FEIBA NF Dosage
Generic name: ANTI-INHIBITOR COAGULANT COMPLEX 1000U in 20mL;
Dosage form: injection
Medically reviewed by Drugs.com. Last updated on Jan 6, 2025.
For intravenous use after reconstitution only.
Dose
A guide for dosing FEIBA is provided in Table 1.
| Dose (unit/kg) | Frequency of Doses (hours) | Duration of Therapy | |
|---|---|---|---|
| Control and Prevention of Bleeding | |||
| Joint Hemorrhage | 50 – 100 | 12 | Until pain and acute disabilities are improved. |
| Mucous Membrane Bleeding | 50 – 100 | 6 | At least 1 day or until bleeding is resolved. |
| Soft Tissue Hemorrhage (e.g., retroperitoneal bleeding) |
100 | 12 | Until resolution of bleed. |
| Other Severe Hemorrhage (e.g., CNS bleeds) |
100 | 6 – 12 | Until resolution of bleed. |
| Perioperative Management | |||
| Preoperative | 50 – 100 | One time dose | Immediately prior to surgery. |
| Postoperative | 50 – 100 | 6 – 12 | Until resolution of bleed and healing are achieved. |
| Routine Prophylaxis | |||
| 85 | Every other day | ||
- Dosage and duration of treatment depend on the location and extent of bleeding, and the patient's clinical condition. Careful monitoring of replacement therapy is necessary in cases of major surgery or life-threatening bleeding episodes.
- Each vial of FEIBA contains the labeled amount of factor VIII inhibitor bypassing activity in units.
- Base the dose and frequency of FEIBA on the individual clinical response. Clinical response to treatment with FEIBA may vary by patient and may not correlate with the patient's inhibitor titer.
- Record the name of the patient and batch number of the product in order to maintain a link between the patient and the batch of the product.
- Do not exceed a single dose of 100 units per kg body weight or a daily dose of 200 units per kg body weight.
Preparation and Reconstitution
- Use aseptic technique throughout the entire reconstitution process.
- If the patient uses more than one vial per injection, reconstitute each vial according to the following instructions.
- Allow the vials of FEIBA and Sterile Water for Injection (diluent) to reach room temperature, if refrigerated.
- Remove the plastic caps from the concentrate and diluent vials.
- Wipe the stoppers of both vials with a sterile alcohol swab and allow them to dry prior to use.
- Open the package of BAXJECT II Hi-Flow device by peeling away the lid completely without touching the inside (Fig. A). Do not remove the device from the package. Do not touch the clear spike.
- Place the diluent vial on a flat and solid surface. Turn the package over and insert the clear plastic spike through the diluent stopper by pressing straight down (Fig. B).
- Grip the BAXJECT II Hi-Flow device package at the edges and pull the package off the device (Fig. C). Do not remove the blue protective cap from the BAXJECT II Hi-Flow device. Do not touch the purple spike.
- Turn the system over, so that the diluent vial is on top. Quickly insert the purple spike of the BAXJECT II Hi-Flow device fully into the FEIBA vial. The vacuum will draw the diluent into the FEIBA vial (Fig. D). The connection of the two vials should be done expeditiously to close the open fluid pathway created by the first insertion of the spike to the diluent vial.
- Gently swirl (do not shake) the vial until FEIBA is completely dissolved. Make sure that FEIBA has been dissolved completely; otherwise, active material will not pass through the device filter. The reconstituted solution should be inspected visually for particulate matter before administration. The solution should be discarded if it is not clear or is discolored.
- After reconstitution of FEIBA is complete, the injection or infusion should begin immediately and must be completed within three hours following reconstitution. Do not refrigerate after reconstitution. Discard unused portion.
Figure A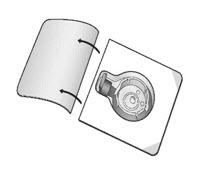 |
Figure B |
Figure C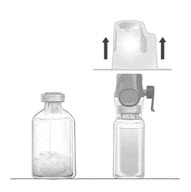 |
Figure D |
Administration
For intravenous injection or intravenous infusion after reconstitution only.
- Inspect the reconstituted FEIBA solution visually for particulate matter and discoloration prior to administration. The appearance of the solution should be colorless to slightly yellowish. Do not use if particulate matter or discoloration is observed.
- Flush venous access lines with isotonic saline prior to and after infusion of FEIBA. Do not administer in the same tubing or container with other medicinal products.
- Use plastic Luer lock syringes because protein such as FEIBA tends to stick to the surface of all-glass syringes.
- Remove the blue protective cap from the BAXJECT II Hi-Flow device. Tightly connect the syringe to the BAXJECT II Hi-Flow device (DO NOT DRAW AIR INTO THE SYRINGE) by turning the syringe in clockwise direction until stop position. Use of a Luer lock syringe is highly recommended to ensure a tight connection between the syringe and the BAXJECT II Hi-Flow device (Fig. E).
- Invert the system so that the dissolved FEIBA product is on top. Draw the dissolved product carefully into the syringe by pulling the plunger back slowly to avoid foaming (Fig. F).
- Ensure that the tight connection between the BAXJECT II Hi-Flow device and the syringe is maintained.
- Disconnect the syringe.
- Attach a suitable needle and inject or infuse intravenously at a rate that does not exceed 10 units per kg of body weight per minute. A syringe pump may be used to control the rate of administration. For a patient with a body weight of 75 kg, this corresponds to an infusion rate up to 15 mL per minute.
Figure E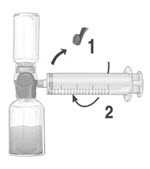 |
Figure F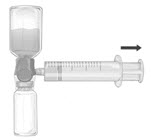 |
More about Feiba NF (anti-inhibitor coagulant complex)
Patient resources
Other brands
Professional resources
See also:
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.

