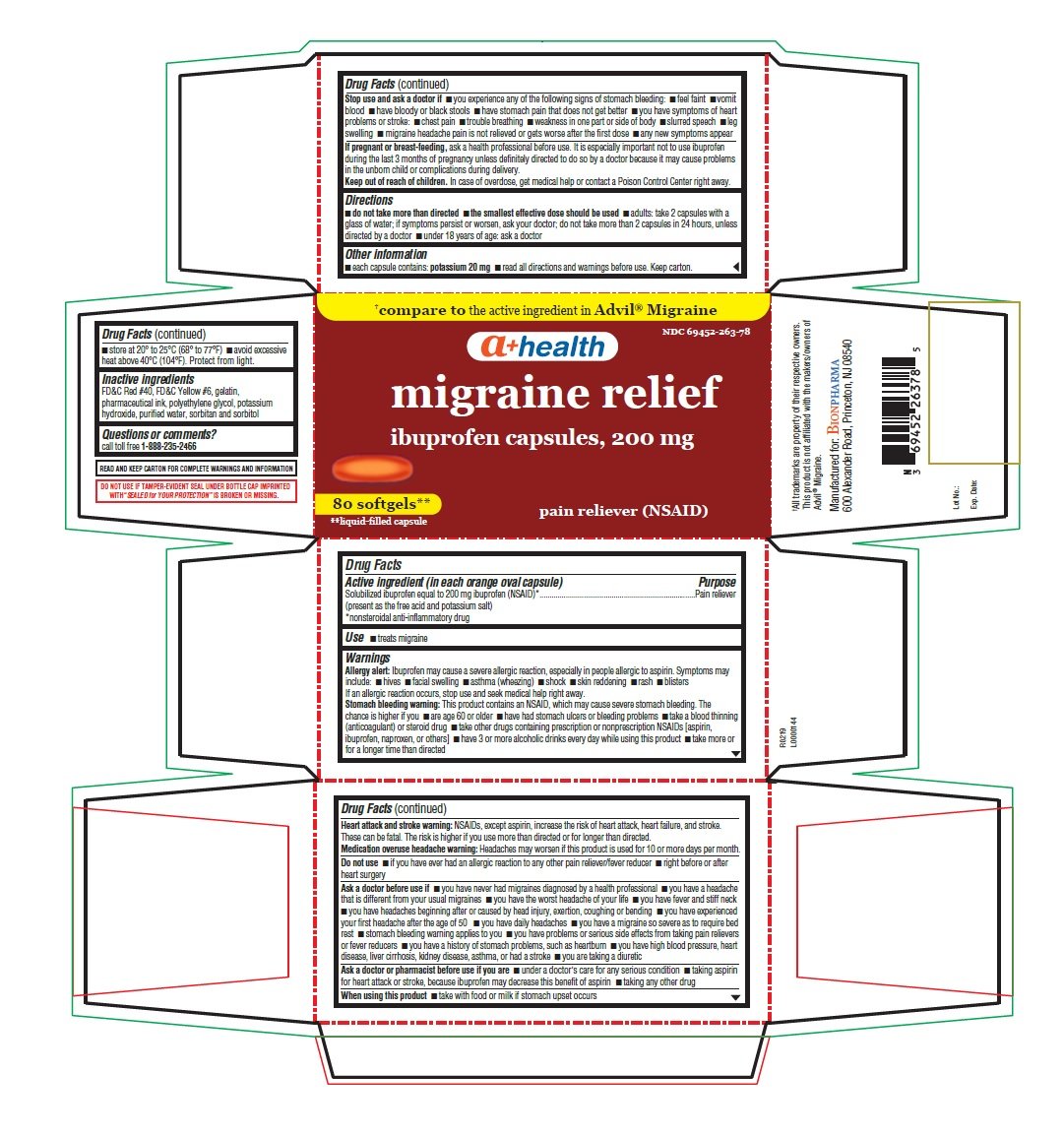
Migraine Relief
Dosage form: capsule, liquid filled
Ingredients: IBUPROFEN 200mg
Labeler: Bionpharma Inc.
NDC code: 69452-263
Medically reviewed by Drugs.com. Last updated on Apr 14, 2025.
Solubilized ibuprofen equal to 200 mg ibuprofen (NSAID)*
(present as the free acid and potassium salt)
*nonsteroidal anti-inflammatory drug
Pain reliever
- treats migraine
Allergy alert: Ibuprofen may cause a severe allergic reaction, especially in people allergic to aspirin. Symptoms may include:
- hives
- facial swelling
- asthma (wheezing)
- shock
- skin reddening
- rash
- blisters
If an allergic reaction occurs, stop use and seek medical help right away.
Stomach bleeding warning: This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you
- are age 60 or older
- have had stomach ulcers or bleeding problems
- take a blood thinning (anticoagulant) or steroid drug
- take other drugs containing prescription or nonprescription NSAIDs [aspirin, ibuprofen, naproxen, or others]
- have 3 or more alcoholic drinks every day while using this product
- take more or for a longer time than directed
Heart attack and stroke warning: NSAIDs, except aspirin, increase the risk of heart attack, heart failure, and stroke. These can be be fatal. The risk is higher if you use more than directed or for longer than directed.
Medication overuse headache warning: Headaches may worsen if this product is used for 10 or more days per month.
- if you have ever had an allergic reaction to any other pain reliever/fever reducer
- right before or after heart surgery
- you have never had migraines diagnosed by a health professional
- you have a headache that is different from your usual migraines
- you have the worst headache of your life
- you have fever and stiff neck
- you have headaches beginning after or caused by head injury, exertion, coughing or bending
- you have experienced your first headache after the age of 50
- you have daily headaches
- you have a migraine so severe as to require bed rest
- stomach bleeding warning applies to you
- you have problems or serious side effects from taking pain relievers or fever reducers
- you have a history of stomach problems, such as heartburn
- you have high blood pressure, heart disease, liver cirrhosis, kidney disease, asthma, or had a stroke
- you are taking a diuretic
- under a doctor's care for any serious condition
- taking aspirin for heart attack or stroke, because ibuprofen may decrease this benefit of aspirin
- taking any other drug
- take with food or milk if stomach upset occurs
- you experience any of the following signs of stomach bleeding:
- feel faint
- vomit blood
- have bloody or black stools
- have stomach pain that does not get better
- you have symptoms of heart problems or stroke:
- chest pain
- trouble breathing
- weakness in one part or side of body
- slurred speech
- leg swelling
- migraine headache pain is not relieved or gets worse after the first dose
- any new symptoms appear
ask a health professional before use. It is especially important not to use ibuprofen during the last 3 months of pregnancy unless definitely directed to do so by a doctor because it may cause problems in the unborn child or complications during delivery.
In case of overdose, get medical help or contact a Poison Control Center right away.
- do not take more than directed
- the smallest effective dose should be used
- adults: take 2 capsules with a glass of water; if symptoms persist or worsen, ask your doctor; do not take more than 2 capsules in 24 hours, unless directed by a doctor
- under 18 years of age: ask a doctor
- each capsule contains: potassium 20 mg
- read all warnings and directions before use. Keep carton.
- store at 20° to 25°C (68° to 77°F)
- avoid excessive heat above 40°C (104°F). Protect from light.
FD&C Red #40, FD&C Yellow #6, gelatin, pharmaceutical ink, polyethylene glycol, potassium hydroxide, purified water, sorbitan and sorbitol
call toll free 1-888-235-2466
READ AND KEEP CARTON FOR COMPLETE WARNINGS AND INFORMATION
DO NOT USE IF TAMPER-EVIDENT SEAL UNDER BOTTLE CAP IMPRINTED WITH “SEALED for YOUR PROTECTION” IS BROKEN OR MISSING.
†All trademarks are property of their respective owners. This product is not affiliated with the makers/owners of Advil® Migraine.
Manufactured for:BIONPHARMA
600 Alexander Road, Princeton, NJ 08540
R0219
L0000144
| MIGRAINE RELIEF
ibuprofen capsule, liquid filled |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Bionpharma Inc. (079637826) |
| Registrant - Bionpharma Inc. (079637826) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Patheon Softgels Inc. | 002193829 | manufacture(69452-263) | |
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.