Zepol Topical Analgesic
Dosage form: ointment
Ingredients: CAMPHOR (SYNTHETIC) 6.55g in 100g, MENTHOL 6.55g in 100g
Labeler: Laboratorios Zepol S.A.
NDC code: 55715-001
Medically reviewed by Drugs.com. Last updated on Oct 14, 2024.
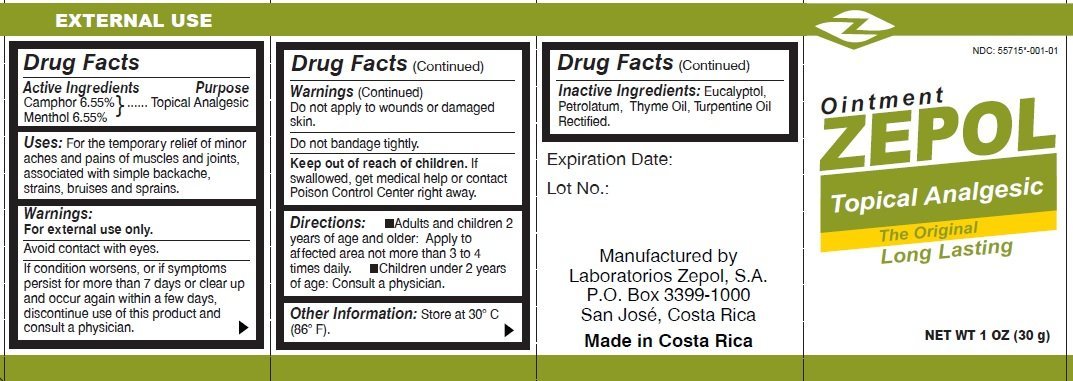
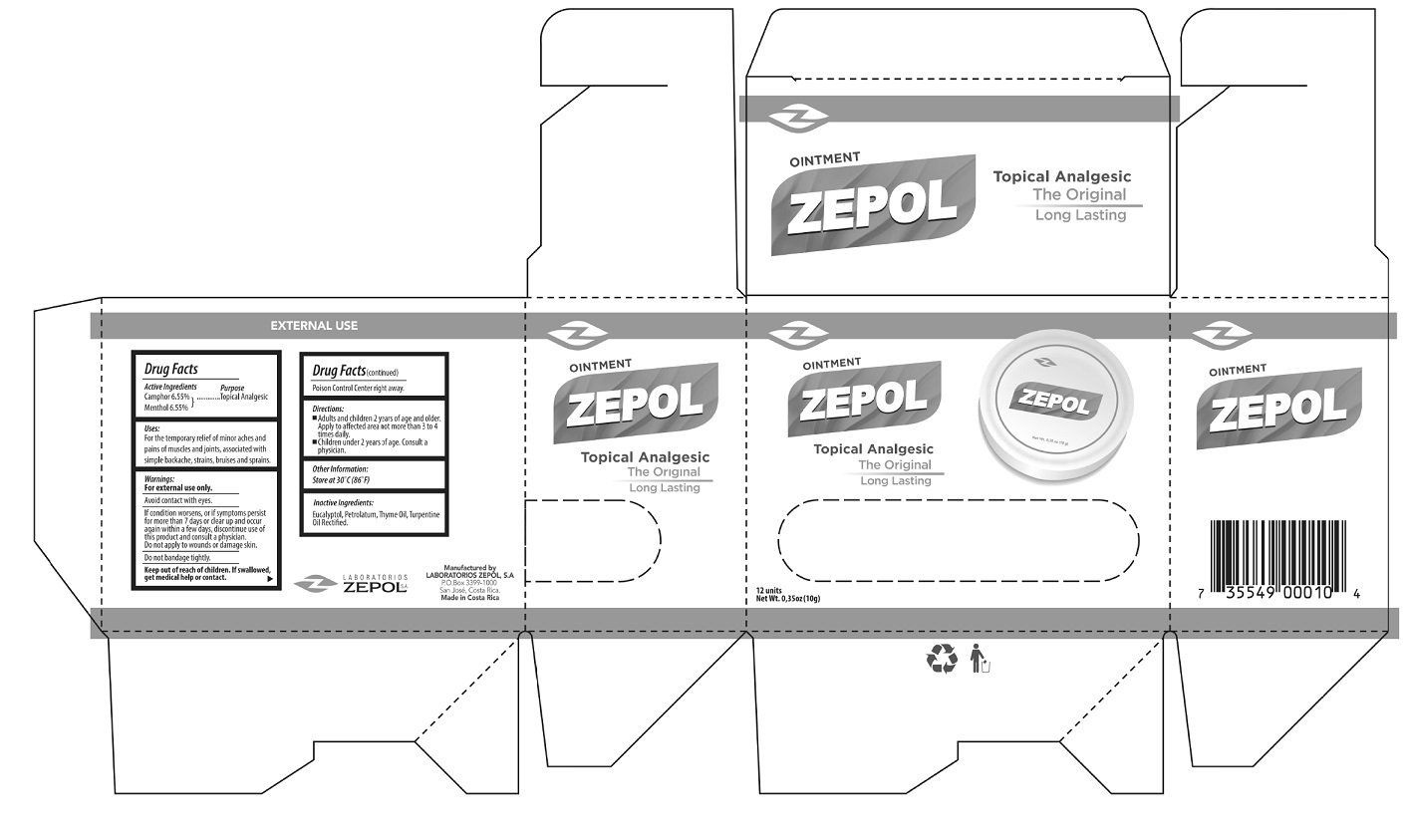
Camphor 6.55%
Menthol 6.55%
Topical Analgesic
For the temporary relief of minor aches and pains of muscles and joints, associated with simple backache, strains, bruises and sprains.
For external use only.
Avoid contact with eyes.
If condition worsens, or if symptoms persist for more than 7 days or clear up and occur again within a few days, discontinue use of this product and consult a physician.
to wounds or damaged skin.
Do not bandage tightly.
If swallowed, get medical help or contact Poison Control Center right away.
-Adults and children 2 years of age and older: Apply to affected area not more than 3 to 4 times daily. -Children under 2 years of age: Consult a physician.
Store at 30 degrees C (86 degrees F).
Eucalyptol, Petrolatum, Thyme Oil, Turpentine Oil Rectified.







| ZEPOL TOPICAL ANALGESIC
camphor, menthol ointment |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| Labeler - Laboratorios Zepol S.A. (853070985) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Laboratorios Zepol S.A | 853070985 | manufacture(55715-001) | |
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
