Praxbind Dosage
Generic name: IDARUCIZUMAB 50mg in 1mL
Dosage form: injection
Drug class: Anticoagulant reversal agents
Medically reviewed by Drugs.com. Last updated on Jan 23, 2024.
Recommended Dose
The recommended dose of PRAXBIND is 5 g, provided as two separate vials each containing 2.5 g/50 mL idarucizumab (see Figure 1). Both vials are packaged together in one carton.
For intravenous use only.
There is limited data to support administration of an additional 5 g of PRAXBIND.
Preparation
- Remove both vials (each containing 2.5 g/50 mL idarucizumab) from carton.
- Ensure aseptic handling when preparing the infusion.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
- Once solution has been removed from the vial, administration should begin promptly. The solution in vials may be stored at room temperature, 25°C (77°F), but must be used within 6 hours.
Administration
- Do not mix with other medicinal products. Use aseptic technique when administering PRAXBIND.
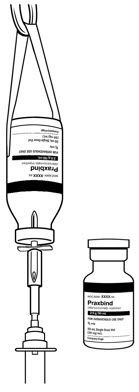
- Intravenously administer the dose of 5 g (2 vials, each contains 2.5 g) as
- A pre-existing intravenous line may be used for administration of PRAXBIND. The line must be flushed with sterile 0.9% Sodium Chloride Injection, USP solution prior to infusion. No other infusion should be administered in parallel via the same intravenous access.
- PRAXBIND treatment can be used in conjunction with standard supportive measures, which should be considered as medically appropriate.
 |
 |
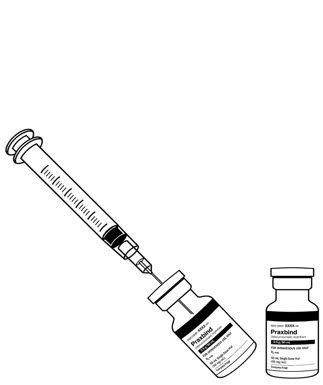 |
| Figure 1 Recommended dose of PRAXBIND provided as two vials. | Figure 2 Two consecutive infusions by hanging vials. | Figure 3 Inject both vials consecutively via syringe. |
Restarting Antithrombotic Therapy
Patients being treated with dabigatran therapy have underlying disease states that predispose them to thromboembolic events. Reversing dabigatran therapy exposes patients to the thrombotic risk of their underlying disease. To reduce this risk, resumption of anticoagulant therapy should be considered as soon as medically appropriate.
Idarucizumab is a specific reversal agent for dabigatran, with no impact on the effect of other anticoagulant or antithrombotic therapies.
Pradaxa treatment can be initiated 24 hours after administration of PRAXBIND.
More about Praxbind (idarucizumab)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Side effects
- During pregnancy
- FDA approval history
- Drug class: anticoagulant reversal agents
- Breastfeeding
- En español
Patient resources
Professional resources
Related treatment guides
See also:
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.

