Floriva Tablets: Package Insert / Prescribing Info
Package insert / product label
Generic name: sodium fluoride, multivitamins
Dosage form: chewable tablets
Drug class: Vitamin and mineral combinations
Medically reviewed by Drugs.com. Last updated on Feb 4, 2024.
On This Page
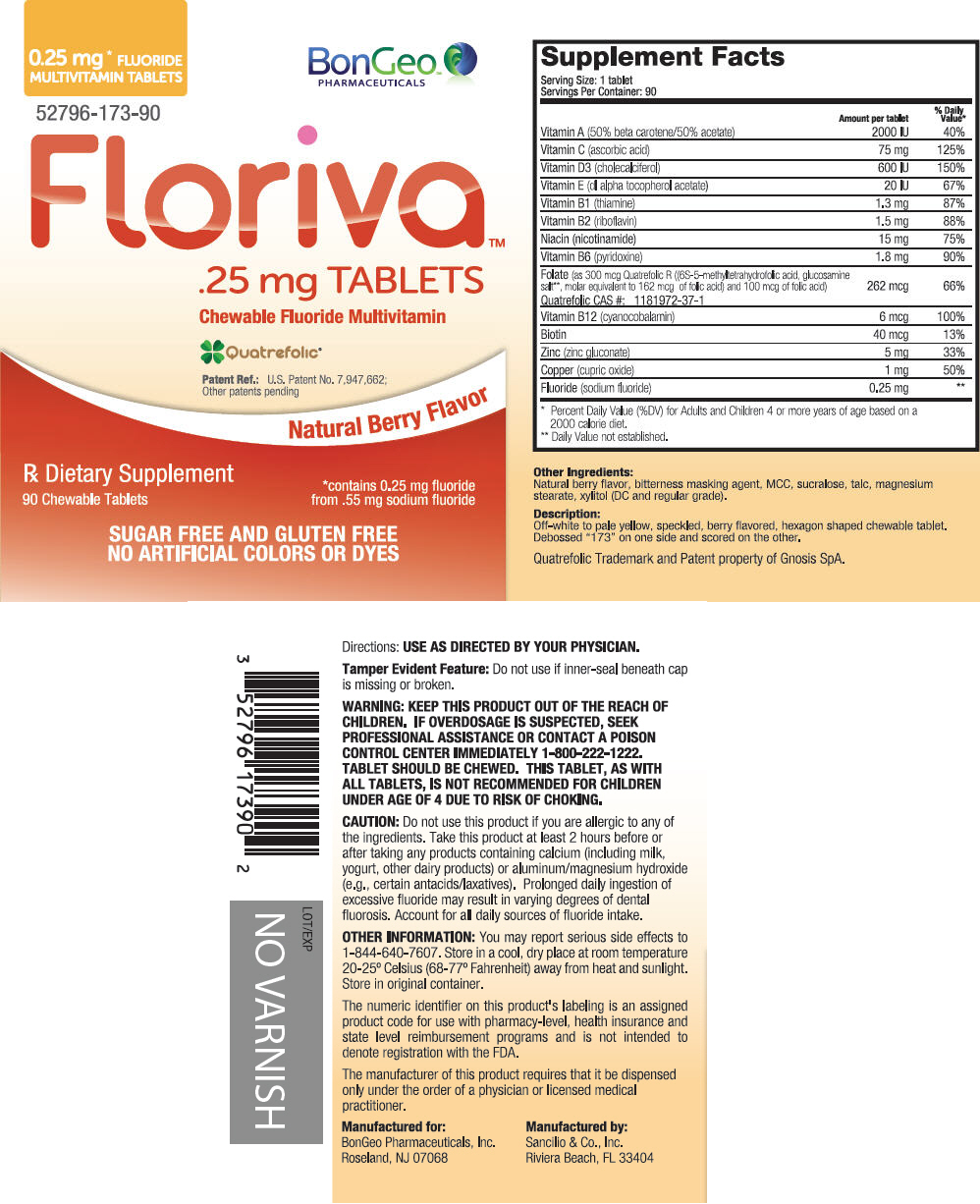
| Supplement Facts | ||
|---|---|---|
| Serving Size: 1 tablet Servings Per Container: 90 | ||
| Amount per tablet | % Daily Value* | |
| Vitamin A (50% beta carotene/50% acetate) | 2000 IU | 40% |
| Vitamin C (ascorbic acid) | 75 mg | 125% |
| Vitamin D3 (cholecalciferol) | 600 IU | 150% |
| Vitamin E (dl alpha tocopherol acetate) | 20 IU | 67% |
| Vitamin B1 (thiamine) | 1.3 mg | 87% |
| Vitamin B2 (riboflavin) | 1.5 mg | 88% |
| Niacin (nicotinamide) | 15 mg | 75% |
| Vitamin B6 (pyridoxine) | 1.8 mg | 90% |
| Folate (as 300 mcg Quatrefolic R ((6S-5-methyltetrahydrofolic acid, glucosamine salt†, molar equivalent to 162 mcg of folic acid) and 100 mcg of folic acid) Quatrefolic CAS #: 1181972-37-1 | 262 mcg | 66% |
| Vitamin B12 (cyanocobalamin) | 6 mcg | 100% |
| Biotin | 40 mcg | 13% |
| Zinc (zinc gluconate) | 5 mg | 33% |
| Copper (cupric oxide) | 1 mg | 50% |
| Fluoride (sodium fluoride) | 0.25 mg | † |
Other Ingredients
Natural berry flavor, bitterness masking agent, MCC, sucralose, talc, magnesium stearate, xylitol (DC and regular grade).
Floriva Tablets Description
Off-white to pale yellow, speckled, berry flavored, hexagon shaped chewable tablet. Debossed "173" on one side and scored on the other.
Quatrefolic Trademark and Patent property of Gnosis SpA.
Related/similar drugs
Warnings
KEEP THIS PRODUCT OUT OF THE REACH OF CHILDREN. IF OVERDOSAGE IS SUSPECTED, SEEK PROFESSIONAL ASSISTANCE OR CONTACT A POISON CONTROL CENTER IMMEDIATELY 1-800-222-1222. TABLET SHOULD BE CHEWED. THIS TABLET, AS WITH ALL TABLETS, IS NOT RECOMMENDED FOR CHILDREN UNDER AGE OF 4 DUE TO RISK OF CHOKING.
Precautions
Do not use this product if you are allergic to any of the ingredients. Take this product at least 2 hours before or after taking any products containing calcium (including milk, yogurt, other dairy products) or aluminum/magnesium hydroxide (e.g., certain antacids/laxatives). Prolonged daily ingestion of excessive fluoride may result in varying degrees of dental fluorosis. Account for all daily sources of fluoride intake.
Storage and Handling
You may report serious side effects to 1-844-640-7607. Store in a cool, dry place at room temperature 20-25° Celsius (68-77° Fahrenheit) away from heat and sunlight. Store in original container.
The numeric identifier on this product's labeling is an assigned product code for use with pharmacy-level, health insurance and state level reimbursement programs and is not intended to denote registration with the FDA.
The manufacturer of this product requires that it be dispensed only under the order of a physician or licensed medical practitioner.
Manufactured for:
BonGeo Pharmaceuticals, Inc.
Roseland, NJ 07068
Manufactured by:
Sancilio & Co., Inc.
Riviera Beach, FL 33404
PRINCIPAL DISPLAY PANEL - 90 Tablet Bottle Label
0.25 mg* FLUORIDE
MULTIVITAMIN TABLETS
BonGeo™
PHARMACEUTICALS
52796-173-90
Floriva™
.25 mg TABLETS
Chewable Fluoride Multivitamin
Quatrefolic®
Patent Ref.: U.S. Patent No. 7,947,662;
Other patents pending
Natural Berry Flavor
Rx Dietary Supplement
90 Chewable Tablets
*contains 0.25 mg fluoride
from .55 mg sodium fluoride
SUGAR FREE AND GLUTEN FREE
NO ARTIFICIAL COLORS OR DYES
| FLORIVA
vitamin a acetate, .beta.-carotene, ascorbic acid, cholecalciferol, .alpha.-tocopherol, thiamine, riboflavin, niacinamide, pyridoxine, levomefolate glucosamine, folic acid, cyanocobalamin, biotin, zinc gluconate, cupric oxide, and sodium fluoride tablet, chewable |
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - BonGeo Pharmaceuticals, Inc. (964822022) |
More about multivitamin with fluoride
- Check interactions
- Compare alternatives
- Pricing & coupons
- Drug images
- Side effects
- Dosage information
- Drug class: vitamin and mineral combinations
- En español
Patient resources
- Multivitamins with fluoride drug information
- Pediatric Multivitamin Chewables with Fluoride
- Pediatric Multivitamin Drops with Fluoride
Professional resources
- Multi Vitamin Fluoride Drops prescribing information
- Multi Vitamin with Fluoride (FDA)
- Multi-Vit with Fluoride Drops (FDA)
- MultiVit with Fluoride Chewable Tablets (FDA)
- Multivitamin with Fluoride Chewable Tablets (FDA)
- Vitamins A, C, D and Fluoride (FDA)
Other brands
MVC-Fluoride, TRI-VIT With Fluoride, Tri-Vite Drops with Fluoride

