Diabet Aid
Dosage form: lotion
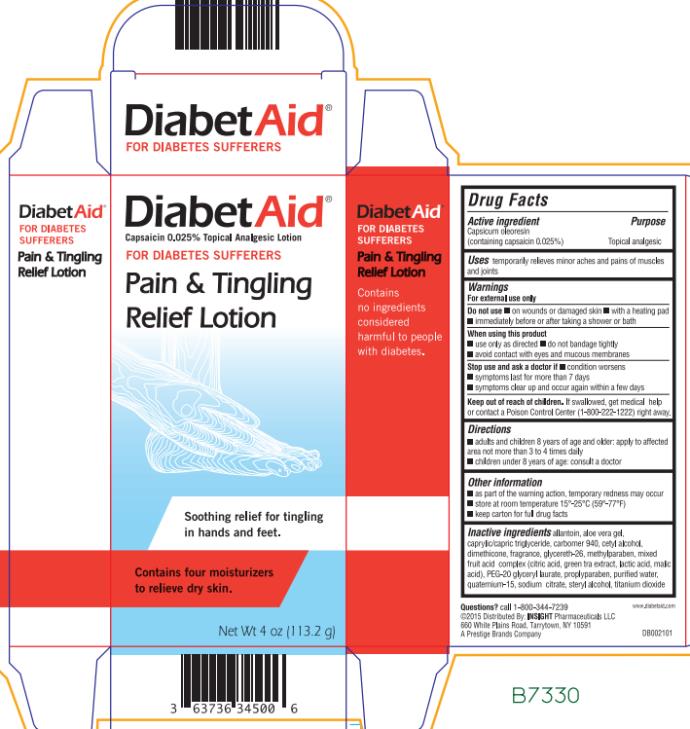
Ingredients: CAPSICUM OLEORESIN 0.25mg in 1g
Labeler: Insight Pharmaceuticals LLC
NDC code: 63736-404
Medically reviewed by Drugs.com. Last updated on Oct 14, 2024.
DIABET AID- capsicum oleoresin lotion
Diabet
Aid®
Drug Facts
Capsicum oleoresin (containing capsaicin 0.025%)
Topical analgesic
temporarily relieves minor aches and pains of muscles and joints
For external use only
- on wounds or damaged skin
- with a heating pad
- immediately before or after taking a shower or bath
- avoid contact with eyes
- do not bandage tightly
- do not apply to wounds or damaged skin
- condition gets worse
- symptoms last for more than 7 days
- symptoms clear up and occur again within a few days
If swallowed, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
| Adults and children 8 years of age and older | apply to affected area not more than 3 to 4 times daily |
| Children under 8 years of age | consult a doctor |
- as part of the warming action, temporary redness may occur
- store at room temperature 15° - 25°C (59° - 77°F)
- keep carton for full drug facts
allantoin, aloe vera gel, caprylic/capric triglyceride, carbomer, cetyl alcohol, dimethicone, fragrance, methylparaben, mixed fruit acid complex (citric acid, green tea extract, lactic acid, malic acid), PEG-20 glyceryl laurate, PEG-30 glyceryl laurate, propylparaben, purified water, quaternium-15, sodium citrate, stearyl alcohol, titanium dioxide.
call 1-800-344-7239
| DIABET AID
capsicum oleoresin lotion |
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
| Labeler - Insight Pharmaceuticals LLC (055665422) |
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.