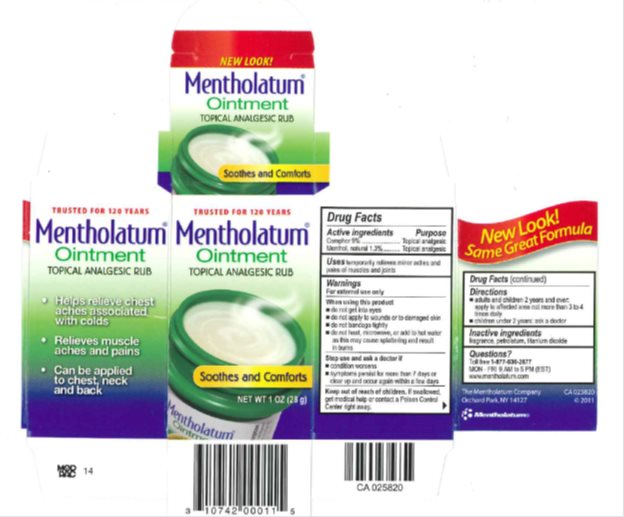
Mentholatum
Dosage form: ointment
Ingredients: CAMPHOR (SYNTHETIC) 90mg in 1g, MENTHOL, UNSPECIFIED FORM 13mg in 1g
Labeler: The Mentholatum Company
NDC code: 10742-0001
Active ingredients
Camphor 9%
Menthol, natural 1.3%
Purpose
Camphor - Topical Analgesic
Menthol, natural - Topical Analgesic
Uses
temporarily relieves minor aches and pains of muscles and joints
Warnings
For external use only
When using this product
- •
- do not heat
- •
- do not microwave
- •
- do not add to hot water or any container where heating water. May cause splattering and result in burns.
- •
- do not get into eyes
- •
- do not apply to wounds or to damaged skin
- •
- do not bandage tightly
Stop use and ask a doctor if
- •
- condition worsens
- •
- symptoms persist for more than 7 days or clear up and occur again within a few days
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- •
- adults and children 2 years and over: apply to affected area not more than 3 to 4 times daily
- •
- children under 2 years: ask a doctor
Inactive ingredients
fragrance, petrolatum, titanium dioxide
Questions?
Toll free 1-877-636-2677 MON-FRI 9AM to 5PM (EST)
www.mentholatum.com
| MENTHOLATUM
camphor, menthol ointment |
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
| Labeler - The Mentholatum Company (002105757) |
Revised: 12/2016
Document Id: 83a1534a-79e1-45db-b6f4-e4921183156c
Set id: 0c633ac9-7cc4-477d-8296-2a891ed4a867
Version: 5
Effective Time: 20161206
The Mentholatum Company
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.