Ruzurgi User Reviews & Ratings
Ruzurgi has an average rating of 10.0 out of 10 from a total of 2 reviews on Drugs.com. 100% of reviewers reported a positive experience, while 0% reported a negative experience.
| Condition | Avg. Rating | Reviews | Compare |
|---|---|---|---|
| Lambert-Eaton Myasthenic Syndrome | 2 reviews for Lambert-Eaton Myasthenic Syndrome | 2 medications |
- Sue...
- Taken for 2 to 5 years
- April 29, 2020
For Lambert-Eaton Myasthenic Syndrome "For me, this drug was like a miracle drug. I could barely walk, and after a short time after taking the medicine, I walked just about normal."
See also:
Reviews may be edited to correct grammar/spelling or remove inappropriate content. Reviews appearing to come from parties with a vested interest are not published. This information is not intended to endorse any medication and should not replace the expertise and judgment of healthcare professionals.
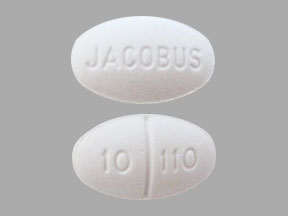
More about Ruzurgi (amifampridine)
- Check interactions
- Compare alternatives
- Reviews (2)
- Drug images
- Latest FDA alerts (1)
- Side effects
- Dosage information
- During pregnancy
- FDA approval history
- Drug class: cholinergic muscle stimulants
- Breastfeeding


For Lambert-Eaton Myasthenic Syndrome "I have been on Ruzurgi (which used to be called 3,4 DAP) since 2014, and it is a miracle drug. I could barely walk, and when I took this drug, I was walking nearly normal after 20 minutes. In 2018, Catalyst patented Firdapse, and we were forced to go on it in January 2019. I had so many side effects from Firdapse that I never had on Ruzurgi. Luckily for me, Ruzurgi got approved for children from ages 6-17, and my doctor can now prescribe it off-label. I was never so happy in my life to get back the Ruzurgi."