55910-346), pack(55910-346) | |
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
