Gastromark: Package Insert / Prescribing Info
Package insert / product label
Generic name: ferumoxsil
Dosage form: oral suspension
Drug class: Magnetic resonance imaging contrast media
Medically reviewed by Drugs.com. Last updated on Mar 25, 2025.
On This Page
Gastromark Description
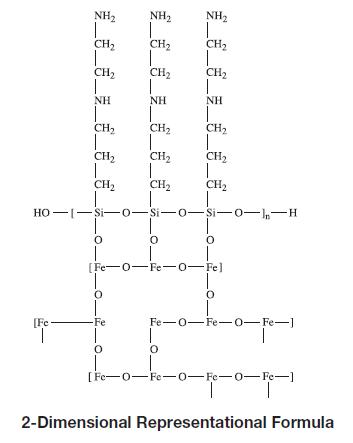
Gastromark™ (ferumoxsil, oral suspension) is an aqueous suspension of silicone-coated, superparamagnetic iron oxide, intended for oral administration as a magnetic resonance imaging contrast media. Gastromark is designated chemically as poly [N-(2-aminoethyl)-3-aminopropyl] siloxane coated non-stoichiometric magnetite (FeOx[C5H13N2SiO2]y), which has been manufactured to obtain a small uniform particle size of approximately 0.4 microns.
Gastromark is a turbid, slightly viscous, dark brown to orange-brown liquid formulation prepared for oral administration. The formulation contains water, sodium chloride, sorbitol, saccharin, carboxymethylcellulose, methylparaben, propylparaben, yellow dye #6, red dye #40, and flavoring. Each milliliter of Gastromark contains 175 micrograms of iron. Each milliliter also contains 1.4 milligrams of parabens as antimicrobial agents. The osmolality of the suspension is 250 mOsm; specific gravity is 1.01 grams per milliliter. The pH is 5.5 to 9.0, adjusted with sodium hydroxide.
The product is supplied in 360 mL bottles containing 300 mL of Gastromark.
Gastromark - Clinical Pharmacology
General
Gastromark is an oral aqueous suspension of a superparamagnetic magnetic resonance imaging (MRI) contrast agent. After oral administration of Gastromark, the agent fills the stomach and small intestine by 30 to 45 minutes after ingestion. The imaging agent passes distally to the large intestine by 4 to 7 hours after ingestion. Gastromark is primarily eliminated in the feces.
Pharmacokinetics
Absorption – Following the administration of 600 mL of Gastromark containing 10μCi of 59Fe (105 mg of iron) to 3 healthy, male volunteers and the same Gastromark dose containing 9.5μCi of 59Fe (105 mg of iron) to 3 male patients with inflammatory bowel diseases, the blood concentration of 59Fe in all subjects/patients for Days 1, 3, 7, and 14 after dosing were low (respectively, 0 to 0.024, 0 to 0.015, 0 to 0.058 and 0 to 0.044 nCi/mL). When normalized to a 70 kg man with 5000 mL of blood for Days 1, 3, 7, and 14 after dosing, these concentrations amount to 1.3%, 0.8%, 3.0% and 2.3% of the doses of 59Fe administered. This study is too small to reliably predict differences in absorption between healthy people and patients with inflammatory bowel disease. Absorption of Gastromark has not been studied in women.
Generally, the extent of absorption of iron from the gastrointestinal tract is expected to depend upon the existing body stores of iron. The lower the body iron stores, the more iron may be absorbed from Gastromark.
Distribution and Metabolism – Iron absorbed from Gastromark enters the hematopoietic pathway and is incorporated into the hemoglobin of the red blood cells or into ferritin for storage. The extent to which silicone is absorbed or metabolized in humans is not known.
Elimination – Unabsorbed iron in Gastromark is eliminated in feces. Based upon literature information, iron absorbed into the blood is highly conserved. In healthy normal adult men, about 10% of the body’s iron store is lost per year (1 mg per day). Iron is excreted from the gastrointestinal tract in extravasated red cells, in bile and in exfoliated mucosal cells. Small amounts of iron are lost in the urine and in desquamated skin. Additional iron loss occurs in menstrual flow in women.
Food Effect – Gastromark is intended for administration under fasting conditions. The effect of food on the disposition of Gastromark has not been studied.
Special Populations – Studies have not been conducted to evaluate the disposition of Gastromark in special populations such as patients with hepatic and renal diseases, or patients with hemochromatosis, hemosiderosis and other blood disorders.
The amount of iron absorbed in patients with active inflammatory bowel disease has not been studied adequately.
Drug-Drug Interactions – Drug interaction studies have not been conducted with Gastromark. The effect of iron supplements or drugs that increase or decrease gastrointestinal transit time on imaging with Gastromark has not been studied.
Clinical Trials
Gastromark was evaluated in a total of 256 people (63 normal volunteers and 193 patients). Of these, 141 were men and 115 were women. Racial demographics were: 225 Caucasian, 22 Black, 7 Asian, and 2 from other races.
Of the above, 186 patients were enrolled in two identical clinical studies of 93 subjects each. Patients were referred for magnetic resonance imaging (MRI) and had known or suspected abdominal masses. Gastromark was evaluated for its ability to enhance the bowel and improve the definition of anatomic markings in the MRI. Confirmation of the final diagnosis for lesions external to the bowel was not obtained.
The MRI images from the two studies were evaluated by blinded readers for bowel marking and the delineation of anatomy. The before and after Gastromark images were rated for the percent of images that were “better, same, worse, or not evaluable.” The table below lists the percentage of patients in each study with MRls in different regions of the abdomen that were “better” after Gastromark. After Gastromark, artifacts were rated as present (yes) and absent (no).
| Image Sequence | T1 | T2 | ||
|---|---|---|---|---|
|
* The mean volume of Gastromark administered was 699 mL (122 mg of iron). ** In comparison to Gastromark 30 minute images of the hepatic & splenic flexure and the sigmoid, in a subset of 30 patients there is a trend towards improved contrast at 4 to 7 hours; however, these findings have not been confirmed and the impact of dilution is not known. |
||||
| Study ID and number of patients | Study A N=93 | Study B N=93 | Study A N=93 | Study B N=93 |
|
IMAGING WITHIN 30 MINUTES OF GASTROMARK INGESTION** |
||||
|
Marking of the Bowel |
||||
|
Upper GI Overall Stomach Duodenal Sweep Jejunum & Ileum Lower GI Overall Hepatic & Splenic Flexure Sigmoid |
54 (58%) 44 (47%) 26 (28%) 38 (41%) 23 (25%) 18 (19%) 4 ( 4%) |
49 (53%) 58 (62%) 42 (45%) 52 (56%) 26 (28%) 25 (29%) 4 ( 4%) |
71 (77%) 51 (55%) 50 (54%) 58 (63%) 37 (40%) 23 (25%) 10 (11%) |
51 (55%) 48 (52%) 40 (43%) 68 (73%) 43 (46%) 41 (44%) 8 ( 9%) |
|
Delineation of Anatomic Features |
||||
|
Pancreas Anterior Kidney |
15 (16%) 6 ( 7%) |
32 (34%) 3 ( 3%) |
32 (34%) 15 (16%) |
28 (30%) 3 ( 3%) |
After Gastromark, depending upon the site evaluated, 11 to 70% of the patients had images rated “the same” as those without Gastromark; and 1 to 3% of the patients had images rated worse or not evaluable. After Gastromark, artifacts were seen in 1 to 3% of the MRI images.
The presence or absence of mass lesions was further evaluated in a subset of 67 of the 186 patients (36%), 32 with pancreatic/gastric masses and 35 with possible bowel obstruction. For these patients, in comparison to MRls before Gastromark and depending upon the anatomic location, the visualized abnormalities on MRI images after Gastromark increased confidence excluding the mass in 31 to 41% of the patients; increased confidence in delineating the mass in 44 to 49% of the patients; and in 16 to 26% of the patients the information was not helpful. Confirmation of findings or MRI images judged normal after Gastromark was not obtained. Gastromark was not evaluated for the contribution of the Gastromark MRI to the final diagnosis or patient management.
Indications and Usage for Gastromark
Gastromark™ is indicated in adult patients for oral use with magnetic resonance imaging to enhance the delineation of the bowel to distinguish it from organs and tissues that are adjacent to the upper regions of the gastrointestinal tract.
Gastromark’s usefulness in the lower gastrointestinal tract and retroperitoneal region is limited (by transit time and dilution).
Gastromark is not recommended for iron supplementation.
Contraindications
Like other large volume oral contrast agents, Gastromark is contraindicated in patients with known or suspected intestinal perforation or obstruction.
Gastromark is contraindicated in patients with known allergy to its active or inactive ingredients.
Warnings
The ingestion of Gastromark may cause abdominal pain, diarrhea, nausea and vomiting. In 78/256 (30%) of patients and normal volunteers, gastrointestinal adverse events occurred within the first 2 hours after ingestion of Gastromark. In 30/256 (12%), gastrointestinal events had their onset within 30 minutes. In 63/256 (24%), diarrhea occurred within 24 hours.
Vomiting can be associated with aspiration. Precautions should be taken to avoid aspiration.
The effects of Gastromark on human peritoneal tissues are not known. In animal studies, intraperitoneal injection of Gastromark was associated with a foreign body reaction that persisted for at least 30 days.
Precautions
General
Gastromark is associated with nausea, vomiting, diarrhea, and abdominal cramping. In patients who have these symptoms before Gastromark, the symptoms may increase in severity. This could confound the ability to distinguish adverse effects of Gastromark from the signs and symptoms of obstruction or perforation, and from the pre-existing conditions.
Patients who have a current or recent history of hiatal hernia, esophageal reflux, nausea, vomiting, or abdominal pain may not be able to tolerate Gastromark. Studies have not been conducted in these patients.
Gastromark should be given with caution to patients who cannot tolerate large fluid shifts and who are on specific fluid intake requirements.
The safety of Gastromark in patients with inflammatory bowel disease has not been well studied.
Studies have not been done to describe the effects of drugs that increase or decrease gastrointestinal transit time on the Gastromark image quality or on gastrointestinal adverse events.
Iron containing products should be used with caution in patients with disorders associated with iron overload (e.g., hemosiderosis, chronic hemolytic anemia with frequent blood transfusions, or chronic iron replacement).
Diagnostic procedures that involve the use of contrast agents should be carried out under the direction of a physician with the prerequisite training and a thorough knowledge of the procedure to be performed.
Information For Patients
Patients receiving Gastromark should be instructed to inform the physician and technologist if they are pregnant or nursing. Patients should be informed that:
Gastromark is prescribed for gastrointestinal tract enhancement during MRI.
Gastromark can cause significant nausea, vomiting, diarrhea or abdominal pain, and other intestinal discomfort. In patients who have these symptoms before ingesting Gastromark, the symptoms might increase in severity.
Patients should be asked if they are able to rapidly drink approximately 20 to 30 ounces (1/2 to 1 quart of fluid, or 600 to 900 mL) over a 15 to 30 minute period. They should be asked if they have hiatal hernias or problems with regurgitation when they lie on their backs after eating.
Laboratory Test Findings
Gastromark contains iron oxide. Although iron absorption from Gastromark is low, the extent of gastrointestinal absorption of any available elemental iron from Gastromark is expected to depend upon the patient’s existing iron stores. The lower the body iron stores, the more iron will be absorbed from the GI tract. The iron may change serum measurements of iron, iron binding,
transferrin, ferritin, and other studies which reflect iron metabolism. In patients with iron deficiency anemia, the extent to which the iron absorption may affect supplemental or parenteral iron dosing requirements is not known (see CLINICAL PHARMACOLOGY section for details on iron absorption).
Eosinophilia was noted in patients who received Gastromark.
Carcinogenesis, Mutagenesis and Impairment of Fertility
Long-term animal studies have not been performed to evaluate the carcinogenic potential of Gastromark. Gastromark was not found to cause genetic toxicity in the Ames test, in a chromosome aberration assay in CHO cells, in the CHO/HGPRT forward mutation assay, or in an unscheduled DNA synthesis assay in isolated rat hepatocytes. Gastromark has not been evaluated for potential to impair fertility.
Pregnancy Category B
Reproduction studies have been performed with Gastromark dosed by iron content. During gestation in rats and rabbits respectively, doses were up to 52.5 mg Fe/kg and 26.25 mg Fe/kg (respectively about 17 and 8 times the dose for a 50 kg human on a mg/kg basis, and 8 and 2.5 times on a mg/m2 basis). Maternal toxicity was not observed and these doses were not associated with evidence of harm to the fetus. Adequate and well-controlled studies have not been conducted in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Adverse Reactions/Side Effects
Gastromark was evaluated in 256 patients and normal volunteers in clinical trials. Of these, 147 (57%) had at least one adverse event. Deaths did not occur during the study period of 7 days. In 15/256 (6%) patients, the ingestion of Gastromark was either interrupted or stopped because of vomiting or because they could not ingest any more fluid. One normal volunteer had prolonged abdominal cramping for 4 hours. One patient had post-operative ileus 2 days after ingesting Gastromark.
There is a suggestion of differences in rates of adverse experiences in patients who have or do not have pancreatic or gastric masses. In patients with gastric or pancreatic masses, vomiting was reported in 4/35 (11%); in patients without gastric or pancreatic masses, vomiting was reported in 10/221 (5%) (p = 0.07). Abdominal pain was reported in 1/35 (3%) of patients with pancreatic or gastric masses and in 22/221 (10%) of patients without these lesions. The clinical relevance of these findings is not known.
The following table of adverse events is based upon clinical trials with Gastromark in 256 subjects.
| Adverse Event | Before Gastromark N = 241 | After Gastromark N = 256 |
|---|---|---|
|
* The mean volume of Gastromark administered was 701 mL (122.7 mg of iron). ** Includes bloating and gas. |
||
|
Patients with Any Adverse Event |
83 (34%) |
147 (57%) |
|
Body as a Whole |
0 (0%) |
25 (10%) |
|
Headache |
0 (0%) |
3 (1%) |
|
Digestive System |
80 (33%) |
130 (51%) |
|
Abdominal Pain/Cramps |
18 (8%) |
23 (9%) |
|
Diarrhea |
38 (16%) |
67 (26%) |
|
Dyspepsia |
14 (6%) |
16 (6%) |
|
Flatulence** |
41 (17%) |
55 (21%) |
|
Nausea |
18 (8%) |
43 (17%) |
|
Vomiting |
5 (2%) |
14 (6%) |
|
Nervous System |
0 (0%) |
44 (17%) |
|
Paresthesia, Oral |
0 (0%) |
44 (17%) |
|
Skin & Appendages |
0 (0%) |
3 (1%) |
|
Pruritus |
0 (0%) |
2 (0.8%) |
In the safety database of 256 people, the type of events was similar in people of each gender and in patients and healthy volunteers. The sample size was too small to determine whether there is any association of adverse events to body weight. However, for nausea and diarrhea, as shown in the following table, the rate of adverse events was much higher in women, both in patients and in volunteers.
| Adverse Event | Men | Women | ||
|---|---|---|---|---|
|
Healthy Volunteers |
Patients (N = 99) |
Healthy Volunteers |
Patients |
|
|
Nausea |
4 (10%) |
9 (9%) |
10 (48%) |
20 (21%) |
|
Diarrhea |
9 (21%) |
23 (23%) |
12 (57%) |
23 (25%) |
Other events reported in less than 0.5% of 256 healthy volunteers and patients who received Gastromark are: chills, fever, post-operative ileus, eructation, itching, urticarial rash, pustular rash, stomatitis, taste alteration, and edema of extremities. One healthy volunteer had prolonged abdominal cramping for 4 hours.
Related/similar drugs
Overdosage
Overdose has not been reported with Gastromark. If overdose occurs, it is likely that the toxicity would be related to iron, volume intolerance, or allergic-hypersensitivity reactions.
Gastromark Dosage and Administration
The recommended dosage of Gastromark is 600 mL (105 mg Fe) administered orally at a rate of about 300 mL over 15 minutes. The maximum oral dose is 900 mL (157.5 mg Fe).
Gastromark should be taken after fasting at least 4 hours.
Before use, the bottles should be shaken vigorously for at least 1 full minute to assure uniform suspension of contents. Chilling before ingestion improves palatability. Any unused portion should be discarded.
Gastromark contains 175 μg iron per milliliter. The amount of iron absorbed varies with the patient’s iron stores (see CLINICAL PHARMACOLOGY section).
Imaging: After ingestion of Gastromark, imaging may begin immediately. Generally, optimal results occur within 30 minutes. In some patients, delayed images (4 to 7 hours after ingestion) may be useful in delineating the lower gastrointestinal regions.
How is Gastromark supplied
Gastromark™ is packaged in white polyethylene bottles sealed with tamper-evident screw closures. Each bottle contains 300 mL of Gastromark.
Gastromark is supplied in boxes of twelve bottles each.
12 x 300 mL bottle .....NDC 0019-1120-06
Gastromark is distributed by Mallinckrodt Inc., Raleigh, NC, under license from AMAG Pharmaceuticals, Inc., Cambridge, MA.
Gastromark is a trademark of Mallinckrodt Inc.
Storage: Store at controlled room temperature or refrigerate between 2° to 25°C (36° to 77°F). Protect from freezing. The product should be discarded if frozen.
The following patents have claims directed to the drug: USP 4,695,392, USP 4,695,393, USP 4,770,183, USP 4,827,945, USP 4,951,675, USP 5,055,288, USP 5,069,216, USP 5,219,554.
tyco
Healthcare
MALLINCKRODT
Mallinckrodt Inc.
St. Louis, MO 63042 USA
www.Mallinckrodt.com
MKR 11200210
Revised 02/10
Printed in U.S.A.
PACKAGE LABEL - PRINCIPAL DISPLAY PANEL - 300 mL Bottle
Gastromark™
0019-1120-06
(ferumoxsil oral suspension)
300 mL, 175 μg Fe/mL
For Oral Use Only
Rx Only
Before use, shake vigorously for at least one full minute.
Store between: 2° to 25°C (36° to 77°F)
May be stored at controlled room temperature or refrigerated. Do not freeze. Discard product if frozen.
Ingredients: Each mL contains 175 μg inorganic iron from silicone-coated superparamagnetic iron oxide; sodium hydroxide to adjust pH; sodium chloride to adjust osmolality.
Inactive ingredients: Carboxymethylcellulose sodium, methylparaben, orange color, orange flavor, propylparaben, saccharin sodium, sorbitol solution.
Usual Dosage: The recommended dosage is 600 mL.
Manufactured by:
Mallinckrodt Inc.
St. Louis, MO 63042 USA
www.Mallinckrodt.com
10850210
Under License from:
AMAG Pharmaceuticals, Inc.
Cambridge, MA 02138
tyco
Healthcare
MALLINCKRODT
| GASTROMARK
ferumoxsil suspension |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| Labeler - Mallinckrodt Inc. (047021092) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Mallinckrodt Inc.-Pharmaceuticals Group | 097722284 | analysis, manufacture | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| AMAG Pharmaceuticals, Inc. | 017511155 | api manufacture | |

