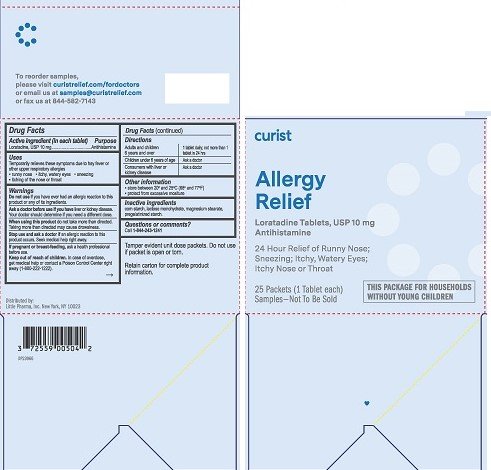
Curist Allergy Relief
Dosage form: tablet
Ingredients: LORATADINE 10mg
Labeler: Little Pharma, Inc.
NDC code: 72559-006
Medically reviewed by Drugs.com. Last updated on Jun 3, 2025.
Drug Facts
Active ingredient (in each tablet)
Loratadine, USP 10 mg
Purpose
Antihistamine
Uses
Temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- itchy, watery eyes
- sneezing
- itching of the nose or throat
Warnings
Do not use if you have ever had an allergic reaction to this product or any of its ingredients.
Ask a doctor before you if you have liver or kidney disease. Your doctor should determine if you need a different dose.
When using this product do not take more than directed. Taking more than directed may cause drowsiness.
Stop use and ask a doctor if an allergic reaction to this product occurs. Seek medical help right away.
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Directions
|
Adults and children 6 years and over |
1 tablet daily; not more than 1 tablet in 24 hours |
|
Children under 6 years of age |
Ask a doctor |
|
Consumers with liver and kidney disease |
Ask a doctor |
Other information
- store between 20° and 25°C (68° and 77°F)
- protect from excessive moisture
Inactive ingredients
corn starch, lactose monohydrate, magnesium stearate, pregelatinized starch
Questions or comments? Call 1-844-243-1241
Tamper evident unit dose packets. Do not use if packet is open or torn.
Retain carton for complete product information.
Distributed by:
Little Pharma, Inc.
New York, NY 10023
curist
Allergy Relief
Loratadine Tablets, USP 10 mg
Antihistamine
24 Hour Relief of Runny Nose; Sneezing; Itchy, Watery Eyes; Itchy Nose or Throat
25 Packets (1 Tablet each)
Samples - Not To Be Sold
THIS PACKAGE FOR HOUSEHOLDS WITHOUT YOUNG CHILDREN
| CURIST ALLERGY RELIEF
loratadine tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Little Pharma, Inc. (074328189) |
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
