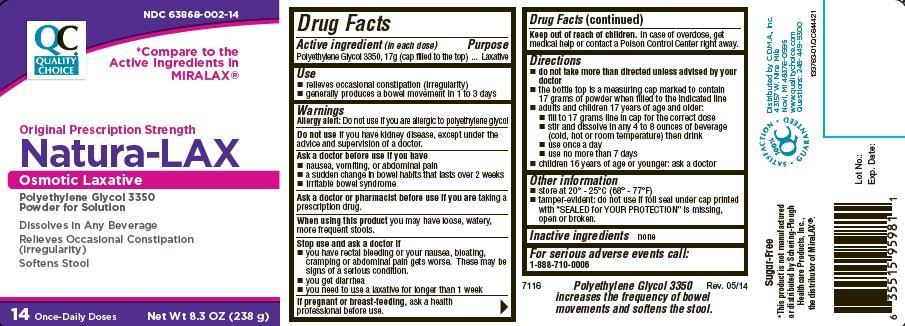
NATURA-LAX
Dosage form: powder, for solution
Ingredients: POLYETHYLENE GLYCOL 3350 17g in 17g
Labeler: Chain Drug Marketing Association
NDC code: 63868-002
Medically reviewed by Drugs.com. Last updated on Apr 14, 2025.
Polyethylene Glycol 3350, 17g (cap filled to the top)
Laxative
- relieves occasional constipation (irregularity)
- generally produces a bowel movement in 1 to 3 days
Allergy alert: Do not use if you are allergic to polyethylene glycol
Do not use if you have kidney disease, except under the advice and supervision of a doctor
Ask a doctor before use if you have
- nausea, vomiting or abdominal pain
- a sudden change in bowel habits that lasts over 2 weeks
- irritable bowel syndrome
Ask a doctor or pharmacist before use if you are taking a prescription drug
When using this product you may have loose, watery, more frequent stools
Stop use and ask a doctor if
- you have rectal bleeding or your nausea, bloating, cramping or abdominal pain gets worse. These may be signs of a serious condition.
- you get diarrhea
- you need to use a laxative for longer than 1 week
If pregnant or breast feeding ask a health professional before use.
Keep out of reach of children. In case of overdose , get medical help or contact a Poison Control Center right away.
- do not take more than directed unless advised by your doctor
- the bottle top is a measuring cap marked to contain 17 grams of powder when filled to the indicated line
- adults and children 17 years of age and older:
-
- fill to 17 grams line in cap for the correct dose
-
- stir and dissolve in any 4 to 8 ounces of beverage (cold, hot or room temperature) then drink
-
- use once daily
-
- use no more than 7 days
- children 16 years of age or younger: ask a doctor
- store at 20° - 25°C (68° - 77°F)
- tamper-evident: do not use if foil seal under cap printed with “SEALED for YOUR PROTECTION” is missing, open or broken.
none
1-888-710-0006
NDC 63868-002-14
Quality Choice
compare to the active ingredient in miralax
original prescription strength
Natura-LAX
Osmotic Laxative
polyethylene glycol 3350
powder for solution
Dissolves in Any Beverage
relieves occasional constipation (irregularity)
softens stool
14 once daily doses
net wt 8.3 oz (238 g)
| NATURA-LAX
polyethylene glycol 3350 powder, for solution |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Chain Drug Marketing Association (011920774) |
| Registrant - Geri-Care Pharmaceutical Corp (611196254) |
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
