"4810" Pill Images
The following drug pill images match your search criteria.

1 / 2
Loading
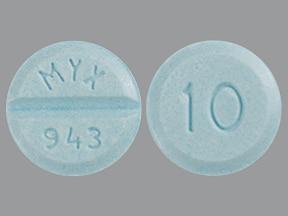
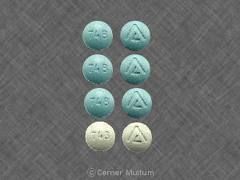
4810
1 / 3
Loading
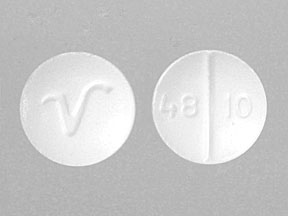
V 48 10
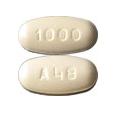
A48 1000

NIV-143 100 mg
Temozolomide
- Strength
- 100 mg
- Imprint
- NIV-143 100 mg
- Color
- Beige / Peach
- Shape
- Capsule/Oblong
Featured


Ozempic
Learn about Ozempic (semaglutide) for type 2 diabetes treatment, weight management, cardiovascular ...
Reviews & ratings

443 10/325
Acetaminophen and Oxycodone Hydrochloride
- Strength
- 325 mg / 10 mg
- Imprint
- 443 10/325
- Color
- White
- Shape
- Capsule/Oblong

1 / 5
Loading
BP 643 10
Acetaminophen and Hydrocodone Bitartrate
- Strength
- 300 mg / 10 mg
- Imprint
- BP 643 10
- Color
- White
- Shape
- Capsule/Oblong
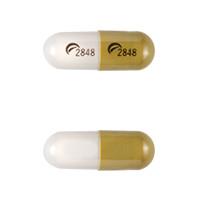
Logo (Actavis) 2848 Logo (Actavis) 2848
Fluvoxamine Maleate Extended-Release
- Strength
- 100 mg
- Imprint
- Logo (Actavis) 2848 Logo (Actavis) 2848
- Color
- Gray / Green
- Shape
- Capsule/Oblong
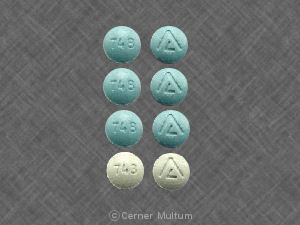
748 Logo
Previfem
- Strength
- ethinyl estradiol 0.035 mg / norgestimate 0.25 mg
- Imprint
- 748 Logo
- Color
- Blue
- Shape
- Round
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.