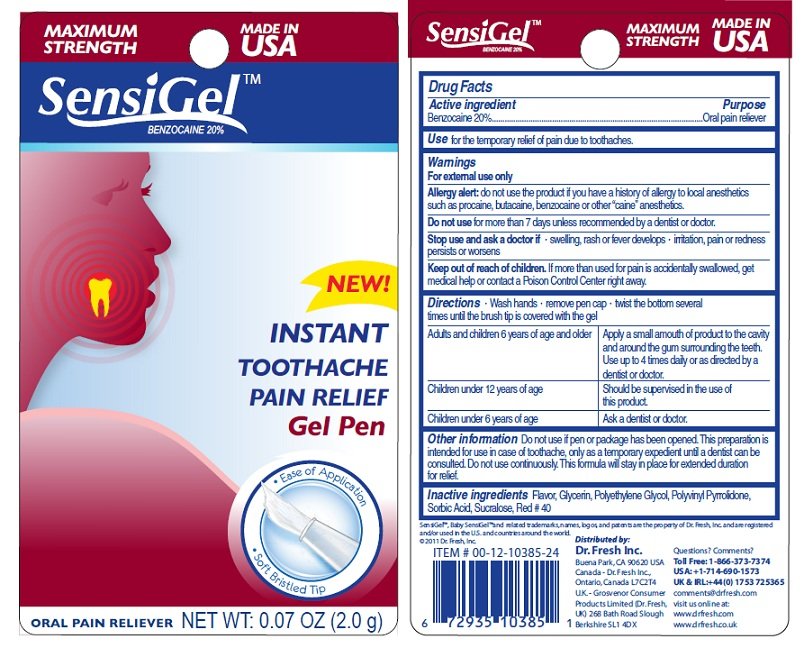
SensiGel
Dosage form: gel
Ingredients: BENZOCAINE 20g in 100g
Labeler: Dr. Fresh LLC
NDC code: 64893-385
Medically reviewed by Drugs.com. Last updated on Jan 6, 2025.
Benzocaine 20%
Oral Pain Reliever
for the temporary relief of pain due to toothaches.
For external use only.
Allergy Alert: do not use the product if you havd a history of allergy to local anesthetics such as procaine, butacaine, benzocaine or other "caine" anesthetics.
for more than 7 days unless recommended by a dentist or doctor.
swelling, rash or fever develops - irritation, pain or redness persists or worsens
If more than used for pain is accidentally swallowed, get medical help or contact a Poison Control Center right away.
Wash hands - remove pen cap - twist the bottom several times until the brush tip is covered with the gel.
Adults and children 6 years of age and older: Apply a small amount of product to the cavity and around the gum surrounding the teeth. Use up to 4 times daily or as directed by a dentist or doctor. Children under 12 years of age - Should be supervised in the use of this product. Children under 6 years of age: Ask a dentist or doctor.
Do not use if pen or package has been opened. This preparation is intended for use in case of toothache, only as a temporary expedient until a dentist can b consulted. Do not use continuously. This formula will stay in place for an extended duration for relief.
Flavor, Glycerin, Polyethylene Glycol, Polyvinyl Pyrrolidone, Sorbic Acid, Sucralose, Red No. 40
| SENSIGEL
benzocaine gel |
||||||||||||||
|
||||||||||||||
|
||||||||||||||
|
||||||||||||||
|
||||||||||||||
|
||||||||||||||
| Labeler - Dr. Fresh LLC (117376803) |
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.