Nicomide: Package Insert / Prescribing Info
Package insert / product label
Generic name: niacinamide, folic acid, zinc oxide and cupric oxide
Dosage form: tablet
Drug class: Vitamin and mineral combinations
Medically reviewed by Drugs.com. Last updated on Mar 24, 2025.
On This Page
DESCRIPTION: Oval white tablet for oral administration of niacin, folate, zinc, and copper to supplement the diet. Tablets are debossed with “342” on one side and plain on the other.
| Supplement Facts
Serving Size: 1 Tablet | ||
| Each Tablet Contains: | % Daily Value | |
| Niacin (as niacinamide) | 750 mg | 3,750% |
| Folate (as 926 mcg Quatrefolic® ((6S)-5,- methyltetrahydrofolate glucosamine salt*) CAS 1181972-37-1) | 500 mcg | 125% |
| Zinc (as zinc oxide) | 25 mg | 167% |
| Copper (as cupric oxide) | 1.5 mg | 75% |
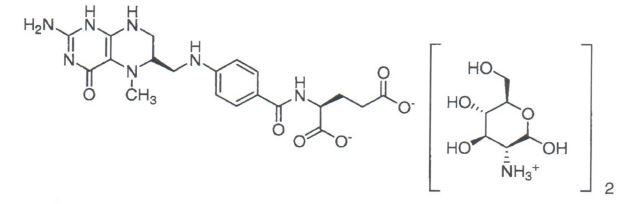
The chemical structure for Quatrefolic® is:
OTHER INGREDIENTS: Hypromellose, Microcrystalline cellulose, Magnesium Stearate, Stearic acid, Silica, and pharmaceutical glaze.
Quatrefolic® ((6S)-5,-methyltetrahydrofolate, glucosamine salt) is the fourth generation folate which has enhanced stability as well as high water solubility and bioavailability.
INDICATIONS: Nicomide® tablets are indicated for patients who are deficient in, or at risk of deficiency in, one or more of the components in this product.
CONTRAINDICATIONS: This product is contraindicated in patients with a known hypersensitivity to any of the ingredients.
WARNING: Folate alone is improper treatment of pernicious anemia and other megaloblastic anemias where Vitamin B12 is deficient.
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
PRECAUTIONS: Large dose of NICOMIDE® should be administered with caution in patients with a history of jaundice, liver disease or diabetes mellitus.
Folate above 0.1 mg daily may obscure pernicious anemia (hematologic remission may occur while neurological manifestations remain progressive).
Those with chronic liver failure and chronic renal failure should exercise extreme caution in the use of supplements containing copper.
If you are pregnant, nursing, or taking medication, consult your doctor before use.
Abnormal liver functions tests have been reported in persons taking daily doses of 500 mg or more of niacinamide. Folate intake should not exceed 250% of the Daily Value (1,000 mcg).
ADVERSE REACTIONS: Allergic sensitization has been reported rarely following oral and parental administration of Folate.
DOSAGE AND ADMINISTRATION: Usual adult dose is one tablet taken once or twice a day or prescribed by a physician.
| NICOMIDE
niacinamide, folic acid, zinc oxide and cupric oxide tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Avion Pharmaceuticals, LLC (965450542) |
Related/similar drugs
More about Nicomide (multivitamin with minerals)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Drug images
- Side effects
- Dosage information
- Drug class: vitamin and mineral combinations
Professional resources
Other brands
Dolomite, Bacmin, Centratex, Dialyvite Supreme D, ... +7 more